Abstract
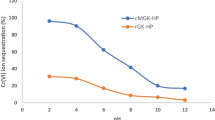
The effects of various experimental parameters on adsorption of Zn2+ metal ion from its aqueous solution by castor seed hull and also by activated carbon have been investigated using batch adsorption experiments. It has been found that the amount of zinc adsorbed per unit mass of the hull increases with the initial metal ion concentration, contact time, solution pH and with the amount of the adsorbent. Kinetic experiments clearly indicate that adsorption of zinc on both castor hull and activated carbon is a three-step process—a rapid adsorption of the metal ion, a transition phase, and an almost flat plateau. This has also been confirmed by the intraparticle diffusion model. It has also been found that the zinc adsorption process followed pseudo-second order kinetics. The kinetic parameters including rate constants have been determined at different initial metal ion concentration, pH, amount, and type of adsorbent, respectively. The Langmuir and Freundlich adsorption isotherm models have been used to interpret the equilibrium adsorption data. The Langmuir model yields better correlation coefficients. The monolayer adsorption capacities (q m ) of castor hull and activated carbon have been compared with those for others reported in the literature. The value of separation factor (R L ) derived from the Langmuir model gives an indication of favorable adsorption. Finally, from comparative studies, it has been found that castor hull is a potentially attractive adsorbent as compared to commercial activated carbon for the removal of zinc from aqueus effluents.














Similar content being viewed by others
References
Acharya, J., Sahu, J. N., Mohanty, C. R., & Meikap, B. C. (2009). Removal of Pb(II) from wastewater by activated carbon developed from tamarind wood activated with zinc chloride. Chemical Enineering Journal, 149, 249–262.
Ajmal, M., Rao, A. K. R., & Ahmad, J. (2000). Adsorption studies on Citrus reticulata: removal and recovery of Ni(II) from electroplating wastewater. Journal of Hazardous Materials, B79(2000), 117–131.
Annadurai, G., Juang, R., & Lee, D.-J. (2002). Use of cellulose-based wastes for adsorption of dyes from aqueous solutions. Journal of Hazardous Materials, 92, 263–274.
Arias, F., & Sen, T. K. (2009). Removal of zinc metal ion (Zn2+) from its aqueous solution by kaolin clay minerals: A kinetic and equilibrium study. Colloids and Surfaces, 348, 100–108.
Babel, S., & Kurniawan, T. A. (2003). Low-cost adsorbents for heavy metals uptake from contaminated water: a review. Journal of Hazardous Materials, 97, 219–225.
Bhattacharyya, K. G., & Sen Gupta, S. (2008). Adsorption of a few heavy metals on natural and modified kaolinite and montmorillonite: a review. Advances in Colloid and Interface Science, 140, 114–131.
Bhattacharya, A. K., Mandal, S. N., & Das, S. K. (2006). Adsorption of Zn(II) from aqueous solution by using different adsorbents. Chemical Enineering Journal, 123, 43–51.
Choi, J. Y., & Kim, D.-S. (2002). Adsorption behavior of zinc and cadmium on granular activated carbon in singular and binary systems and the influence of nitrilotricetic acid as a complexing agent. Journal of Environmental Science and Health, A37(9), 1701–1719.
Demiral, H., Demiral, L., Tumsek, F., & Karabacakoglu, B. (2008). Adsorption of chromium (VI) from aqueous solution by activated carbon derived from olive bagasse and applicability of different adsorption models. Chemical Enineering Journal., 144, 188–196.
Gharaibeh, S. H., Abu-EI-Shae, W. Y., & Al-Kofahi, M. M. (1998). Removal of selected heavy metals from aqueous solutions using processed solid residue of olive mill products. Water Research, 32, 498–502.
Gurses, A., Dogar, C., Yalcin, Y., Acikyildiz, M., Bayrak, R., & Karaca, S. (2006). The adsorption kinetics of the cationic dye, methylene blue, onto clay. Journal of Hazardous Materials, 131(1–3), 217–228.
Hall, K. R., Eagleton, L. C., Acrivos, A., & Vermeulen, T. (1966). Pore and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Industrial and Engineering Chemistry Fundamentals, 5, 212–223.
Jain, C. K. (2001). Adsorption of zinc onto bed sediments of the river Ganga: adsorption models and kinetics. Hydrological Sciences Journal, 46, 419–434.
Kandah, M. I. (2004). Zinc and cadmium adsorption on low-grade phosphate. Separation and Purification Technology., 35, 61–70.
Kargi, F., & Cikla, S. (2007). Kinetics of zinc(II) ion biosorption onto powdered waste sludge (PWS) at different operating conditions. Environmental Engineering Science, 24, 687–695.
Leyva-Ramos, R., Rangel-Mendez, J. R., Mendoza-Barron, J., Fuentes-Rubio, L., & Guerrero-Coronado, R. M. (1997). Adsorption of cadmium(II) from aqueous solution onto activated carbon. Water Science and Technology, 35, 205–211.
Marshall, W. E., & Johns, M. M. (1996). Agricultural by-products as metal adsorbents: sorption properties and resistance to mechanical abrasion. Journal of Chemical Technology and Biotechnology, 66, 192–198.
Meena, A. K., Mishra, G. K., Rai, P. K., Rajagopal, C., & Nager, P. N. (2005). Removal of heavy metal ions from aqueous solutions using carbon aerogel as an adsorbent. Journal of Hazardous Materials, 122, 161–170.
Mohan, D., & Singh, K. P. (2002). Single and multi-component adsorption of cadmium and zinc using activated carbon derived from bagasse—an agricultural waste. Water Research, 36, 2304–2318.
Monser, L., & Adhoum, N. (2002). Modified activated carbon for the removal of copper, zinc, chromium and cyanide from wastewater. Separation and Purfication Technology, 26, 137–146.
Naiya, T. K., Chowdhury, P., Bhattacharya, A. K., & Das, S. K. (2008). Saw dust and neem bark as low-cost natural biosorbent for adsorptive removal of Zn(II) and Cd(II) ions from aqueous solutions. Chemical Enineering Journal. doi:10.1016/J.cej.2008.08.002.
Norton, L., Baskaran, K., & McKenzie, T. (2004). Biosorption of zinc from aqueous solutions using biosolids. Advances in Environmental Research, 8, 629–635.
Oliveira, W. E., Franca, A. S., Oliveira, L. S., & Rocha, D. (2008). Untreated coffee husks as biosorbents for the removal of heavy metals from aqueous solutions. Journal of Hazardous Materials, 15, 1073–1081.
Ong, S. A., Toorisaka, E., Hirata, M., & Hano, T. (2005). The behavior of Ni(II), Cr(III) and Zn(II) in biological wastewater treatment process. Acta Hydrochimica Hydrobiologica, 33, 95–103.
Ricordel, S., Taha, S., Cisse, I., & Dorange, G. (2001). Heavy metals removal by adsorption onto peanut husks carbon: characterization, kinetic study and modeling. Separation and Puriication Technollogy, 24, 389–401.
Sen, T. K., & Khilar, K. C. (2006). Review on subsurface colloids and colloid-associated contaminant transport in saturated porous media. Advances in Colloid and Interface Science., 119, 71–96.
Sen, T. K., & Meimon, V. S. (2008). Removal of cadmium metal ion (Cd2+) from its aqueous solution by aluminium oxide (Al2O3): a kinetic and equilibrium study. Chemical Enineering Journal, 142, 256–262.
Sen, T. K., Mahajan, S. P., & Khilar, K. C. (2002). Adsorption of Cu2+ and Ni2+ on iron oxide and kaolin and its importance on Ni2+ transport in Porous Media. Colloids and Surfaces A, 211, 91–102.
Shukla, S. R., & Pai, R. S. (2005). Adsorption of Cu(II), Ni(II) and Zn(II) on dye loaded groundnut shells and sawdust. Separation and Purification Technology, 43, 1–8.
Snoeyink, V. L., & Jenkins, D. (1980). Water chemistry. New York, USA: Wiley.
Ucer, A., Uyanik, A., & Aygun, S. F. (2006). Adsorption of Cu(II), Cd(II), Zn(II), Mn(II) and Fe(III) ions by tannic acid immobilised activated carbon. Separation and Purification Technology, 47, 113–118.
Vadivelan, V., & Kumar, K. V. (2005). Equilibrium, kinetics, mechanism and process design for the sorption of methylene blue onto rice husk. Journal of Colloid and Interface Science, 286, 90–100.
Weber, W. J., & Morris, J. C. (1963). Kinetics of adsorption on carbon from solution. Journal of Sanitary Engineering Division, American Society of Civil Engineers., 89, 31–60.
Weng, C. H., & Huang, C. P. (2004). Adsorption characteristics of Zn(II) from dilute aqueous solution by fly ash. Colloids and Surfaces A, 247, 137–143.
Wu, F. C., Tseng, R. L., & Juang, R. S. (2002). Adsorption of dyes and humic acid from water using chitosan-encapsulated activated carbon. Journal of Chemical Technology and Biotechnology, 77, 1269–1279.
Zacaria, R., Gerente, C., Andres, Y., & Cloirec, P. L. (2002). Adsorption of several metal ions on to low cost biosorbent: kinetic and equilibrium studies. Environmental Science and Technology., 36, 2067–2073.
Zhu, C., Luan, Z., Wang, Y., & Shau, X. (2007). Removal of cadmium from aqueous solution by adsorption on granular red mud 9GRM. Separation and Purification Technology, 57, 167–175.
Acknowledgement
The authors gratefully acknowledge the Universiti Teknologi PETRONAS (UTP), Malaysia for providing necessary research facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohammod, M., Sen, T.K., Maitra, S. et al. Removal of Zn2+ from Aqueous Solution using Castor Seed Hull. Water Air Soil Pollut 215, 609–620 (2011). https://doi.org/10.1007/s11270-010-0503-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-010-0503-0