Abstract
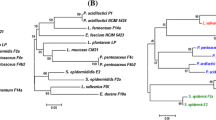
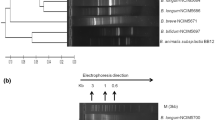
In this study, 105 bacterial colonies were isolated from the feces of newborn healthy Bos indicus calves and 37 isolates were confirmed using morphological, biochemical tests, and genus-specific PCR as lactobacilli. 11 isolates were then short-listed for in vitro probiotic testing based on their ability to dwell under acid and bile stress. Species-level identification using 16S rRNA gene sequencing revealed that they were Ligilactobacillus salivarius. These isolates flourished in 0.4% phenol, depicting resistance in adverse conditions encountered in the gastrointestinal tract. The results of cell surface hydrophobicity were found to be 74.50% for RBL12 and 62.62% for RBL09 in hexadecane and xylene, respectively, and that of auto-aggregation was highest in RBL26 (58.92%). These isolates also produced digestive enzymes like amylase, protease, and β-galactosidase. Further assays reiterated their antimicrobial and coaggregation potential against diarrhea-causing pathogens like Escherichia coli ATCC-25922 and Salmonella arizonae ATCC-13314. Biosafety assessment revealed that none of the tested isolates were hemolytic and mucinolytic in nature. Furthermore, the antioxidant potential of the isolates was also confirmed using 1,1‑diphenyl‑2‑picrylhydrazyl (DPPH) and ferric ion-reducing antioxidant power (FRAP) assay. Along with efficient utilization of inulin, isolates showed promising adhesion ability to the HT-29 cell line. The current findings hence conclude that these Lactobacillus isolates can be exploited as animal probiotics for potential application in young calves to foster gut health and immunity.











Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
Andriantsoanirina V, Allano S, Butel MJ, Aires J (2013) Tolerance of Bifidobacterium human isolates to bile, acid and oxygen. Anaerobe 21:39–42. https://doi.org/10.1016/j.anaerobe.2013.04.0051
Arena MP, Capozzi V, Russo P, Drider D, Spano G, Fiocco D (2018) Immunobiosis and probiosis: antimicrobial activity of lactic acid bacteria with a focus on their antiviral and antifungal properties. Appl Microbiol Biotechnol 102(23):9949–9958. https://doi.org/10.1007/s00253-018-9403-9
Balamurugan R, Chandragunasekaran AS, Chellappan G, Rajaram K, Ramamoorthi G, Ramakrishna BS (2014) Probiotic potential of lactic acid bacteria present in homemade curd in southern India. Indian J Med Res 140(3):345–355
Botes M, Loos B, van Reenen CA, Dicks LMT (2008) Adhesion of the probiotic strains Enterococcus mundtii ST4SA and Lactobacillus plantarum 423 to Caco-2 cells under conditions simulating the intestinal tract, andin the presence of antibiotics and anti-inflammatory medicaments. Arch Microbiol 190(5):573–584. https://doi.org/10.1007/s00203-008-0408-0
Caro-Quintero A, Ochman H (2015) Assessing the unseen bacterial diversity in microbial communities. Genome Biol Evol 7(12):3416–3425. https://doi.org/10.1093/gbe/evv234
Charteris WP, Kelly PM, Morelli L, Collins JK (1998) Antibiotic susceptibility of potentially probiotic Lactobacillus species. J Food Prot 61(12):1636–1643
CLSI (2015) Performance standards for antimicrobial susceptibility testing; Twenty-fifth informational supplement. CLSI Document M100-S25https://file.qums.ac.ir/repository/mmrc/CLSI2015.pdf
Collado MC, Meriluoto J, Salminen S (2008) Adhesion and aggregation properties of probiotic and pathogen strains. Eur Food Res Technol 226(5):1065–1073. https://doi.org/10.1007/s00217-007-0632-x
Coman MM, Verdenelli MC, Cecchini C et al (2019) Probiotic characterization of Lactobacillus isolates from canine faeces. J Appl Microbiol 126(4):1245–1256. https://doi.org/10.1111/jam.14197
Das D, Goyal A (2015) Antioxidant activity and γ-aminobutyric acid (GABA) producing ability of probiotic Lactobacillus plantarum DM5 isolated from Marcha of Sikkim. LWT 61(1):263–268. https://doi.org/10.1016/j.lwt.2014.11.013
Das S, Mishra BK, Hati S (2020) Techno-functional characterization of indigenous Lactobacillus isolates from the traditional fermented foods of Meghalaya, India. Curr Res Food Sci 3:9–18. https://doi.org/10.1016/j.crfs.2020.01.002
dos Santos KMO, Vieira ADS, Buriti FCA, do Nascimento JCF, de Melo MES, Bruno LM, de Fátima Borges M, Rocha CRC, de Souza Lopes AC, de Melo Franco BDG, Todorov SD (2015) Artisanal Coalho cheeses as source of beneficial Lactobacillus plantarum and Lactobacillus rhamnosus strains. Dairy Sci Technol 95(2), 209-230
Dowarah R, Verma AK, Agarwal N (2017) The use of Lactobacillus as an alternative of antibiotic growth promoters in pigs: A review. Anim Nutr 3(1):1–6. https://doi.org/10.1016/j.aninu.2016.11.002
Duary RK, Rajput YS, Batish VK, Grover S (2011) Assessing the adhesion of putative indigenous probiotic lactobacilli to human colonic epithelial cells. Indian J Med Res 134(5):664–671. https://doi.org/10.4103/0971-5916.90992
Dubernet S, Desmasures N, Guéguen M (2002) A PCR-based method for identification of lactobacilli at the genus level. FEMS Microbiol Lett 214(2):271–275. https://doi.org/10.1111/j.1574-6968.2002.tb11358.x
Ekmekci H, Aslim B, Ozturk S (2009) Characterization of vaginal lactobacilli coaggregation ability with Escherichia coli. Microbiol Immunol 53(2):59–65. https://doi.org/10.1111/j.1348-0421.2009.00115.x
Figueroa-González I, Rodríguez-Serrano G, Gómez-Ruiz L, García-Garibay M, Cruz-Guerrero A (2019) Prebiotic effect of commercial saccharides on probiotic bacteria isolated from commercial products. Food Sci Technol 39(3):747–753. https://doi.org/10.1590/fst.07318
Flint HJ, Duncan SH, Scott KP, Louis P (2007) Interactions and competition within the microbial community of the human colon: links between diet and health. Environ Microbiol 9(5):1101–1111. https://doi.org/10.1111/j.1462-2920.2007.01281.x
Gheytanchi E, Heshmati F, Shargh BK, Nowroozi J, Movahedzadeh F (2010) Study on β-galactosidase enzyme produced by isolated lactobacilli from milk and cheese. Afr J Microbiol Res 4:454–458
Gilliland SE, Staley TE, Bush LJ (1984) Importance of bile tolerance of Lactobacillus acidophilus used as a dietary adjunct. J Dairy Sci 67(12):3045–3051. https://doi.org/10.3168/jds.S0022-0302(84)81670-7
Grujović MŽ, Mladenović KG, Nikodijević DD et al (2019) Autochthonous lactic acid bacteria-presentation of potential probiotics application. Biotechnol Lett 41:1319–1331. https://doi.org/10.1007/s10529-019-02729-8
Gueimonde M, Sánchez B, de los Reyes-Gavilán C, Margolles A, (2013) Antibiotic resistance in probiotic bacteria. Front Microbiol 4:202. https://doi.org/10.3389/fmicb.2013.00202
Gupta M et al (2021) Functional and probiotic characterization of Ligilactobacillus salivarius CPN60 isolated from calf faeces and its appraisal in rats. J Biosci Bioeng 132(6):575–584. https://doi.org/10.1016/j.jbiosc.2021.09.001
Halder D, Mandal M, Chatterjee SS, Pal NK, Mandal S (2017) Indigenous probiotic Lactobacillus isolates presenting antibiotic like activity against human pathogenic bacteria. Biomedicines 5(2). https://doi.org/10.3390/biomedicines5020031
Hamasalim HJ (2016) Synbiotic as feed additives relating to animal health and performance. Adv Microbiol 6(4):288–302. https://doi.org/10.4236/aim.2016.640283
Hassanzadazar H, Ehsani A, Mardani K, Hesari J (2012) Investigation of antibacterial, acid and bile tolerance properties of lactobacilli isolated from Koozeh cheese. Vet Res Forum 3:181–185
Hill C, Guarner F, Reid G et al (2014) The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol 11(8):506–514. https://doi.org/10.1038/nrgastro.2014.66
Huebner J, Wehling RL, Hutkins RW (2007) Functional activity of commercial prebiotics. Int Dairy J 17(7):770–775. https://doi.org/10.1016/j.idairyj.2006.10.006
Indian Council of Medical Research Task Force, Co-ordinating Unit ICMR, Co-ordinating Unit DBT (2011) ICMR-DBT guidelines for evaluation of probiotics in food. Indian J Med Res 134(1):22–25
Jiang X, Xu HJ, Cui ZQ, Zhang YG (2020) Effects of supplementation with Lactobacillus plantarum 299v on the performance, blood metabolites, rumen fermentation and bacterial communities of preweaning calves. Livest Sci 239.
Johan SO (2020) Gut health, stress, and immunity in neonatal dairy calves: the host side of host-pathogen interactions. J Anim Sci Biotechnol 11(1):105. https://doi.org/10.1186/s40104-020-00509-3
Kaplan H, Hutkins RW (2003) Metabolism of fructooligosaccharides by Lactobacillus paracasei 1195. Appl Environ Microbiol 69(4):2217–2222. https://doi.org/10.1128/AEM.69.4.2217-2222.2003
Kim MJ, Ku S, Kim SY, Lee HH, Jin H, Kang S, Li R, Johnston TV, Park MS, Ji GE (2018) Safety evaluations of Bifidobacterium bifidum BGN4 and Bifidobacterium longum BORI. Int J Mol Sci 19(5):1422
Kim PI, Jung MY, Chang YH, Kim S, Kim SJ, Park YH (2007) Probiotic properties of Lactobacillus and Bifidobacterium strains isolated from porcine gastrointestinal tract. Appl Microbiol Biotechnol 74(5):1103–1111. https://doi.org/10.1007/s00253-006-0741-7
Kumar M, Sarma DK, Shubham S, Kumawat M, Verma V, Nina PB, Jp D, Kumar S, Singh B, Tiwari RR (2021a) Futuristic Non-antibiotic Therapies to Combat Antibiotic Resistance: A Review. Front Microbiol 12:16. https://doi.org/10.3389/fmicb.2021.609459
Kumar S, Pattanaik AK, Jadhav SE (2021b) Potent health-promoting effects of a synbiotic formulation prepared from Lactobacillus acidophilus NCDC15 fermented milk and Cichorium intybus root powder in Labrador dogs. Curr Res Biotechnol 3:209–214. https://doi.org/10.1016/j.crbiot.2021.06.001
Kumar S, Pattanaik AK, Sharma S, Jadhav SE, Dutta N, Kumar A (2017) Probiotic potential of a Lactobacillus bacterium of canine faecal-origin and its impact on select gut health indices and immune response of dogs. Probiotics Antimicrob Proteins 9(3):262–277. https://doi.org/10.1007/s12602-017-9256-z
Kumar S, Pattanaik AK, Sharma S, Jadhav SE (2016) Species-specific probiotics Lactobacillus johnsonii CPN23 supplementation modulates blood biochemical profile and erythrocytic antioxidant indices in Labrador dogs. Indian J Anim Sci 86:918–924
Kumar S, Varada VV, Banakar PS, Tyagi N, Chouraddi R, Hogarehalli Mallapa R, Tyagi AK (2022) Screening and characterization of Sahiwal cattle calves-origin lactic acid bacteria based on desired probiotic attributes for potential application. Anim Biotechnol. https://doi.org/10.1080/10495398.2022.2043885
Kurdi P, Kawanishi K, Mizutani K, Yokota A (2006) Mechanism of growth inhibition by free bile acids in lactobacilli and bifidobacteria. J Bacteriol 188(5):1979–1986. https://doi.org/10.1128/JB.188.5.1979-1986.2006
Lähteinen T, Malinen E, Koort JMK et al (2010) Probiotic properties of Lactobacillus isolates originating from porcine intestine and feces. Anaerobe 16(3):293–300. https://doi.org/10.1016/j.anaerobe.2009.08.002
Lee BJ, Kim JS, Kang YM et al (2010) Antioxidant activity and γ-aminobutyric acid (GABA) content in sea tangle fermented by Lactobacillus brevis BJ20 isolated from traditional fermented foods. Food Chem 122(1):271–276. https://doi.org/10.1016/j.foodchem.2010.02.071
Li M, Wang Y, Cui H, Li Y, Sun Y, Qiu HJ (2020) Characterization of lactic acid bacteria isolated from the gastrointestinal tract of a wild boar as potential probiotics. Front Vet Sci 7:49. https://doi.org/10.3389/fvets.2020.00049
Liu C, Zhang ZY, Dong K, Yuan JP, Guo XK (2009) Antibiotic resistance of probiotic strains of lactic acid bacteria isolated from marketed foods and drugs. Biomed Environ Sci 22(5):401–412. https://doi.org/10.1016/S0895-3988(10)60018-9
Mallappa RH, Singh DK, Rokana N, Pradhan D, Batish VK, Grover S (2019) Screening and selection of probiotic Lactobacillus strains of Indian gut origin based on assessment of desired probiotic attributes combined with principal component and heatmap analysis. LWT 105:272–281. https://doi.org/10.1016/j.lwt.2019.02.002
Markowiak P, Śliżewska K (2017) Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 9(9). https://doi.org/10.3390/nu9091021
Mishra V, Shah C, Mokashe N, Chavan R, Yadav H, Prajapati J (2015) Probiotics as potential antioxidants: a systematic review. J Agric Food Chem 63(14):3615–3626. https://doi.org/10.1021/jf506326t
Monteagudo-Mera A, Rastall RA, Gibson GR, Charalampopoulos D, Chatzifragkou A (2019) Adhesion mechanisms mediated by probiotics and prebiotics and their potential impact on human health. Appl Microbiol Biotechnol 103(16):6463–6472. https://doi.org/10.1007/s00253-019-09978-7
Moussavi M, Adams MC (2010) An in vitro study on bacterial growth interactions and intestinal epithelial cell adhesion characteristics of probiotic combinations. Curr Microbiol 60(5):327–335. https://doi.org/10.1007/s00284-009-9545-1
Nami Y, Vaseghi Bakhshayesh R, Mohammadzadeh Jalaly H, Lotfi H, Eslami S, Hejazi MA (2019) Probiotic properties of Enterococcus isolated from artisanal dairy products. Front Microbiol 10:300. https://doi.org/10.3389/fmicb.2019.00300
Neumann B, Pospiech A, Schairer HU (1992) Rapid isolation of genomic DNA from gram-negative bacteria. Trends Genet 8(10):332–333. https://doi.org/10.1016/0168-9525(92)90269-a
Padmavathi T, Bhargavi R, Priyanka PR, Niranjan NR, Pavitra PV (2018) Screening of potential probiotic lactic acid bacteria and production of amylase and its partial purification. J Genet Eng Biotechnol 16(2):357–362. https://doi.org/10.1016/j.jgeb.2018.03.005
Palframan R, Gibson GR, Rastall RA (2003) Development of a quantitative tool for the comparison of the prebiotic effect of dietary oligosaccharides. Lett Appl Microbiol 37(4):281–284. https://doi.org/10.1046/j.1472-765x.2003.01398.x
Parsana Y, Yadav M, Kumar S (2023) Microencapsulation in the chitosan-coated alginate-inulin matrix of Limosilactobacillus reuteri SW23 and Lactobacillus salivarius RBL50 and their characterization. Carbohydr Pol Technol Appl 100285.
Patil MM, Pal A, Anand T, Ramana KV (2010) Isolation and characterization of lactic acid bacteria from curd and cucumber. Indian J Biotechnol 9:166–172
Prabhurajeshwar C, Chandrakanth K (2019) Evaluation of antimicrobial properties and their substances against pathogenic bacteria in-vitro by probiotic Lactobacilli strains isolated from commercial yoghurt. Clin Nutr Exp 23:97–115. https://doi.org/10.1016/j.yclnex.2018.10.001
Prete R, Garcia-Gonzalez N, Di Mattia CD, Corsetti A, Battista N (2020) Food-borne Lactiplantibacillus plantarum protect normal intestinal cells against inflammation by modulating reactive oxygen species and IL-23/IL-17 axis. Sci Rep 10(1):16340. https://doi.org/10.1038/s41598-020-73201-1
Reddy PRK, Ravi Kanth MMMY, Elghandour MMMY, Salem, AZM, Yasaswin, D, Reddy PPR, Reddy AN, Hyder I AZM, et al (2020) Plant secondary metabolites as feed additives in calves for antimicrobial stewardship. Anim Feed Sci Technol 264:114469. doi: https://doi.org/10.1016/j.anifeedsci.2020.114469
Tynkkynen KV, Singh PV (1998) Vancomycin resistance factor of Lactobacillus rhamnosus GG in relation to enterococcal vancomycin resistance (van) genes. Int J Food Microbiol 41(3):195–204. https://doi.org/10.1016/s0168-1605(98)00051-8
Sharma K, Attri S, Goel G (2019) Selection and evaluation of probiotic and functional characteristics of autochthonous lactic acid bacteria isolated from fermented wheat flour dough babroo. Probiotics Antimicrob Proteins 11(3):774–784. https://doi.org/10.1007/s12602-018-9466-z
Sharma AN, Kumar S, Tyagi AK (2018) Effects of mannan-oligosaccharides and Lactobacillus acidophilus supplementation on growth performance, nutrient utilization and faecal characteristics in Murrah buffalo calves. J Anim Physiol Anim Nutr 102:679–689. https://doi.org/10.1111/jpn.12878
Sharma P, Tomar SK, Sangwan V, Goswami P, Singh R (2016) Antibiotic resistance of Lactobacillus sp. isolated from commercial probiotic preparations. J Food Saf 36(1):38–51. https://doi.org/10.1111/jfs.12211
Shin JH, Lee JS, Seo JG (2015) Assessment of cell adhesion, cell surface hydrophobicity, autoaggregation, and lipopolysaccharide-binding properties of live and heat-killed Lactobacillus acidophilus CBT LA1. Korean J Microbiol 51(3):241–248. https://doi.org/10.7845/kjm.2015.5029
Singh A, Kumar S, Vinay VV et al (2021) Autochthonous Lactobacillus spp. isolated from Murrah buffalo calves show potential application as probiotic. Curr Res Biotechnol 3:109–119. https://doi.org/10.1016/j.crbiot.2021.04.002
Tang W, Xing Z, Li C, Wang J, Wang Y (2017) Molecular mechanisms and in vitro antioxidant effects of Lactobacillus plantarum MA2. Food Chem 221:1642–1649
Tauno M (2016) Statistical analysis of multivariate data in bioinformatics. Published online. http://dspace.ut.ee/bitstream/handle/10062/50409/metsalu_tauno.pdf
Todorov SD, Dicks LMT (2008) Evaluation of lactic acid bacteria from kefir, molasses and olive brine as possible probiotics based on physiological properties. Ann Microbiol 58(4):661. https://doi.org/10.1007/BF03175572
Touré R, Kheadr E, Lacroix C, Moroni O, Fliss I (2003) Production of antibacterial substances by bifidobacterial isolates from infant stool active against Listeria monocytogenes. J Appl Microbiol 95(5):1058–1069. https://doi.org/10.1046/j.1365-2672.2003.02085.x
Tyagi AK, Kumar S, Choudhury PK, Tyagi B, Tyagi N (2020) Conjugated linoleic acid producing potential of lactobacilli isolated from goat (AXB) rumen fluid samples. Asian-Australas J Anim Sci 33(8):1233–1241. https://doi.org/10.5713/ajas.19.0080
USDA (2014) Dairy Cattle Management Practices in the United States, USDA–APHIS–VS–CEAH–NAHMS Fort Collins CO 6920216
Varada VV, Tyagi AK, Banakar PS et al (2022) Autochthonous Limosilactobacillus reuteri BFE7 and Ligilactobacillus salivarius BF17 probiotics consortium supplementation improves performance, immunity, and selected gut health indices in Murrah buffalo calves. Vet Res Commun. https://doi.org/10.1007/s11259-022-09896-6
Xanthopoulos V, Litopoulou-Tzanetaki E, Tzanetakis N (2000) Characterization of Lactobacillus isolates from infant faeces as dietary adjuncts. Food Microbiol 17(2):205–215. https://doi.org/10.1006/fmic.1999.0300
Zhou JS, Gopal PK, Gill HS (2001) Potential probiotic lactic acid bacteria Lactobacillus rhamnosus (HN001), Lactobacillus acidophilus (HN017) and Bifidobacterium lactis (HN019) do not degrade gastric mucin in vitro. Int J Food Microbiol 63(1–2):81–90. https://doi.org/10.1016/s0168-1605(00)00398-6
Zhou JS, Pillidge CJ, Gopal PK, Gill HS (2005) Antibiotic susceptibility profiles of new probiotic Lactobacillus and Bifidobacterium strains. Int J Food Microbiol 98(2):211–217. https://doi.org/10.1016/j.ijfoodmicro.2004.05.011
Acknowledgements
The authors would like to express their sincere gratitude to Director, ICAR-NDRI, Karnal, for providing all the necessary facilities to carry out the research work.
Funding
This work was supported by grants from ICAR-NCVTCC, Hisar, India (F.No: NCVTC/Budget/2019–20/860).
Author information
Authors and Affiliations
Contributions
RC: Writing ‐ review & editing, Methodology. SK: Conceptualization, Funding acquisition, Project administration, Writing ‐ review & editing, Formal analysis. BK: Writing – review & editing, Software. MB: Methodology, Formal analysis. VV: Writing ‐ review & editing, Software. NT: review the manuscript. RHM: Conceptualization, Formal analysis, Software. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The experiments were done in compliance with appropriate rules and guidelines laid down by the Institutional Animal Ethics Committee (IAEC) registered (Registration number: 1705/GO/ac/13/CPCSEA) Committee for the Purpose of Control and Supervision of Experiments in Animals (CPCSEA), New Delhi, Ministry of Environment, Forests and Climate Change, Government of India.
Consent for publication
All authors give their consent for the publication of this manuscript.
Consent to participate
All authors give their consent to participate in the publication of this manuscript.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11259_2023_10077_MOESM1_ESM.jpg
Supplementary file1 Survivability percentage of lactobacilli isolatesexposed to acid (pH 2) and bile (Ox-bile 0.3%); Values are mean ± S.E.M. ofthree replicates (JPG 29 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chouraddi, R., Kumar, S., Kumar, B. et al. Techno-functional characterization of fecal lactobacilli isolates of Bos indicus calves for probiotic properties. Vet Res Commun 47, 1285–1302 (2023). https://doi.org/10.1007/s11259-023-10077-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10077-2