Abstract
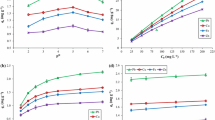
Materials of different origins have been applied as adsorbents in the treatment of metal-containing liquid solutions. In this study, we evaluated the potential of the dry biomass of two species of macrophytes in the adsorption of Cu2+ and Pb2+ ions from aqueous solution. The investigation included analyses of particle size, pH at the point of zero charge (pHPZC), kinetics of adsorption of the metal ions from the solution (equilibrium time × removal efficiency), and generation of adsorption isotherms. Results showed that particle size was not a determining factor for adsorption efficiency and capacity. The point of zero charge was similar between the adsorbents. Equilibrium times were 120 min for Pontederia rotundifolia and 240 min for Salvinia biloba. There was a high rate of removal of the metal ions from the solution, which occurred in the first minutes for S. biloba and in seconds for P. rotundifolia, at all initial concentrations tested. Equilibrium data fitted the Langmuir model. The adsorbent S. biloba is recommended for Cu2+ removal from solutions, while both adsorbents can be indicated for removal of Pb2+. The biomass of P. rotundifolia and S. biloba has promising for application in systems aimed at the treatment or filtering of liquid solutions with the Cu2+ and Pb2+ ions, features being highlighted by the fast removal speed, good adsorption capacity, and low cost.




Similar content being viewed by others
References
Aguiar, M. R. M. P., & Novaes, A. C. (2002). Removal of heavy metals from wastewaters by aluminosilicate. Química Nova, 25(6), 1145–1154. https://doi.org/10.1590/S0100-40422002000700015.
Ali, H., Khan, E., & Sajad, M. A. (2013). Phytoremediation of heavy metals-concepts and applications. Chemosphere, 91(7), 869–881. https://doi.org/10.1016/j.chemosphere.2013.01.075.
Ali, R. M., Hamad, H. A., Hussein, M. M., & Malash, G. F. (2016). Potential of using green adsorbent of heavy metal removal from aqueous solutions: adsorption kinetics, isotherm, thermodynamic, mechanism and economic analysis. Ecological Engineering, 91, 317–332. https://doi.org/10.1016/j.ecoleng.2016.03.015.
Andreazza, R., Camargo, F. A. O., Antoniolli, Z. I., Quadro, M. S., & Barcelos, A. A. (2013). Bioremediation of copper contaminated areas. Revista de Ciências Agrárias, 36(2), 127–136.
Azevedo, F. A., & Chasin, A. A. M. (2003). Metais: Gerenciamento da Toxicidade. São Paulo: Editora Atheneu.
Barać, N., Škrivanj, S., Mutić, J., Manojlović, D., Bukumirić, Z., Živojinović, D., et al. (2016). Heavy metals fractionation in agricultural soils of Pb/Zn mining region and their transfer to selected vegetables. Water. Water Air and Soil Pollution, 227(12), 481. https://doi.org/10.1007/s11270-016-3177-4.
Barros, A. J. M., Prasad, S., Leite, V. D., & Souza, A. G. (2006). Thermogravimetric study of the sorption process of heavy metals in organic solid waste. Revista Engenharia Sanitária, 11(2), 184–190. https://doi.org/10.1590/S1413-4152200600020001.
Bhargava, A., Carmona, F. F., Bhargava, M., & Srivastava, S. (2012). Approaches for enhanced phytoextraction of heavy metals. Journal of Environmental Management, 105, 103–120. https://doi.org/10.1016/j.jenvman.2012.04.002.
Boniolo, M. R., Yamaura, M., & Monteiro, R. A. (2010). Residual biomass for removal of uranyl ions. Química Nova, 33(3), 547–551. https://doi.org/10.1590/S0100-40422010000300010.
Bueno, C. I. C., & Carvalho, W. A. (2007). Lead (II) removal in discontinous systems by carbon activated by phosphoric acid and vapor. Química Nova, 30(8), 1911–1918. https://doi.org/10.1590/S0100-40422007000800022.
Bueno, B. M., Molina, F. F., Torem, M. L., de Mesquita, L. M. S., & Pino, G. A. H. (2009). Removal of Pb (II) from aqueous solutions by biosorption with R. opacus. International Engineering Journal, 62(4), 487–494. https://doi.org/10.1590/S0370-44672009000400012.
Bulgariu, D., & Bulgariu, L. (2013). Sorption of Pb (II) onto a mixture of algae waste biomass and anion exchanger resin in a packed-bed column. Bioresource Technology, 129, 374–380. https://doi.org/10.1016/j.biortech.2012.10.142.
Calfa, B. A., & Torem, M. L. (2007). Bioreagents—their use in the removal of heavy metals from liquid streams by biosorption/bioflotation. International Engineering Journal, 60(3), 537–542. https://doi.org/10.1590/S0370-44672007000300015.
Caliskan, N., Kul, A. R., Alkan, S., Sogut, E. G., & Alacabey, I. (2011). Adsorption of zinc (II) on diatomite: a Kinect and equilibrium study. Journal of Hazardous Materials, 193, 27–36. https://doi.org/10.1016/j.jhazmat.2011.06.058.
Chojnacka, K. (2010). Biosorption and bioaccumulation-the prospects for practical applications. Environment International, 36, 299–307. https://doi.org/10.1016/j.envint.2009.12.001.
Ciszewski, D., & Grygar, T. M. A. (2016). Review of flood-related storage and remobilization of heavy metal pollutants in river systems. Water, Air, and Soil Pollution, 227, 239. https://doi.org/10.1007/s11270-016-2934-8.
Coelho, G. F., Gonçalves Jr., A. C., de Sousa, R. F. B., Schwantes, D., Miola, A. J., & Domingues, C. V. R. (2014). Use of adsorption techniques utilizing agroindustrial waste in the removal of contaminants in waters. Journal of Agronomic Sciences, 3, 291–317.
CONAMA Resolution (2011). Provides for effluent release conditions and standards, complements and amends National Environmental Council Resolution N°. 357 of March 17, 2005. http://www.mma.gov.br/port/conama/legiabre.cfm?codlegi=646.
Conceição, F. T., Pichinelli, B. C., da Silva, M. S. G., Antunes, M. L. P. A., Lopes, M. S., & Moruzzi, R. B. (2017). Adsorption of Cd2+, Ni2+ and Zn2+ in aqueous solutions using anhydrite and red mud. Engenharia Sanitária e Ambiental, 22(4), 665–670. https://doi.org/10.1590/s1413-41522017146698.
Cooney, D. O. (1999). Adsorption design for wastewater treatment. Boca Raton: Editora CRC Press.
Della Vecchia, J. F., Cruz, C., Silva, A. F., Cerveira Jr., W. R., & Garlich, N. (2016). Macrophyte bioassay applications for monitoring pesticides in the aquatic environment. Planta Daninha, 34(3), 597–603. https://doi.org/10.1590/s0100-83582016340300021.
Esteves, F. A. (1998). Fundamentos de Limnologia. Rio de Janeiro: Interciência.
Ferreira, R. M., Domingues, A. L. C., & Stapelfeldt, D. M. A. (2017). Studies of selective adsorption, desorption and reuse of chemically altered biomass produced from aquatic macrophytes for treatment of metal-containing wastewater. Water, Science and Technology, 75, 2083–2093. https://doi.org/10.2166/wst.2017.090.
Fia, F. R. L., Matos, A. T., Fia, R., Borges, A. C., & Cecon, P. R. (2017). Effect of vegetation in constructed wetlands treating swine wastewater. Engenharia Sanitária e Ambiental, 22(2), 303–311. https://doi.org/10.1590/s1413-41522016123972.
França, J. B. A., Moraes, T. V., Vaz, D. C., Ferreira, A. A., & Soares, F. A. L. (2014). Tratamento de efluente doméstico com macrófitas aquáticas para reuso na fertirrigação. Brazilian Journal of Irrigation and Drainage, 1, 85–93. https://doi.org/10.15809/irriga.2014v1n1p85.
Freitas, F., Lunardi, S., Souza, L. B., Von Der Osten, J. C., Arruda, R. S., Andrade, R. L. T., & Battirola, L. D. (2018). Accumulation of copper by the aquatic macrophyte Salvinia biloba Raddi (Salviniaceae). Brazilian Journal of Biology, 78(1), 133–139. https://doi.org/10.1590/1519-6984.166377.
Gasper, J., & Pitol-Filho, L. (2016). Treatment of aqueous solutions of calcium ions using rice husks in batch and packed bed. Journal of Chemical Engineering and Chemistry, 2(1), 029–038. https://doi.org/10.18540/2446941601012016029.
Girard, J. E. (2013). Princípios de Química Ambiental. Rio de Janeiro: LTC.
Gonçalves-Junior, A. C. G., Selzlein, C., & Nacke, H. (2009). Uso de biomassa seca de aguapé (Eichhornia crassipes) visando à remoção de metais pesados de soluções contaminadas. Acta Scientiarum Technology, 31(1), 103–108. https://doi.org/10.4025/actascitechnol.v31i1.3166.
Gonçalves-Junior, A. C., Meneghel, A. P., Rubio, F., Strey, L., Dragunski, D. C., & Coelho, G. F. (2013). Applicability of Moringa oleifera lam. Pie as an adsorbent for removal of heavy metals from waters. Revista Brasileira de Engenharia Agrícola e Ambiental, 17(1), 94–99. https://doi.org/10.1590/S1415-43662013000100013.
Guimarães, M. G. Q., Moreira, A. D. R., & Petean Bove, C. P. (2017). Flora of Rio de Janeiro: Pontederiaceae. Rodriguésia, 68(1), 103–108. https://doi.org/10.1590/2175-7860201768118.
Guinesi, L. S., Esteves, A. A., & Cavalheiro, E. T. G. (2007). Adsorção de íons cobre (II) pela quitosana usando coluna em sistema sob fluxo hidrodinâmico. Quimica Nova, 30(4), 809–814. https://doi.org/10.1590/S0100-40422007000400010.
Han, S., Naito, W., & Masunaga, S. (2016). Impact of bioavailability incorporation on ecological risk assessment of nickel, copper, and zinc in surface waters. Water, Air, and Soil Pollution, 227(12), 480. https://doi.org/10.1007/s11270-016-3090-x.
Higarashi, M. M., Kunz, A., & Mattei, R. M. (2008). Adsorption applied to the removal of ammonia from pre-treated piggery wastewater. Química Nova, 31(5), 1156–1160. https://doi.org/10.1590/S0100-40422008000500043.
Honorato, A. C., Machado, J. M., Celante, G., Borges, W. G. P., Dragunski, D. C., & Caetano, J. (2015). Biosorption of methylene blue using agro-industrial residues. Revista Brasileira de Engenharia Agrícola e Ambiental, 19(7), 705–710. https://doi.org/10.1590/1807-1929/agriambi.v19n7p705-710.
Kelly-Vargas, K., Cerro-Lopez, M., Reyna-Tellez, S., Bandala, E. R., & Sanchez-Salas, J. L. (2012). Biosorption of heavy metals in polluted water, using different waste fruit cortex. Physics and Chemistry of the Earth, 37-39, 26–29. https://doi.org/10.1016/j.pce.2011.03.006.
Langmuir, I. (1916). The dissociation of hydrogen into atoms. III. The mechanism of the reaction. Journal of the American Chemical Society, 38(6), 1145–1156.
Li, Y., Yue, Q., & Gao, B. (2010). Adsorption kinetics and desorption of cu (II) and Zn (II) from aqueous solution onto humic acid. Journal of Hazardous Material, 178(1–3), 455–461. https://doi.org/10.1016/j.jhazmat.2010.01.103.
Lima, L. K. S., Silva, M. G. C., & Vieira, M. G. A. (2016). Study of binary and single biosorption by the floating aquatic macrophyte Salvinia natans. Brazilian Journal of Chemical Engineering, 33(3), 649–660. https://doi.org/10.1590/0104-6632.20160333s20150483.
Lucena, G. L., Silva, A. G., Honório, L. M. C., & Santos, V. D. (2012). Adsorption kinetics of copper (II) using bioadsorbents. Scientia Plena, 8, 1–6.
Lunardi, S., Freitas, F., de Souza, L. B., Von Der Osten, J. C., Arruda, R. S., Battirola, L. D., & Andrade, R. L. T. (2017). Effect of concentration and exposure time on copper accumulation in Eichhornia crassipes (Mart.) Solms. (Pontederiaceae). Scientific Electronic Archives, 10(6), 56–63.
Melo, D. Q., de Lima, A. C. A., de Barros, A. L., Vidal, C. B., Raulino, G. S. C., & Nascimento, R. F. (2014). Equilíbrio de Adsorção. In R. F. Nascimento, A. C. A. Lima, C. B. Vidal, D. Q. Melo, & G. S. C. Raulino (Eds.), Adsorção: Aspectos teóricos e Aplicações Ambientais (pp. 23–50). Fortaleza: Imprensa Universitária.
Mishra, S. P. (2014). Adsorption-desorption of heavy metal ions. Current Science, 107(4), 601–612.
Módenes, A. N., Pietrobelli, J. M. T. A., Quiñones, F. R. E., Suzaki, P. Y. R., Alflen, V. L., & Klen, M. R. S. (2009). Biosorption potential of zinc by Egeria dense macrophytes. Engenharia Sanitária e Ambiental, 14(4), 465–470. https://doi.org/10.1590/S1413-41522009000400006.
Módenes, A. N., Ross, A. A., Souza, B. V., Dotto, J., Geraldi, C. Q., Espinoza-Quiñones, F. R., & Kroumov, A. D. (2013). Biosorption of BF-4B reactive red dye by using leaves of macrophyte Eichhornia crassipes. International Journal Bioautomation, 17(1), 33–44.
Moreira, S. A., Sousa, F. W., Oliveira, A. G., Nascimento, R. F., & Brito, E. S. (2009). Metal removal from aqueous solution using cashew bagasse. Química Nova, 32(7), 1717–1722. https://doi.org/10.1590/S0100-40422009000700007.
Mudhoo, A., Garg, V. K., & Wang, S. (2012). Removal of heavy metals by biosorption. Environmental Chemistry Letters, 10(2), 109–117. https://doi.org/10.1007/s10311-011-0342-2.
Nair, C. S., & Kani, K. M. (2016). Phytoremediation of dairy effluent using aquatic macrophytes. International Journal of Scientific and Engineering Research, 7(4), 253–259.
Nascimento, H. C. E., de Andrade, I. M., Silva, M. F. S., & Matias, L. Q. (2013). Pontederiaceae in the coastal region of Piauí, Brazil. Rodriguésia, 64(3), 625–634. https://doi.org/10.1590/S2175-78602013000300013.
Nascimento, R. F., Lima, A. C. A., Vidal, C. B., Melo, D. Q., & Raulino, G. S. C. (2014a). Adsorção: Aspectos teóricos e aplicações ambientais. Fortaleza: Imprensa Universitária.
Nascimento, J. M., Silva, B. S., Chaves, M. D., & Oliveira, J. D. (2014b). Biossorção dos íons Cd2+ e Pb2+ utilizando a biomassa casca de pequi (Caryocar brasiliensi Camb) modificada com ácido nítrico. Revista de Ciências Ambientais, 8(1), 57–69.
Pietrobelli, J. M. T. A., Corrêa, A. R., Leichtweis, W. A., Pietrobelli, S., Módenes, A. N., Trigueros, D. E., et al. (2013). Avaliação do potencial de biossorção dos íons Cd (II), Cu (II) e Zn (II) pela macrófita Egeria densa. Revista Brasileira de Ciências Ambientais, 28, 1–9.
Pino, G. H., & Torem, M. L. (2011). Fundamental aspects of biosorption of non-ferrous metals. Tecnologia em Metalurgia Materiais e Mineração, 8(1), 57–63.
Pitelli, R. L. C. M., Pitelli-Merenda, A. M. C., Pitelli, R. A., Siqueira, R. C., Barbosa, H. O., & Jesus, L. (2014). Aquatic macrophytes community and colonization on Aimorés reservoir. Planta Daninha, 32(3), 475–482. https://doi.org/10.1590/S0100-83582014000300002.
Pitol-Filho, L. (2011). Sustainable applications of biomass: New perspectives. Revista da Unifebe, 9, 100–109.
Pompêo, M. (2008). Monitoramento e manejo de macrófitas aquáticas. Oecologia Brasiliensis, 12(3), 406–424. https://doi.org/10.11606/9788585658670.
Prado, J. (2006). Criptógamos do Parque Estadual das Fontes do Ipiranga, São Paulo, SP. Pteridophyta: Salviniaceae. Hoehnea, 33(1), 107–110.
Rajczykowski, K., & Loska, K. (2018). Stimulation of heavy metal adsorption process by using a strong magnetic field. Water, Air, and Soil Pollution, 229(20). https://doi.org/10.1007/s11270-017-3672-2.
Regalbuto, J. R., & Robles, J. (2004). The engineering of Pt/carbon catalyst preparation. Chicago: University of Illinois.
Rodrigues, R. F., Trevenzoli, R. L., Santos, L. R. G., Versiane, A. L., & Botaro, V. R. (2006). Heavy metals sorption on treated wood sawdust. Engenharia Sanitária e Ambiental, 11(1), 21–26. https://doi.org/10.1590/S1413-41522006000100004.
Rodrigues, A. C. D., Santos, A. M., Santos, F. S., Pereira, A. C. C., & Sobrinho, N. M. B. A. (2016). Response mechanisms of plants to heavy metal pollution: Possibility of using macrophytes for remediation of contaminated aquatic environments. Revista Virtual Química, 8, 262–276. https://doi.org/10.5935/1984-6835.20160017.
Rodrigues, A. C. D., Amaral Sobrinho, N. M. B., dos Santos, F. S., dos Santos, A. M., Pereira, A. C. C., & Lima, E. S. A. (2017). Biosorption of toxic metals by water lettuce (Pistia stratiotes) biomass. Water, Air, and Soil Pollution, 228(156), 1–11. https://doi.org/10.1007/s11270-017-3340-6.
Saier Jr., M. H., & Trevors, J. T. (2010). Phytoremediation. Water, Air, and Soil Pollution, 205, 61–63. https://doi.org/10.1007/s11270-008-9673-4.
Schneider, I. A. H., Rubio, J., & Smith, R. W. (2011). Biosorption of metals into plant biomass: Exchange adsorption or surface precipitation? International Journal of Mineral Processing, 62, 111–120. https://doi.org/10.1016/S0301-7516(00)00047-8.
Seolatto, A. A., Martins, T. D., Bergamasco, R., Tavares, C. R. G., Cossich, E. S., & da Silva, E. A. (2014). Biosorption study of Ni2+ and Cr3+ by Sargassum filipendula: Kinetics and equilibrium. Brazilian Journal of Chemical Engineering, 31(1), 211–227. https://doi.org/10.1590/S0104-66322014000100020.
Shinzato, M. C. (2007). Remoção de metais pesados em solução por zeólitas naturais: revisão crítica. Revista do Instituto Geológico, São Paulo, 27, 65–78.
Shinzato, M. C., Montanheiro, T. J., Janasi, V. A., Andrade, S., & Yamamoto, J. K. (2009). Removal of Pb2+ and Cr3+ from aqueous solution by natural zeolites associated with eruptive rocks from the Serra geral formation, paraná sedimentary basin. Química Nova, 32(8), 1989–1994. https://doi.org/10.1590/S0100-40422009000800002.
Silva, J. L. B. C., Pequeno, O. T. B. L., Rocha, L. K. Y. S., Araújo, E. C. O. A., Marciel, T. A. R., & Barros, A. J. M. B. (2014). Biosorption heavy metals: a review. Revista Saúde e Ciência, 3(3), 137–149.
Souza, J. I., Schoenlei-Crusius, I. H., Pires-Zottarelli, C. L. A., & Schoenlein, N. C. (2008). Copper, manganese and cadmium biosorption by Saprolegnia subterranea (Dissmann) R. L. Seym. and Pythium torulosum Coker & P. Patt. (Oomycet). Acta Botanica Brasilica, 22(1), 217–223. https://doi.org/10.1590/S0102-33062008000100021.
Souza, R. S., Carvalho, S. M. L., Garcia Júnior, M. R. L., & Sena, R. S. F. (2009). Chromium (VI) adsorption by GAC from diluted solutions in batch system and controlled ph. Acta Amazonica, 39(3), 661–668. https://doi.org/10.1590/S0044-59672009000300022.
Sprynskyy, M., Buszewski, B., Terzyk, A. P., & Namiesnik, J. (2006). Study of the selection mechanism of heavy metal (Pb2+, Cu2+, Ni2+, and Cd2+) adsorption on clinoptilolite. Journal of Colloid and Interface Science, 304, 21–28. https://doi.org/10.1016/j.jcis.2006.07.068.
Tasar, S., Kaya, F., & Ozer, A. (2014). Biosorption of lead (II) ions from aqueous solution by peanut shells: Equilibrium, thermodynamic and kinetic studies. Environmental Chemical Engineering, 2, 1018–1026. https://doi.org/10.1016/j.jece.2014.03.015.
Veit, M. T., Silva, E. A., Fagundes-Klen, M. R., Tavares, C. R. G., & Gonçalves, J. C. (2008). Biosorption of chromium (III) in fixed bed column. Estudos Tecnológicos, 4(2), 88–104.
Vidal, C. B., de Lima, A. C. A., Raulino, G. S. C., Melo, D. Q., & do Nascimento, R. F. (2014). Princípios básicos. In R. F. Nascimento, A. C. A. Lima, C. B. Vidal, D. Q. Melo, & G. S. C. Raulino (Eds.), Adsorção: Aspectos teóricos e Aplicações Ambientais (pp. 51–71). Fortaleza: Imprensa Universitária.
Volesky, B. (2004). Sorption and biosorption. Saint-Lambert: BV-Sorbex.
Volesky, B. (2007). Biosorption and me. Water Research, 41, 4017–4029. https://doi.org/10.1016/j.watres.2007.05.062.
Worch, E. (2012). Adsorption technology in water treatment: Fundamentals, processes, and modeling. Germany: Dresden University of Technology Institute of Water Chemistry.
Zhou, H. Y., & Wong, M. H. (2000). Mercury accumulation in freshwater fish with emphasis on the dietary influence. Water Research, 34(17), 4234–4242. https://doi.org/10.1016/S0043-1354(00)00176-7.
Acknowledgements
The authors thank Apolônia Grade (Orchids of the Amazon, Alta Floresta, MT) for the support in obtaining the plants for the experiments.
Funding
This work was financially supported by the Research Support Foundation of the State of Mato Grosso, FAPEMAT (227320/2015) by the Post-Graduate Program in Environmental Sciences, PPGCAM/UFMT-Sinop; by the Coordination for the Improvement of Higher Education Personnel (CAPES); by the Integrated Laboratory of Chemical Research, LIPEQ/UFMT-Sinop; and by the Funding of Innovation and Research (FINEP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Freitas, F., Battirola, L.D. & de Andrade, R.L.T. Adsorption of Cu2+ and Pb2+ Ions by Pontederia rotundifolia (L.f.) (Pontederiaceae) and Salvinia biloba Raddi (Salviniaceae) Biomass. Water Air Soil Pollut 229, 349 (2018). https://doi.org/10.1007/s11270-018-4005-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-018-4005-9