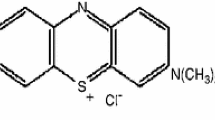
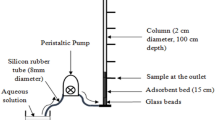
Abstract
The fixed-bed column biosorption process is the most widely used system, which aims to expand to industrial scale in wastewater treatment, and requires for its design thermodynamic equilibrium data. In this study, fixed-bed column experiments using malt bagasse, an industrial byproduct, were performed to quantitatively evaluate the dye biosorption capacity, as well as perform mathematical modeling of the breakthrough curves and obtain mass transfer parameters. The FTIR spectrum of malt bagasse was performed before and after the dye biosorption process. The best operating condition of the column occurred at the feed flow of 4mLmin− 1. The Langmuir isotherm (qmax = 38.44mgg− 1; R2 = 0.98) was used to represent the equilibrium data between the phases (fluid/solid) in column. The phenomenological mathematical model, which considered the resistance to internal diffusion as a limiting step of mass transfer, was able to adequately predict the dynamic behavior of the biosorption column operation, constituting a useful tool for analysis, description, and design of dye biosorption in fixed-bed column.





Similar content being viewed by others
References
Alardhi, S. M., Albayati, T. M., & Alrubaye, J. M. (2020). Adsorption of the methyl green dye pollutant from aqueous solution using mesoporous materials MCM-41 in a fixed-bed column. Heliyon, 6(1), e03253. https://doi.org/10.1016/j.heliyon.2020.e03253, https://linkinghub.elsevier.com/retrieve/pii/S2405844020300980https://linkinghub.elsevier.com/retrieve/pii/S2405844020300980.
Apiratikul, R. (2020). Application of analytical solution of advection-dispersion-reaction model to predict the breakthrough curve and mass transfer zone for the biosorption of heavy metal ion in a fixed bed column. Process Safety and Environmental Protection, 137, 58–65. https://doi.org/10.1016/j.psep.2020.02.018, https://linkinghub.elsevier.com/retrieve/pii/S0957582019323596https://linkinghub.elsevier.com/retrieve/pii/S0957582019323596.
Borba, C. E., Guirardello, R., Silva, E. A., Veit, M. T., & Tavares, C. R. G. (2006). Removal of nickel(II) ions from aqueous solution by biosorption in a fixed bed column: Experimental and theoretical breakthrough curves. Biochemical Engineering Journal, 30(2), 184–191. https://doi.org/10.1016/j.bej.2006.04.001, https://linkinghub.elsevier.com/retrieve/pii/S1369703X0600091Xhttps://linkinghub.elsevier.com/retrieve/pii/S1369703X0600091X.
Chen, S., Yue, Q., Gao, B., Li, Q., Xu, X., & Fu, K. (2012). Adsorption of hexavalent chromium from aqueous solution by modified corn stalk: A fixed-bed column study. Bioresource Technology, 113, 114–120. https://doi.org/10.1016/j.biortech.2011.11.110, https://linkinghub.elsevier.com/retrieve/pii/S0960852411017214https://linkinghub.elsevier.com/retrieve/pii/S0960852411017214.
Cordeiro, L. G., El-Aouar, A. A., & de Araújo, C V B. (2013). Energetic characterization of malt bagasse by calorimetry and thermal analysis. Journal of Thermal Analysis and Calori- metry, 112(2), 713–717. https://doi.org/10.1007/s10973-012-2630-x.
Crini, G. (2006). Non-conventional low-co st adsorbents for dye removal: A review. Bioresource Technology, 97 (9), 1061– 1085. https://doi.org/10.1016/j.biortech.2005.05.001, https://linkinghub.elsevier.com/retrieve/pii/S0960852405002452https://linkinghub.elsevier.com/retrieve/pii/S0960852405002452.
Cussler, E. L. (1997). Diffusion - Mass Transfer In Fluid Systems. Cambridge University Press.
Da Silva, E. A., Cossich, E. S., Tavares, C. R. G., Filho, L. C., & Guirardello, R. (2002). Modeling of copper(II) biosorption by marine alga Sargassum sp. in fixed-bed column. Process Biochemistry, 38(5), 791–799. https://doi.org/10.1016/S0032-9592(02)00231-5.
de Franco, M. A. E., de Carvalho, C. B., Bonetto, M. M., de Pelegrini Soares, R., & Féris, L. A. (2018). Diclofenac removal from water by adsorption using activated carbon in batch mode and fixed-bed column: Isotherms, thermodynamic study and breakthrough curves modeling. Journal of Cleaner Production, 181, 145–154. https://doi.org/10.1016/j.jclepro.2018.01.138, https://linkinghub.elsevier.com/retrieve/pii/S0959652618301604https://linkinghub.elsevier.com/retrieve/pii/S0959652618301604.
da Silva, B. C., Zanutto, A., & Pietrobelli, J. M. T. A. (2019). Biosorption of reactive yellow dye by malt bagasse. Adsorption Science & Technology, 37(3-4), 236–259. https://doi.org/10.1177/0263617418823995.
dos Santos, D. M., Bukzem, A. L., Ascheri, D. P. R., Signini, R., & de Aquino, G. L. B. (2015). Microwave-assisted carboxymethylation of cellulose extracted from brewer’s spent grain. Carbohydrate Polymers, 131, 125–133. https://doi.org/10.1016/j.carbpol.2015.05.051, https://linkinghub.elsevier.com/retrieve/pii/S0144861715004579https://linkinghub.elsevier.com/retrieve/pii/S0144861715004579.
Evy Alice Abigail, M., & Chidambaram, R. (2016). Rice husk as a low cost nanosorbent for 2,4-dichlorophenoxyacetic acid removal from aqueous solutions. Ecological Engineering, 92, 97–105. https://doi.org/10.1016/j.ecoleng.2016.03.020, https://www.sciencedirect.com/science/article/abs/pii/S0925857416301690.
Ferandin Honorio, J., Veit, M. T., Suzaki, P. Y. R., Coldebella, P. F., Sloboda Rigobello, E., & Tavares, C. R. G. (2020). Adsorption of naturals hormones estrone, 17 β -estradiol, and estriol by rice husk: monocomponent and multicomponent kinetics and equilibrium. Environmental Technology, 41(9), 1075–1092. https://doi.org/10.1080/09593330.2018.1521472.
Ferraz, A. I., Amorim, C., Tavares, T., & Teixeira, J. A. (2015). Chromium(III) biosorption onto spent grains residual from brewing industry: equilibrium, kinetics and column studies. International Journal of Environmental Science and Technology, 12(5), 1591–1602. https://doi.org/10.1007/s13762-014-0539-6.
Fontana, I. B., Peterson, M., & Cechinel, M. A. P. (2018). Application of brewing waste as biosorbent for the removal of metallic ions present in groundwater and surface waters from coal regions. Journal of Environmental Chemical Engineering, 6(1), 660–670. https://doi.org/10.1016/j.jece.2018.01.005, https://linkinghub.elsevier.com/retrieve/pii/S2213343718300058.
Fontana, K. B., Chaves, E. S., Sanchez, J. D. S., Watanabe, E. R. L. R., Pietrobelli, J. M. T. A., & Lenzi, G. G. (2016). Textile dye removal from aqueous solutions by malt bagasse: Isotherm, kinetic and thermodynamic studies. Ecotoxicology and Environmental Safety, 124, 329–336. https://doi.org/10.1016/j.ecoenv.2015.11.012, https://linkinghub.elsevier.com/retrieve/pii/S0147651315301561https://linkinghub.elsevier.com/retrieve/pii/S0147651315301561.
Geankoplis, C. J. (1993). Transport processes and unit operations, 4th edn. New York: PTR Prentice Hall.
Glueckauf, E., & Coates, J. I. (1947). Theory of chromatography. Part IV. The influence of incomplete equilibrium on the front boundary of chromatograms and on the effectiveness of separation. Journal of the Chemical Society (Resumed), 1315. https://doi.org/10.1039/jr9470001315.
Gokulan, R., Ganesh Prabhu, G., & Jegan, J. (2019). A novel sorbent Ulva lactuca-derived biochar for remediation of Remazol Brilliant Orange 3R in packed column. Water Environment Research, 91(7), 642–649. https://doi.org/10.1002/wer.1092.
Guaratini, C. C. I., & Zanoni, V. B. (1999). Revis ao: Corantes Têxteis. Química Nova, 23(1), 71–78.
Jain, S. N., & Gogate, P. R. (2018). Efficient removal of Acid Green 25 dye from wastewater using activated Prunus Dulcis as biosorbent: Batch and column studies. Journal of Environmental Management, 210, 226–238. https://doi.org/10.1016/j.jenvman.2018.01.008, https://linkinghub.elsevier.com/retrieve/pii/S0301479718300082https://linkinghub.elsevier.com/retrieve/pii/S0301479718300082.
Juchen, P. T., Piffer, H. H., Veit, M. T., da Cunha Gonçalves, G., Palácio, S. M., & Zanette, J. C. (2018). Biosorption of reactive blue BF-5G dye by malt bagasse: kinetic and equilibrium studies. Journal of Environmental Chemical Engineering, 6(6), 7111–7118. https://doi.org/10.1016/j.jece.2018.11.009, https://linkinghub.elsevier.com/retrieve/pii/S2213343718306882https://linkinghub.elsevier.com/retrieve/pii/S2213343718306882.
Kezerle, A., Velić, N., Hasenay, D., & Kovačević, D. (2018). Lignocellulosic Materials as Dye Adsorbents: Adsorption of Methylene Blue and Congo Red on Brewers’ Spent Grain. Croatica Chemica Acta, 91(1), 53–64. https://doi.org/10.5562/cca3289, https://hrcak.srce.hr/file/288988.
Khalfa, L., Sdiri, A., Bagane, M., & Cervera, M. L. (2021). A calcined clay fixed bed adsorption studies for the removal of heavy metals from aqueous solutions. Journal of Cleaner Production, 278, 123935. https://doi.org/10.1016/j.jclepro.2020.123935, https://linkinghub.elsevier.com/retrieve/pii/S0959652620339809.
Ko, D. C. K., Porter, J. F., & McKay, G. (2001). Film-pore diffusion model for the fixed-bed sorption of copper and cadmium ions onto bone char. Water Research, 35(16), 3876–3886. https://doi.org/10.1016/S0043-1354(01)00114-2, https://linkinghub.elsevier.com/retrieve/pii/S0043135401001142.
Langmuir, I. (1918). The adsorption of gases on plane surfaces of glass, mica and platinum. Journal of the American Chemical Society, 40(9), 1361–1403.
Li nán-Montes, A., de la Parra-Arciniega, S. M., Garza-González, M. T., García-Reyes, R. B., Soto-Regalado, E., & Cerino-Córdova, F. J. (2014). Characterization and thermal analysis of agave bagasse and malt spent grain. Journal of Thermal Analysis and Calorimetry, 115(1), 751–758. https://doi.org/10.1007/s10973-013-3321-y.
Machado, F. M., Bergmann, C. P., Fernandes, T. H. M., Lima, E. C., Royer, B., Calvete, T., & Fagan, S. B. (2011). Adsorption of Reactive Red M-2BE dye from water solutions by multi-walled carbon nanotubes and activated carbon. Journal of Hazardous Materials, 192(3), 1122–1131. https://doi.org/10.1016/j.jhazmat.2011.06.020, https://linkinghub.elsevier.com/retrieve/pii/S0304389411007825.
Marin, P., Borba, C. E., Módenes, A. N., Espinoza-Qui nones, F. R., de Oliveira, S. P. D., & Kroumov, A. D. (2014). Determination of the mass transfer limiting step of dye adsorption onto commercial adsorbent by using mathematical models. Environmental Technology, 35(18), 2356–2364. https://doi.org/10.1080/09593330.2014.904445.
McCabe, W. L., Smith, J. C., & Harriott, P. (2001). Unit operations of chemical engineering, 6th edn. New York: McGraw-Hill.
Nelder, J. A., & Mead, R. (1965). A Simplex Method for Function Minimization. The Computer Journal, 7(4), 308–313. https://doi.org/10.1093/comjnl/7.4.308.
Nguyen, T. A. H., Ngo, H. H., Guo, W. S., Pham, T. Q., Li, F. M., Nguyen, T. V., & Bui, X. T. (2015). Adsorption of phosphate from aqueous solutions and sewage using zirconium loaded okara (ZLO): Fixed-bed column study. Science of The Total Environment, 523, 40–49. https://doi.org/10.1016/j.scitotenv.2015.03.126, https://linkinghub.elsevier.com/retrieve/pii/S0048969715004106.
O’Mahony, T., Guibal, E., & Tobin, J. M. (2002). Reactive dye biosorption by Rhizopus arrhizus biomass. Enzyme and Microbial Technology, 31(4), 456–463. https://doi.org/10.1016/S0141-0229(02)00110-2, https://linkinghub.elsevier.com/retrieve/pii/S0141022902001102.
Pedro Silva, J., Sousa, S., Rodrigues, J., Antunes, H., Porter, J. J., Gonçalves, I., & Ferreira-Dias, S. (2004). Adsorption of acid orange 7 dye in aqueous solutions by spent brewery grains. Separation and Purification Technology, 40(3), 309–315. https://doi.org/10.1016/j.seppur.2004.03.010, https://linkinghub.elsevier.com/retrieve/pii/S1383586604000991https://linkinghub.elsevier.com/retrieve/pii/S1383586604000991.
Reis, H. C. O., Cossolin, A. S., Santos, B. A. P., Castro, K. C., Pereira, G. M., Silva, V. C., Sousa, Jr. P.T., Dall’Oglio, E. L., Vasconcelos, L. G., & Morais, E. B. (2018). Malt Bagasse Waste as Biosorbent for Malachite Green: An Ecofriendly Approach for Dye Removal from Aqueous Solution. International Journal of Biological, Life and Agricultural Sciences, 12(4), 118–127. https://doi.org/10.5281/zenodo.1340591.
Robinson, T., McMullan, G., Marchant, R., & Nigam, P. (2001). Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresource Technology, 77(3), 247–255. https://doi.org/10.1016/S0960-8524(00)00080-8.
Rosenbrock, H. H. (1963). Some general implicit processes for the numerical solution of differential equations. The Computer Journal, 5(4), 329–330. https://doi.org/10.1093/comjnl/5.4.329.
Ruthven, D. S. J. (1984). Principles of adsorption and adsorption process. New York: Wiley.
Sausen, M. G., Scheufele, F. B., Alves, H. J., Vieira, M. G. A., da Silva, M. G. C., Borba, F. H., & Borba, C. E. (2018). Efficiency maximization of fixed-bed adsorption by applying hybrid statistical-phenomenological modeling. Separation and Purification Technology, 207, 477–488. https://doi.org/10.1016/j.seppur.2018.07.002, https://linkinghub.elsevier.com/retrieve/pii/S1383586618300224.
Scheufele, F. B., Módenes, A. N., Borba, C. E., Ribeiro, C., Espinoza-Qui nones, F.R., Bergamasco, R., & Pereira, N. C. (2016). Monolayer-multilayer adsorption phenomenological model: Kinetics, equilibrium and thermodynamics. Chemical Engineering Journal, 284, 1328–1341. https://doi.org/10.1016/j.cej.2015.09.085, https://linkinghub.elsevier.com/retrieve/pii/S138589471501356X.
Sheltami, R. M., Abdullah, I., Ahmad, I., Dufresne, A., & Kargarzadeh, H. (2012). Extraction of cellulose nanocrystals from mengkuang leaves (Pandanus tectorius). Carbohydrate Polymers, 88(2), 772–779. https://doi.org/10.1016/j.carbpol.2012.01.062, https://linkinghub.elsevier.com/retrieve/pii/S0144861712000823https://linkinghub.elsevier.com/retrieve/pii/S0144861712000823.
Sotelo, J. L., Ovejero, G., Rodríguez, A., Álvarez, S., & García, J. (2012). Removal of atenolol and isoproturon in aqueous solutions by adsorption in a fixed-bed column. Industrial and Engineering Chemistry Research, 51(13), 5045–5055. https://doi.org/10.1021/ie300334q.
Suzaki, P. Y. R., Munaro, M. T., Triques, C. C., Kleinübing, S. J., Fagundes Klen, M. R., Bergamasco, R., & de Matos Jorge, L. M. (2017). Phenomenological mathematical modeling of heavy metal biosorption in fixed-bed columns. Chemical Engineering Journal, 326, 389–400. https://doi.org/10.1016/j.cej.2017.05.157.
Tavlieva, M. P., Genieva, S. D., Georgieva, V. G., & Vlaev, L. T. (2015). Thermodynamics and kinetics of the removal of manganese(II) ions from aqueous solutions by white rice husk ash. Journal of Molecular Liquids, 211, 938–947. https://doi.org/10.1016/j.molliq.2015.08.015.
Thomas, H. C. (1944). Heterogeneous Ion Exchange in a Flowing System. Journal of the American Chemical Society, 66(10), 1664–1666. https://doi.org/10.1021/ja01238a017.
Tukarambai, M., & Venakateswarlu, P. (2020). A study of lead removal using sargassum tenerrimum (brown algae): Biosorption incolumn study. Materials Today: Proceedings, 27 (xxxx), 421–425. https://doi.org/10.1016/j.matpr.2019.11.254, https://linkinghub.elsevier.com/retrieve/pii/S2214785319339409https://linkinghub.elsevier.com/retrieve/pii/S2214785319339409.
Tural, B., Erta, E, Enez, B., Fincan, S. A., & Tural, S. (2017). Preparation and characterization of a novel magnetic biosorbent functionalized with biomass of Bacillus Subtilis : Kinetic and isotherm studies of biosorption processes in the removal of Methylene Blue. Journal of Environmental Chemical Engineering, 5(5), 4795–4802. https://doi.org/10.1016/j.jece.2017.09.019, https://linkinghub.elsevier.com/retrieve/pii/S2213343717304578.
Wang, W., Li, M., & Zeng, Q. (2012). Column adsorption of chromium(VI) by strong alkaline anion-exchange fiber. Journal of Applied Polymer Science, 126(5), 1733–1738. https://doi.org/10.1002/app.36634.
Wernke, G., Fagundes-Klen, M. R., Vieira, M. F., Suzaki, P. Y. R., de Souza, H. K. S., Shimabuku, Q. L., & Bergamasco, R. (2020). Mathematical modelling applied to the rate-limiting mass transfer step determination of a herbicide biosorption onto fixed-bed columns. Environmental Technology, 41(5), 638–648. https://doi.org/10.1080/09593330.2018.1508252.
Wernke, G., Triques, C. C., Suzaki, P. Y. R., Fagundes-Klen, M. R., & Bergamasco, R. (2020). Analysis of herbicide biosorption by means of a phenomenological mathematical distributed parameter model. Environmental Technology, 1–8. https://doi.org/10.1080/09593330.2020.1861107.
Yang, L., He, L., Chu, X., Wang, F., & Zhang, L. (2016). Dynamic separation and recovery of osmium using γ-nano-Al2O3 in a packed column adsorption system. Research on Chemical Intermediates, 42 (3), 2691–2705. https://doi.org/10.1007/s11164-015-2175-2.
Zhang, Z., O’Hara, I. M., Kent, G. A., & Doherty, W. O. S. (2013). Comparative study on adsorption of two cationic dyes by milled sugarcane bagasse. Industrial Crops and Products, 42(1), 41–49. https://doi.org/10.1016/j.indcrop.2012.05.008, https://linkinghub.elsevier.com/retrieve/pii/S0926669012002737https://linkinghub.elsevier.com/retrieve/pii/S0926669012002737.
Acknowledgments
The authors thank to CNPq for the financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Juchen, P.T., Veit, M.T., Gonçalves, G.d.C. et al. Biosorption of Dye by Malt Bagasse in a Fixed-Bed Column: Experimental and Theoretical Breakthrough Curves. Water Air Soil Pollut 232, 128 (2021). https://doi.org/10.1007/s11270-021-05041-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05041-2