Abstract
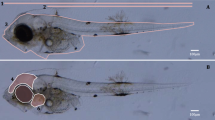
Amoxicillin (AMX) is an antibiotic that has been added to the watch list of substances by the European Union, through the Water Framework Directive (WFD; 2000/60/EC) and its daughter regulation (Decision 2018/840) for which monitoring data have to be gathered with a possibility for future regulation. Previous studies have demonstrated that this antibiotic generates toxic effects, among which oxidative stress in aquatic organisms is noteworthy. The aim of this study was to evaluate the effect on embryonic development and the teratogenic effects induced by AMX at environmentally relevant concentrations in oocytes and embryos of Danio rerio. Furthermore, oxidative stress biomarkers were evaluated at 72 and 96 hpf. The LC50 was 14.192 μgL−1, EC50 was 7.083 μgL−1, and TI was 2.003. Biomarkers of cellular oxidation and antioxidant enzymes were modified in a concentration-dependent way with respect to the control group (p<0.05). The main malformations identified were tail malformation, pericardial edema, yolk sac malformation, scoliosis, pectoral fin absence, and no hatching. The results allow us to conclude that AMX at environmentally relevant concentrations is capable of inducing embryotoxic and teratogenic effects and oxidative damage. This compound represents a risk to aquatic organisms such as Danio rerio.
Clinical Trials Registration
Not applicable.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Aderem, A., & Underhill, D. M. (1999). Mechanisms of phagocytosis in macrophages. Annual Review of Immunology, 17, 593–623. https://doi.org/10.1146/annurev.immunol.17.1.593.
Agathokleous, E., Kitao, M., & Calabrese, E. J. (2018). Human and veterinary antibiotics induce hormesis in plants: Scientific and regulatory issues and an environmental perspective. Environment International, 120, 489–495. https://doi.org/10.1016/j.envint.2018.08.035.
Aldini, G., Gamberoni, L., Orioli, M., Beretta, G., Regazzoni, L., Facino, R. M., & Carini, M. (2006). Mass spectrometric characterization of covalent modification of human serum albumin by 4-hydroxy-trans-2-nonenal. Journal of Mass Spectrometry, 41(9), 1149–1161. https://doi.org/10.1002/jms.1067.
Aldini, G., Vistoli, G., Regazzoni, L., Gamberoni, L., Facino, R. M., Yamaguchi, S., Uchida, K., & Carini, M. (2008). Albumin is the main nucleophilic target of human plasma: A protective role against pro-atherogenic electrophilic reactive carbonyl species? Chemical Research in Toxicology, 21(4), 824–835. https://doi.org/10.1021/tx700349r.
Andreozzi, R., Caprio, V., Ciniglia, C., de Champdoré, M., Lo Giudice, R., Marotta, R., & Zuccato, E. (2004). Antibiotics in the environment: Occurrence in Italian STPs, fate, and preliminary assessment on algal toxicity of amoxicillin. Environmental Science & Technology, 38(24), 6832–6838. https://doi.org/10.1021/es049509a.
Anlas, C., & Ustuner, O. (2016). Genotoxic assessment of amoxicillin in rainbow trout (Oncorhynchus mykiss) by comet assay and micronucleus test. Fresenius Environmental Bulletin, 25(12), 5358–5364.
Azanu, D., Styrishave, B., Darko, G., Weisser, J. J., & Abaidoo, R. C. (2018). Occurrence and risk assessment of antibiotics in water and lettuce in Ghana. Science of the Total Environment, 622-623, 293–305. https://doi.org/10.1016/j.scitotenv.2017.11.287.
Beekhuijzen, M., de Koning, C., Flores-Guillén, M. E., de Vries-Buitenweg, S., Tobor-Kaplon, M., van de Waart, B., & Emmen, H. (2015). From cutting edge to guideline: A first step in harmonization of the zebrafish embryotoxicity test (ZET) by describing the most optimal test conditions and morphology scoring system. Reproductive Toxicology, 56, 64–76.
Blahová, J., Plhalová, L., Hostovský, M., Divišová, L., Dobšíková, R., Mikulíková, I., Štěpánová, S., & Svobodováa, Z. (2013). Oxidative stress responses in zebrafish (Danio rerio) after subchronic exposure to atrazine. Food and Chemical Toxicology, 61, 82–85. https://doi.org/10.1016/j.fct.2013.02.041.
Buege, J. A., & Aust, S. D. (1978). Microsomal lipid peroxidation. Methods in Enzymology, 52, 302–310. https://doi.org/10.1016/S0076-6879(78)52032-6.
Bush, K. (2003). β-lactam antibiotics: Penicillin, and other β-lactam antibiotics. In: Finch RC, Greenwood D, Norrby SR, Whitley RJ. Antibiotic and chemotherapy: Anti-infective agents and their use in therapy (8th ed.). Churchill Livingstone 1000 pp.
Busto, R. V., Roberts, J., Hunter, C., Escudero, A., Helwig, K., & Coelho, L. H. G. (2020). Mechanistic and ecotoxicological studies of amoxicillin removal through anaerobic degradation systems. Ecotoxicology and Environmental Safety, 192, 110207. https://doi.org/10.1016/j.ecoenv.2020.110207.
Castiglioni, S., Bagnati, R., Calamari, D., Fanelli, R., & Zuccato, E. (2005). A multiresidue analytical method using solid-phase extraction and high-pressure liquid chromatography tandem mass spectrometry to measure pharmaceuticals of different therapeutic classes in urban wastewaters. Journal of Chromatography. A, 1092(2), 206–215. https://doi.org/10.1016/j.chroma.2005.07.012.
Çelik, E. S., Kaya, H., & Yılmaz, S. (2012). Effects of phosalone on mineral contents and spinal deformities in common carp (Cyprinus carpio, L. 1758). Turkish Journal of Fisheries and Aquatic Sciences, 12, 259–264. https://doi.org/10.4194/1303-2712-v12_2_10.
Chowdhury, J., Mandal, T. K., & Mondal, S. (2020). Genotoxic impact of emerging contaminant amoxicillin residue on zebra fish (Danio rerio) embryos. Heliyon, 6, e05379. https://doi.org/10.1016/j.heliyon.2020.e05379.
Danner, M.-C., Robertson, A., Behrends, V., & Reiss, J. (2019). Antibiotic pollution in surface fresh waters: Occurrence and effects. Science of the Total Environment, 664(10), 793–804. https://doi.org/10.1016/j.scitotenv.2019.01.406.
Dave, G., & Xiu, R. Q. (1991). Toxicity of mercury, copper, nickel, lead, and cobalt to embryos and larvae of zebrafish Brachydanio rerio. Archives of Environmental Contamination and Toxicology, 21(1), 126–134. https://doi.org/10.1007/BF01055567.
de Oliveira, R. C. S., Oliveira, R., Rodrigues, M. A. C., de Farias, N. O., Sousa-Moura, D., Nunes, N. A., Andrade, T. S., & Grisolia, C. K. (2020). Lethal and sub-lethal effects of nitrofurantoin on zebrafish early-life stages. Water, Air, and Soil Pollution, 231(2), 54. https://doi.org/10.1007/s11270-020-4414-4.
Dennery, P. A. (2007). Effects of oxidative stress on embryonic development. Birth Defects Research. Part C, Embryo Today, 81, 155–162. https://doi.org/10.1002/bdrc.20098.
Denucé, J. M. (1985). How embryos escape from their envelopes: a new look at the (phylogenetically) old problem of hatching. Meded Kon Acad Wet Belgie, 46, 1–30.
Dinh, Q. T., Alliot, F., Moreau-Guigon, E., Eurin, J., Chevreuil, M., & Labadie, P. (2011). Measurement of trace levels of antibiotics in river water using on-line enrichment and triple-quadrupole LC-MS/MS. Talanta., 85(3), 1238–1245. https://doi.org/10.1016/j.talanta.2011.05.013.
Dwyer, D. J., Kohanski, M. A., Hayete, B., & Collins, J. J. (2007). Gyrase inhibitors induce an oxidative damage cellular death pathway in Escherichia coli. Molecular Systems Biology, 3(1), 3–91. https://doi.org/10.1038/msb4100135.
Dwyer, D. J., Belenky, P. A., Yang, J. H., MacDonald, I. C., Martell, J. D., Takahashi, N., Chan, C. T. Y., Lobritz, M. A., Braff, D., Schwarz, E. G., Ye, J. D., Pati, M., Vercruysse, M., Ralifo, P. S., Allison, K. R., Khalil, A. S., Ting, A. Y., Walker, G. C., & Collins, J. J. (2014). Antibiotics induce redox-related physiological alterations as part of their lethality. Proceedings of the National Academy of Sciences of the United States of America, 111(20), E100–E109. https://doi.org/10.1073/pnas.1401876111.
Ebele, A. J., Abdallah, M. A.-E., & Harrad, S. (2017). Pharmaceuticals and personal care products (PPCPs) in the freshwater aquatic environment. Emerging Contaminants, 3(1), 1–16. https://doi.org/10.1016/j.emcon.2016.12.004.
Elizalde-Velázquez, A., Martínez-Rodríguez, H., Galar-Martínez, M., Dublán-García, O., Islas-Flores, H., Rodríguez-Flores, J., Castañeda-Peñalvo, G., Lizcano-Sanz, I., & Gómez-Oliván, L. M. (2017). Effect of amoxicillin exposure on brain, gill, liver, and kidney of common carp (Cyprinus carpio): The role of amoxicilloic acid. Environmental Toxicology, 32(4), 1102–1120. https://doi.org/10.1002/tox.22307.
Faghih, O., Zhang, Z., Ranade, R. M., Gillespie, J. R., Creason, S. A., Huang, W., Shibata, S., Barros-Álvarez, X., Verlinde, C. L. M. J., Hol, W. G. J., Fan, E., & Buckner, F. S. (2017). Development of methionyl-tRNA synthetase inhibitors as antibiotics for Gram-positive bacterial infections. Antimicrobial Agents and Chemotherapy, 61(11). https://doi.org/10.1128/AAC.00999-17.
Farré, M., Pérez, S., Kantiani, L., & Barceló, D. (2008). Fate and toxicity of emerging pollutants, their metabolites and transformation products in the aquatic environment. TrAC, 27(11), 991–1007. https://doi.org/10.1016/j.trac.2008.09.010.
Fatta-Kassinos, D., Meric, S., & Nikolaou, A. (2011). Pharmaceutical residues in environmental waters and wastewater: current state of knowledge and future research. Analytical and Bioanalytical Chemistry, 399(1), 251–275. https://doi.org/10.1007/s00216-010-4300-9.
Fent, K., Weston, A. A., & Caminada, D. (2006). Ecotoxicology of human pharmaceuticals. Aquatic Toxicology, 76(2), 122–159. https://doi.org/10.1016/j.aquatox.2005.09.009.
Fulda, S., Gorman, A. M., Hori, O., & Samali, A. (2010). Cellular stress responses: cell survival and cell death. International Journal of Cell Biology, 2010, 214074. https://doi.org/10.1155/2010/214074.
Gomes, M. P., Richardi, V. S., Bicalho, E. M., da Rocha, D. C., Navarro-Silva, M. A., Soffiatti, P., Garcia, Q. S., & Sant'Anna-Santos, B. F. (2019). Effects of ciprofloxacin and roundup on seed germination and root development of maize. Science of the Total Environment, 651(Pt 2), 2671–2678. https://doi.org/10.1016/j.scitotenv.2018.09.365.
Guérin, P., El Mouatassim, S., & Ménézo, Y. (2001). Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Human Reproduction Update, 7(2), 175–189. https://doi.org/10.1093/humupd/7.2.175.
Haendel, M. A., Tilton, F., Bailey, G. S., & Tanguay, R. L. (2004). Developmental toxicity of the dithiocarbamate pesticide sodium metam in zebrafish. Toxicological Sciences, 81(2), 390–400. https://doi.org/10.1093/toxsci/kfh202.
Hallare, A. V., Schirling, M., Luckenbach, T., Köhler, H.-R., & Triebskorn, R. (2005). Combined effects of temperature and cadmium on developmental parameters and biomarker responses in zebrafish (Danio rerio) embryos. Journal of Thermal Biology, 30(1), 7–17. https://doi.org/10.1016/j.jtherbio.2004.06.002.
Hermsen, S. A. B., van den Brandhof, E.-J., van der Ven, L. T. M., & Piersma, A. H. (2011). Relative embryotoxicity of two classes of chemicals in a modified zebrafish embryotoxicity test and comparison with their in vivo potencies. Toxicology In Vitro, 25(3), 745–753. https://doi.org/10.1016/j.tiv.2011.01.005.
Hill, A. J., Bello, S. M., Prasch, A. L., Peterson, R. E., & Heideman, W. (2004). Water permeability and TCDD-induced edema in zebrafish early-life stages. Toxicological Sciences, 78(1), 78–87. https://doi.org/10.1093/toxsci/kfh056.
Hill, A. J., Teraoka, H., Heideman, W., & Peterson, R. E. (2005). Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicological Sciences, 86(1), 6–19. https://doi.org/10.1093/toxsci/kfi110.
Hirsch, R., Ternes, T. A., Haberer, K., Mehlich, A., Ballwanz, F., & Kratz, K.-L. (1998). Determination of antibiotics in different water compartments via liquid chromatography-electrospray tandem mass spectrometry. Journal of Chromatography. A, 815(2), 213–223. https://doi.org/10.1016/S0021-9673(98)00335-5.
Jiang, Z. Y., Hunt, J. V., & Wolff, S. P. (1992). Ferrous ion oxidation in the presence of xylenol orange for detection of lipid hydroperoxide in low density lipoprotein. Analytical Biochemistry, 202(2), 384–389. https://doi.org/10.1016/0003-2697(92)90122-N.
Kaseva, M. E., Mwegoha, W. J. S., Kihampa, C., & Matiko, S. (2008). Performance of a waste stabilization pond system treating domestic and hospital wastewater and its implications to the aquatic environment-a case study in Dar es Salaam, Tanzania. Journal of Building and Land Development, 15(1-2), 76–88.
Kasprzyk-Hordern, B., Dinsdale, R. M., & Guwy, A. J. (2008). Multi-residue method for the determination of basic/neutral pharmaceuticals and illicit drugs in surface water by solid-phase extraction and ultra-performance liquid chromatography-positive electrospray ionisation tandem mass spectrometry. Journal of Chromatography. A, 1161(1-2), 132–145. https://doi.org/10.1016/j.chroma.2007.05.074.
Khazaei, M., & Aghaz, F. (2017). Reactive oxygen species generation and use of antioxidants during in vitro maturation of oocytes. International Journal of Fertility and Sterility, 11(2), 63–70. https://doi.org/10.22074/ijfs.2017.4995.
Kim, Y., Choi, K., Jung, J., Park, S., & Kim, P. -., & Park, J. (2007). Aquatic toxicity of acetaminophen, carbamazepine, cimetidine, diltiazem and six major sulfonamides, and their potential ecological risks in Korea. Environment International, 33(3), 370–375. https://doi.org/10.1016/j.envint.2006.11.017.
Kim, C., Ryu, H.-D., Chung, E. G., & Kim, Y. (2018). Determination of 18 veterinary antibiotics in environmental water using high-performance liquid chromatography-Q-Orbitrap combined with on-line solid-phase extraction. Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences, 1084, 158–165. https://doi.org/10.1016/j.jchromb.2018.03.038.
Kimmel, C.B., Ballard, W.W., Kimmel, S.R., Ullmann, B., Schilling, T.F., (1995). Stages of embryonic development of the zebrafish. Developmental Dynamics 203, 253–310. https://doi.org/10.1002/aja.1002030302.
Kohanski, M. A., Dwyer, D. J., Hayete, B., Lawrence, C. A., & Collins, J. J. (2007). A common mechanism of cellular death induced by bactericidal antibiotics. Cell, 130(5), 797–810. https://doi.org/10.1016/j.cell.2007.06.049.
Kohanski, M. A., DePristo, M. A., & Collins, J. J. (2010). Sub-lethal antibiotic treatment leads to multidrug resistance via radical-induced mutagenesis. Molecular Cell, 37(3), 311–320. https://doi.org/10.1016/j.molcel.2010.01.003.
Kormoczi, G. F., Wölfel, U. M., Rosenkranz, A. R., Hörl, W. H., Oberbauer, R., & Zlabinger, G. J. (2001). Serum proteins modified by neutrophil-derived oxidants as mediators of neutrophil stimulation. Journal of Immunology, 167(1), 451–460. https://doi.org/10.4049/jimmunol.167.1.451.
Lammer, E., Carr, G. J., Wendler, K., Rawlings, J. M., Belanger, S. E., & Braunbeck, T. (2009). Is the fish embryo toxicity test (FET) with the zebrafish (Danio rerio) a potential alternative for the fish acute toxicity test? Comparative Biochemistry and Physiology, Part C: Toxicology & Pharmacology, 149(2), 196–209. https://doi.org/10.1016/j.cbpc.2008.11.006.
Lawrence, J. W., Claire, D. C., Weissig, V., & Rowe, T. C. (1996). Delayed cytotoxicity and cleavage of mitochondrial DNA in ciprofloxacin-treated mammalian cells. Molecular Pharmacology, 50(5), 1178–1188.
Locatelli, M. A. F., Sodré, F. F., & Jardim, W. F. (2011). Determination of antibiotics in Brazilian surface waters using liquid chromatography-electrospray tandem mass spectrometry. Archives of Environmental Contamination and Toxicology, 60, 385–393. https://doi.org/10.1007/s00244-010-9550-1.
Loos, R., Marinov, D., Sanseverino, I., Napierska, D., & Lettieri, T. (2018). Review of the 1st Watch List under the Water Framework Directive and recommendations for the 2nd Watch List. Publications Office of the European Union 268 pp.
Lowes, D. A., Wallace, C., Murphy, M. P., Webster, N. R., & Galley, H. F. (2009). The mitochondria targeted antioxidant MitoQ protects against fluoroquinolone-induced oxidative stress and mitochondrial membrane damage in human Achilles tendon cells. Free Radical Research, 43(3), 323–328. https://doi.org/10.1080/10715760902736275.
Ma, C., Yang, X., & Lewis, P. J. (2016). Bacterial transcription as a target for antibacterial drug development. Microbiology and Molecular Biology Reviews, 80(1), 139–160. https://doi.org/10.1128/MMBR.00055-15.
Manaia, C. M. (2017). Assessing the risk of antibiotic resistance transmission from the environment to humans: Non-direct proportionality between abundance and risk. Trends in Microbiology, 25(3), 173–181. https://doi.org/10.1016/j.tim.2016.11.014.
Misra, H. P., & Fridovich, I. (1972). The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. The Journal of Biological Chemistry, 247(10), 3170–3175. https://doi.org/10.1016/S0021-9258(19)45228-9.
Mu, X., Pang, S., Sun, X., Gao, J., Chen, J., Chen, X., Li, X., & Wang, C. (2013). Evaluation of acute and developmental effects of difenoconazole via multiple stage zebrafish assays. Environmental Pollution, 175, 147–157. https://doi.org/10.1016/j.envpol.2012.12.029.
Muramoto, S. (1981). Vertebral column damage and decrease of calcium concentration in fish exposed experimentally to cadmium. Environmental Pollution Series A, Ecological and Biological, 24(2), 125–133. https://doi.org/10.1016/0143-1471(81)90074-X.
Mutiyar, P. K., & Mittal, A. K. (2013). Occurrences and fate of an antibiotic amoxicillin in extended aeration-based sewage treatment plant in Delhi, India: a case study of emerging pollutant. Desalination and Water Treatment, 51(31-33), 6158–6164. https://doi.org/10.1080/19443994.2013.770199.
Naidu, R., Arias, E. V. A., Liu, Y., & Jit, J. (2016). Emerging contaminants in the environment: Risk-based analysis for better management. Chemosphere, 154, 350–357. https://doi.org/10.1016/j.chemosphere.2016.03.068.
OECD (2013). Test No. 236: Fish Embryo Acute Toxicity (FET) Test. Guidelines for the testing of chemicals, section 2. https://doi.org/10.1787/9789264203709-en.
Oettl, K., & Stauber, R. E. (2007). Physiological and pathological changes in the redox state of human serum albumin critically influence its binding properties. British Journal of Pharmacology, 151(5), 580–590. https://doi.org/10.1038/sj.bjp.0707251.
Oliveira, R., McDonough, S., Ladewig, J. C. L., Soares, A. M. V. M., Nogueira, A. J. A., & Domingues, I. (2013). Effects of oxytetracycline and amoxicillin on development and biomarkers activities of zebrafish (Danio rerio). Environmental Toxicology and Pharmacology, 36(3), 903–912. https://doi.org/10.1016/j.etap.2013.07.019.
Orozco-Hernández, J. M., Gómez-Oliván, L. M., Heredia-García, G., Luja-Mondragón, M., Islas-Flores, H., SanJuan-Reyes, N., Galar-Martínez, M., García-Medina, S., & Dublán-García, O. (2019). Genotoxic and cytotoxic alterations induced by environmentally-relevant concentrations of amoxicillin in blood cells of Cyprinus carpio. Chemosphere, 236, 124323. https://doi.org/10.1016/j.chemosphere.2019.07.054.
Ozolins, T. R., & Hales, B. F. (1997). Oxidative stress regulates the expression and activity of transcription factor activator protein-1 in rat conceptus. The Journal of Pharmacology and Experimental Therapeutics, 280(2), 1085–1093.
Park, S., & Choi, K. (2008). Hazard assessment of commonly used agricultural antibiotics on aquatic ecosystems. Ecotoxicology, 17(6), 526–538. https://doi.org/10.1007/s10646-008-0209-x.
Pašková, V., Hilscherová, K., & Bláha, L. (2011). Teratogenicity and embryotoxicity in aquatic organisms after pesticide exposure and the role of oxidative stress. Reviews of Environmental Contamination and Toxicology, 211, 25–61. https://doi.org/10.1007/978-1-4419-8011-3_2.
Praveena, S. M., Mohd Rashid, M. Z., Mohd Nasir, F. A., Sze Yee, W., & Aris, A. Z. (2019). Occurrence and potential human health risk of pharmaceutical residues in drinking water from Putrajaya (Malaysia). Ecotoxicology and Environmental Safety, 180, 549–556. https://doi.org/10.1016/j.ecoenv.2019.05.051.
Radi, R., Turrens, J. F., Chang, L. Y., Bush, K. M., Crapo, J. D., & Freeman, B. A. (1991). Detection of catalase in rat heart mitochondria. The Journal of Biological Chemistry, 266(32), 22028–22034. https://doi.org/10.1016/S0021-9258(18)54740-2.
Rao, R., Kaur, S. P., & Nanda, S. (2011). Amoxicillin: A broad-spectrum antibiotic. International Journal of Pharmacy and Pharmaceutical Sciences, 3(3), 30–37.
Reimers, M. J., Flockton, A. R., & Tanguay, R. L. (2004). Ethanol- and acetaldehyde-mediated developmental toxicity in zebrafish. Neurotoxicology and Teratology, 26(6), 769–781. https://doi.org/10.1016/j.ntt.2004.06.012.
Reyns, T., Cherlet, M., De Baere, S., De Backer, P., & Croubels, S. (2008). Rapid method for the quantification of amoxicillin and its major metabolites in pig tissues by liquid chromatography-tandem mass spectrometry with emphasis on stability issues. Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences, 861(1), 108–116. https://doi.org/10.1016/j.jchromb.2007.11.007.
Santos, L. H. M. L. M., Araujo, A. N., Fachini, A., Pena, A., Delerue-Matos, C., & Montenegro, M. C. B. S. M. (2010). Ecotoxicological aspects related to the presence of pharmaceuticals in the aquatic environment. Journal of Hazardous Materials, 175(1-3), 45–95. https://doi.org/10.1016/j.jhazmat.2009.10.100.
Sass, P., & Brotz-Oesterhelt, H. (2013). Bacterial cell division as a target for new antibiotics. Current Opinion in Microbiology, 16(5), 522–530. https://doi.org/10.1016/j.mib.2013.07.006.
Schlisser, A. E., Yan, J., & Hales, B. F. (2010). Teratogen-induced oxidative stress targets glyceraldehyde-3-phosphate dehydrogenase in the organogenesis stage mouse embryo. Toxicological Sciences, 118(2), 686–695. https://doi.org/10.1093/toxsci/kfq287.
Shi, X. J., & Zhou, B. (2010). The role of Nrf2 and MAPK pathways in PFOS-induced oxidative stress in zebrafish embryos. Toxicological Sciences, 115(2), 391–400. https://doi.org/10.1093/toxsci/kfq066.
Singh, V., Pandey, B., & Suthar, S. (2018). Phytotoxicity of amoxicillin to the duckweed Spirodela polyrhiza: Growth, oxidative stress, biochemical traits and antibiotic degradation. Chemosphere, 201, 492–502. https://doi.org/10.1016/j.chemosphere.2018.03.010.
Sinthuchai, D., Boontanon, S. K., Boontanon, N., & Polprasert, C. (2016). Evaluation of removal efficiency of human antibiotics in wastewater treatment plants in Bangkok, Thailand. Water Science and Technology, 73(1), 182–191. https://doi.org/10.2166/wst.2015.484.
Song, C., Gao, N.-Y., & Gao, H.-W. (2010). Transmembrane distribution of kanamycin and chloramphenicol: Insights into the cytotoxicity of antibacterial drugs. Molecular BioSystems, 6(10), 1901–1910. https://doi.org/10.1039/B921810F.
Straub, J. O., Gysel, D., Kastl, U., Klemmer, J., Sonderegger, M., & Studer, M. (2012). Environmental risk assessment for ancillary substances in biotechnological production of pharmaceuticals. Environmental Toxicology and Chemistry, 31, 681–687. https://doi.org/10.1002/etc.1733.
Test No. 236: Fish Embryo Acute Toxicity (FET) Test. (2013). OECD Guidelines for the Testing of Chemicals, Section 2. OECD.
Theodorakis, C. W., Swartz, C. D., Rogers, W. J., Bickham, J. W., Donnelly, K. C., & Adams, S. M. (2000). Relationship between genotoxicity, mutagenicity, and fish community structure in a contaminated stream. Journal of Aquatic Ecosystem Stress and Recovery, 7, 131–143. https://doi.org/10.1023/A:1009971330138.
van Eijk, E., Wittekoek, B., Kuijper, E. J., & Smits, W. K. (2017). DNA replication proteins as potential targets for antimicrobials in drug-resistant bacterial pathogens. The Journal of Antimicrobial Chemotherapy, 72(5), 1275–1284. https://doi.org/10.1093/jac/dkw548.
Wang, H., Che, B., Duan, A., Mao, J., Dahlgren, R. A., Zhang, M., Zhang, H., Zeng, A., & Wang, X. (2013). Toxicity evaluation of β-diketone antibiotics on the development of embryo-larval zebrafish (Danio rerio). Environmental Toxicology, 29(10), 1134–1146. https://doi.org/10.1002/tox.21843.
Wang, G., Zhou, S., Han, X., Zhang, L., Ding, S., Li, Y., Zhang, D., & Zarin, K. (2020). Occurrence, distribution, and source track of antibiotics and antibiotic resistance genes in the main rivers of Chongqing City, southwest China. Journal of Hazardous Materials, 389, 122110. https://doi.org/10.1016/j.jhazmat.2020.122110.
Watkinson, A. J., Murby, E. J., & Costanzo, S. D. (2007). Removal of antibiotics in conventional and advanced wastewater treatment: Implications for environmental discharge and wastewater recycling. Water Research, 41(18), 4164–4176. https://doi.org/10.1016/j.watres.2007.04.005.
Watkinson, A. J., Murby, E. J., Kolpin, D. W., & Costanzo, S. D. (2009). The occurrence of antibiotics in an urban watershed: From wastewater to drinking water. Science of the Total Environment, 407(8), 2711–2723. https://doi.org/10.1016/j.scitotenv.2008.11.059.
Weigt, S., Huebler, N., Strecker, R., Braunbeck, T., & Broschard, T. H. (2011). Zebrafish (Danio rerio) embryos as a model for testing proteratogens. Toxicology, 281(1-3), 25–36. https://doi.org/10.1016/j.tox.2011.01.004.
Weinberg, J. M., Bienholz, A., & Venkatachalam, M. A. (2016). The role of glycine in regulated cell death. Cellular and Molecular Life Sciences, 73(11-12), 2285–2308. https://doi.org/10.1007/s00018-016-2201-6.
World Health Organization (WHO). (2018). WHO report on surveillance of antibiotic consumption 2016-2018 early implementation. World Health Organization 127 pp.
Yamagami, K. (1981). Mechanisms of hatching in fish: Secretion of hatching enzyme and enzymatic choriolysis. American Zoologist, 21(2), 459–471. https://doi.org/10.1093/icb/21.2.459.
Yamashita, N., Yasojima, M., Nakada, N., Miyajima, K., Komori, K., Suzuki, Y., & Tanaka, H. (2006). Effects of antibacterial agents, levofloxacin and clarithromycin, on aquatic organisms. Water Science and Technology, 53(11), 65–72. https://doi.org/10.2166/wst.2006.338.
Yasser, E.-N., & Nabila, E. D. (2015). Toxicity of amoxicillin and erythromycin to fish and mosquitoes. Ecotoxicology and Environmental Contamination, 10(1), 13–21. https://doi.org/10.5132/eec.2015.01.03.
Yumnamcha, T., Roy, D., Devi, M. D., & Nongthomba, U. (2015). Evaluation of developmental toxicity and apoptotic induction of the aqueous extract of Millettia pachycarpa using zebrafish as model organism. Toxicological and Environmental Chemistry, 97(10), 1363–1381. https://doi.org/10.1080/02772248.2015.1093750.
Zhou, L., Limbu, S. M., Shen, M., Zhai, W., Qiao, F., He, A., Du, Z.-Y., & Zhang, M. (2018). Environmental concentrations of antibiotics impair zebrafish gut health. Environmental Pollution, 235, 245–254. https://doi.org/10.1016/j.envpol.2017.12.073.
Zuccato, E., Castiglioni, S., Bagnati, R., Melis, M., & Fanelli, R. (2010). Source, occurrence and fate of antibiotics in the Italian aquatic environment. Journal of Hazardous Materials, 179(1-3), 1042–1048. https://doi.org/10.1016/j.jhazmat.2010.03.110.
Funding
This study was made possible by financial support from the Consejo Nacional de Ciencia y Tecnología (CONACyT, Project 300727).
Author information
Authors and Affiliations
Contributions
Edgar David González-González and Leobardo Manuel Gómez-Oliván performed all the exposure experiments. Leobardo Manuel Gómez-Oliván and Edgar David González-González were involved in the conception. Leobardo Manuel Gómez-Oliván, Edgar David González-González, and Hariz Islas-Flores were involved in the design and interpretation of the data, and the writing of the manuscript was with input from Marcela Galar-Martínez.
Corresponding author
Ethics declarations
Ethics Approval
The study was approved by the Ethics and Research Committee of the Universidad Autónoma del Estado de México, Toluca, Mexico (Approval ID: CEI.UAEMCQ.REC.132.2020).
Consent to Participate
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Plant Reproducibility
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
González-González, E.D., Gómez-Oliván, L.M., Islas-Flores, H. et al. Developmental Effects of Amoxicillin at Environmentally Relevant Concentration Using Zebrafish Embryotoxicity Test (ZET). Water Air Soil Pollut 232, 196 (2021). https://doi.org/10.1007/s11270-021-05148-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05148-6