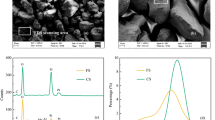
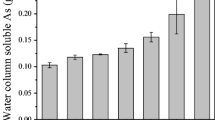
Abstract
The vadose zone acts the last barrier to block radionuclides from entering groundwater, where the adsorption of americium(III) (Am(III)) by natural colloids determines the transport of Am(III) in the vadose zone. However, there are few studies on natural colloids as the third phase in traditional static batch experiments. This study explored the effects of pH (4, 7, 10), electrolyte concentration (CNaCl = 0, 0.1, 5 mM), initial Am(III) concentration (6.54 × 10−10, 1.31 × 10−9, 1.96 × 10−9 M), and natural colloids concentration (300, 400, 500 mg/L) on the adsorption process in Am(III)-sediment (AS) and Am(III)-sediment-natural colloids (ASN) systems. The adsorption process was well fitted by pseudo-second-order kinetic model, indicating mainly a rate-limiting process controlled by chemical reactions. The adsorption percentage (< 70%) of Am(III) in AS was much smaller than that in ASN (> 90%), suggesting that natural colloids significantly affected the adsorption of Am(III) in the vadose zone sediments. Higher pH and lower electrolyte concentration resulted in larger adsorption percentages, because the negative charge on solid phase increased and the Am(III) species distribution changed in aqueous solution. The increase in initial Am(III) concentration can enhance the adsorption percentage, but the adsorption percentage did not rise proportionally with the increase in natural colloid concentration, which may be caused by the presence of critical colloid concentration in the adsorption system. This study provides a basis for evaluating the radioactive nuclide transport in high-risk sites.






Similar content being viewed by others
Data Availability
Data will be made available on request.
Abbreviations
- Am(III):
-
Americium(III)
- AS:
-
Americium(III)-sediment system
- ASN:
-
Americium(III)-sediment-natural colloid system
- NC:
-
Natural colloids
- SEM:
-
Scanning electron microscopy
- XRD:
-
X-ray diffraction
References
Anderson, E. B., Rogozin, Y. M., Smirnova, E. A., Bryzgalova, R. V., Andreeva, N. R., Malimonova, S. I., Shabalev, S. I., & Tochiyama, A. F. (2007). Sorption-barrier properties of granitoids and andesite-basaltic metavolcanites with respect to Am(III) and Pu(IV): 1. Absorption of Am and Pu from groundwater on monolithic samples of granitoids and andesite-basaltic metavolcanites. Radiochemistry, 49, 305–312. https://doi.org/10.1134/S1066362207030186
Baeyens, B., & Bradbury, M. H. (1997). A mechanistic description of Ni and Zn sorption on Na-montmorillonite Part I: titration and sorption measurements. Journal of Contaminant Hydrology, 27, 199–222.
Chen, C., Zhao, D., Tan, X., & Wang, X. (2007). Adsorption kinetic, thermodynamic and desorption studies of Th(IV) on oxidized multi-wall carbon nanotubes. Colloids and Surfaces A Physicochemical and Engineering Aspects, 302, 449–454. https://doi.org/10.1016/j.colsurfa.2007.03.007
Choppin, G. R. (2007). Actinide speciation in the environment. Journal of Radioanalytical and Nuclear Chemistry, 273, 695–703. https://doi.org/10.1007/s10967-007-0933-3
Das, D. K., Pathak, P. N., Kumar, S., & Manchanda, V. K. (2009). Sorption behavior of Am 3+ on suspended pyrite. Journal of Radioanalytical and Nuclear Chemistry, 281, 449–455. https://doi.org/10.1007/s10967-009-0030-x
Dong, W., Wang, X., Du, J., Bian, X., Ma, F., & Tao, Z. (1999). Sorption and desorption characteristics of Eu(III) on red earth. Journal of Radioanalytical and Nuclear Chemistry, 242, 793–797. https://doi.org/10.1007/bf02347397
Dong, W., Wang, X., Bian, X., Wang, A., Du, J., & Tao, Z. (2001). Comparative study on sorption/desorption of radioeuropium on alumina, bentonite and red earth: effects of pH, ionic strength, fulvic acid, and iron oxides in red earth. Applied Radiation and Isotopes : Including Data, Instrumentation and Methods for Use in Agriculture, Industry and Medicine, 54, 603–610. https://doi.org/10.1016/s0969-8043(00)00311-0
El-Sofany, E. A., Zaki, A. A., & Mekhamer, H. S. (2009). Kinetics and thermodynamics studies for the removal of Co2+ and Cs+ from aqueous solution by sand and clay soils. Radiochimica Acta, 97, 23–32. https://doi.org/10.1524/ract.2009.1573
Fan, Q., Shao, D., Lu, Y., Wu, W., & Wang, X. (2009). Effect of pH, ionic strength, temperature and humic substances on the sorption of Ni(II) to Na–attapulgite. Chemical Engineering Journal, 150, 188–195. https://doi.org/10.1016/j.cej.2008.12.024
Fan, Q., Zhang, M., Zhang, Y., Ding, K., Yang, Z., & Wu, W. (2010). Sorption of Eu(III) and Am(III) on attapulgite: effect of pH, ionic strength and fulvic acid. Radiochimica Acta, 98, 19–25. https://doi.org/10.1524/ract.2010.1680
Fuks, L., Oszczak, A., Dudek, J., Majdan, M., & Trytek, M. (2016). Removal of the radionuclides from aqueous solutions by biosorption on the roots of the dandelion (Taraxacum officinale). International Journal of Environmental Science and Technology, 13, 2339–2352. https://doi.org/10.1007/s13762-016-1067-3
Gao, P., Zhang, D., Jin, Q., Chen, Z., Wang, D., Guo, Z., & Wu, W. 2021. Multi-scale study of Am(III) adsorption on Gaomiaozi bentonite: combining experiments, modeling and DFT calculations, Chemical Geology, 581. https://doi.org/10.1016/j.chemgeo.2021.120414
Hamed, M. M., Aly, M. I., & Nayl, A. A. (2015). Kinetics and thermodynamics studies of cobalt, strontium and caesium sorption on marble from aqueous solution. Chemistry and Ecology, 32, 68–87. https://doi.org/10.1080/02757540.2015.1112379
Hormann, V. (2021). A consistent model for estimating the partitioning of Am, Pu and Se in agricultural soils. Journal of Radioanalytical and Nuclear Chemistry, 329, 769–784. https://doi.org/10.1007/s10967-021-07839-0
Iijima, K., Shoji, Y., & Tomura, T. (2010a). Sorption behavior of americium onto bentonite colloid. Radiochimica Acta, 96, 721–730. https://doi.org/10.1524/ract.2008.1558
Iijima, K., Tomura, T., Tobita, M., & Suzuki, Y. (2010b). Distribution of Cs and Am in the solution-bentonite colloids-granite ternary system: effect of addition order and sorption reversibility. Radiochimica Acta, 98, 729–736. https://doi.org/10.1524/ract.2010.1774
Ikaheimoinen, T. K. (2002). Transuranic elements in fishes compared to 137Cs in certain lakes in Finland. Boreal Environment Research, 7, 99–104.
Joseph, C., Schmeide, K., Sachs, S., Brendler, V., Geipel, G., & Bernhard, G. (2011). Sorption of uranium(VI) onto Opalinus Clay in the absence and presence of humic acid in Opalinus Clay pore water. Chemical Geology, 284, 240–250. https://doi.org/10.1016/j.chemgeo.2011.03.001
Lee, M. H., Jung, E. C., Song, K., Han, Y. H., & Shin, H. S. (2010). The influence of humic acid on the pH-dependent sorption of americium(III) onto kaolinite. Journal of Radioanalytical and Nuclear Chemistry, 287, 639–645. https://doi.org/10.1007/s10967-010-0899-4
Li, J., Hu, J., Sheng, G., Zhao, G., & Huang, Q. (2009). Effect of pH, ionic strength, foreign ions and temperature on the adsorption of Cu(II) from aqueous solution to GMZ bentonite. Colloids and Surfaces A Physicochemical and Engineering Aspects, 349, 195–201. https://doi.org/10.1016/j.colsurfa.2009.08.018
Li, P., Liu, Z., Ma, F., Shi, Q., Guo, Z., & Wu, W. (2015). Effects of pH, ionic strength and humic acid on the sorption of neptunium(V) to Na-bentonite. Journal of Molecular Liquids, 206, 285–292. https://doi.org/10.1016/j.molliq.2015.02.014
Lützenkirchen, J. (2012). Summary of studies on (ad)sorption as a “well-established” process within FUNMIG activities. Applied Geochemistry, 27, 427–443. https://doi.org/10.1016/j.apgeochem.2011.09.012
Magal, E., Weisbrod, N., Yechieli, Y., Walker, S. L., & Yakirevich, A. (2011). Colloid transport in porous media: impact of hyper-saline solutions. Water Research, 45, 3521–3532. https://doi.org/10.1016/j.watres.2011.04.021
Missana, T., & Garca-Gutierrez, M. (2007). Adsorption of bivalent ions (Ca(II), Sr(II) and Co(II)) onto FEBEX bentonite. Physics and Chemistry of the Earth Parts A/B/C, 32, 559–67. https://doi.org/10.1016/j.pce.2006.02.052
Missana, T., Alonso, U., García-Gutiérrez, M., & Mingarro, M. (2008). Role of bentonite colloids on europium and plutonium migration in a granite fracture. Applied Geochemistry, 23, 1484–1497. https://doi.org/10.1016/j.apgeochem.2008.01.008
Missana, T., & Geckeis, H. (2006). The CRR Final Project Report Series II: Supporting laboratory experiments with radionuclides and bentonite colloids. Switzerland.
Nawaz, S., Bhatti, H. N., Bokhari, T. H., & Sadaf, S. (2013). Removal of Novacron Golden Yellow dye from aqueous solutions by low-cost agricultural waste: batch and fixed bed study. Chemistry and Ecology, 30, 52–65. https://doi.org/10.1080/02757540.2013.841898
Paatero, J., & Jaakkola, T. (1998). Transfer of plutonium, americium and curium from fallout into reindeer after the Chernobyl accident, Boreal Environment Research, 3(2).
Pan, N., Deng, J., Guan, D., Jin, Y., & Xia, C. (2013). Adsorption characteristics of Th(IV) ions on reduced graphene oxide from aqueous solutions. Applied Surface Science, 287, 178–183. https://doi.org/10.1016/j.apsusc.2013.10.004
Ramirez-Guinart, O., Vidal, M., & Rigol, A. (2016). Univariate and multivariate analysis to elucidate the soil properties governing americium sorption in soils. Geoderma, 269, 19–26. https://doi.org/10.1016/j.geoderma.2016.01.026
Ramirez-Guinart, O., Kaplan, D., Rigol, A., & Vidal, M. (2020). Deriving probabilistic soil distribution coefficients (K(d)). Part 3: Reducing variability of americium K(d) best estimates using soil properties and chemical and geological material analogues. Journal of Environmental Radioactivity, 223–224, 106378. https://doi.org/10.1016/j.jenvrad.2020.106378
Ramírez-Guinart, O., Salaberria, A., Vidal, M., & Rigol, A. (2017). Assessing soil properties governing radiosamarium sorption in soils: can trivalent lanthanides and actinides be considered as analogues? Geoderma, 290, 33–39. https://doi.org/10.1016/j.geoderma.2016.12.010
Righetto, L., Bidoglio, G., Azimonti, G., & Bellobono, I. R. (1991). Competitive actinide interactions in colloidal humic acid-mineral oxide systems. Environmental Science and Technology, 25, 1913–19. https://doi.org/10.1021/es00023a012
Salminen, S., Paatero, J., Jaakkola, T., & Lehto, J. (2005). Americium and curium deposition in Finland from the Chernobyl accident. Radiochimica Acta, 93, 771–779. https://doi.org/10.1524/ract.2005.93.12.771
Schuessler, W., Artinger, R., Kienzler, B., & Kim, J. I. (2000). Conceptual modeling of the humic colloid-borneAmericium(III) migration by a kinetic approach. Environmental Science and Technology, 34, 2608–2611. https://doi.org/10.1021/es991246a
Sun, Y., Pan, D., Wei, X., Xian, D., Wang, P., Hou, J., Xu, Z., Liu, C., & Wu, W. (2020). Insight into the stability and correlated transport of kaolinite colloid: effect of pH, electrolytes and humic substances. Environmental Pollution, 266, 115189. https://doi.org/10.1016/j.envpol.2020.115189
Sun, Y., Zhang, Z., Heng, J., Gao, C., Jin, Q., Chen, Z., & Guo, Z. (2022). Co-transport of U(VI) and colloidal biochar in quartz sand heterogeneous media. Science of the Total Environment, 816, 151606. https://doi.org/10.1016/j.scitotenv.2021.151606
Um, W., & Serne, R. J. (2005). Sorption and transport behavior of radionuclides in the proposed low-level radioactive waste repository at the Hanford site, Washington. Radiochimica Acta, 93, 57–63. https://doi.org/10.1524/ract.93.1.57.58295
Vlasova, I., Petrov, V. G., Kuzmenkova, N., Kashtanov, A., & Hammer, J. (2017). Sorption of radionuclides on the rocks of the exocontact zone of Nizhnekansky granitoid massif. MRS Advances, 1, 4061–4067. https://doi.org/10.1557/adv.2017.190
Wang, X., Dong, W., Gong, Y., Wang, C., & Tao, Z. (2001). Sorption characteristics of radioeuropium on bentonite and kaolinite. Journal of Radioanalytical and Nuclear Chemistry, 250, 267–270. https://doi.org/10.1023/A:1017922924421
Wang, X., Du, J., Tao, Z., & Li, Z. (2004). Evaluation of Eu(III) migration in compacted bentonite. Journal of Radioanalytical and Nuclear Chemistry, 260, 69–73. https://doi.org/10.1023/b:jrnc.0000027063.55710.8a
Wang, S., Dong, Y., He, M., Chen, L., & Yu, X. (2009). Characterization of GMZ bentonite and its application in the adsorption of Pb(II) from aqueous solutions. Applied Clay Science, 43, 164–171. https://doi.org/10.1016/j.clay.2008.07.028
Wang, Y., Zhang, Z., Li, Q., & Liu, Y. (2012). Adsorption of uranium from aqueous solution using HDTMA+-pillared bentonite: isotherm, kinetic and thermodynamic aspects. Journal of Radioanalytical and Nuclear Chemistry, 293, 231–239. https://doi.org/10.1007/s10967-012-1659-4
Wu, W., Fan, Q., Xu, J., Niu, Z., & Lu, S. (2007). Sorption-desorption of Th(IV) on attapulgite: effects of pH, ionic strength and temperature. Applied Radiation and Isotopes, 65, 1108–1114. https://doi.org/10.1016/j.apradiso.2007.05.009
Wu, T., Yang, Z., Hu, R., & Chen, Y. (2023). Three-dimensional visualization reveals pore-scale mechanisms of colloid transport and retention in two-phase flow. Environmental Science & Technology, 57, 1997–2005. https://doi.org/10.1021/acs.est.2c08757
Xie, J., Lin, J., Zhou, X., Li, M., & Zhou, G. (2014a). Plutonium partitioning in three-phase systems with water, colloidal particles, and granites: new insights into distribution coefficients. Chemosphere, 99, 125–133. https://doi.org/10.1016/j.chemosphere.2013.10.049
Xie, J., Lin, J., Zhou, X., Li, M., & Zhou, G. (2014b). Plutonium partitioning in three-phase systems with water, granite grains, and different colloids. Environmental Science and Pollution Research International, 21, 7219–7226. https://doi.org/10.1007/s11356-014-2649-6
Xu, Z., Niu, Z., Pan, D., Zhao, X., Wei, X., Li, X., Tan, Z., Chen, X., Liu, C., & Wu, W. (2021). Mechanisms of bentonite colloid aggregation, retention, and release in saturated porous media: role of counter ions and humic acid. Science of the Total Environment, 793, 148545. https://doi.org/10.1016/j.scitotenv.2021.148545
Yang, S., Li, J., Lu, Y., Chen, Y., & Wang, X. (2009). Sorption of Ni(II) on GMZ bentonite: effects of pH, ionic strength, foreign ions, humic acid and temperature. Applied Radiation and Isotopes : Including Data, Instrumentation and Methods for Use in Agriculture, Industry and Medicine, 67, 1600–1608. https://doi.org/10.1016/j.apradiso.2009.03.118
Ye, Y. (2014). Studies on U(VI), Th(IV) and Eu(III) adsorption on SiO2, TiO2 and bentonite in presence of fulvic acid, Lanzhou University.
Zhang, H., Chen, L., Zhang, L., & Yu, X. (2010). Impact of environmental conditions on the adsorption behavior of radionuclide Ni(II) onto hematite. Journal of Radioanalytical and Nuclear Chemistry, 287, 357–365. https://doi.org/10.1007/s10967-010-0686-2
Zhang, H., Tong, Z., Wei, T., & Tang, Y. (2012). Sorption characteristics of Pb(II) on alkaline Ca-bentonite. Applied Clay Science, 65–66, 21–23. https://doi.org/10.1016/j.clay.2012.06.010
Acknowledgements
We are grateful for the support provided by the China Institute for Radiation Protection for this experiment.
Funding
The funding for this study was provided by the National Natural Science Foundation of China (42177201).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, K., Zhu, J., Gao, H. et al. Influence of Natural Colloids on the Kinetic Adsorption Behavior of Am(III) on Vadose Zone Sediments. Water Air Soil Pollut 235, 210 (2024). https://doi.org/10.1007/s11270-024-07019-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-07019-2