Abstract
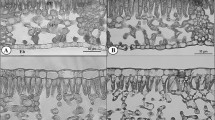
Plants usually respond to the changes of growth irradiance by a combination of the physiological modifications in their preexisting leaves and the production of new leaves. However, those with a determinate growth habit produce certain number of leaves in a growing season and cannot produce new leaves when light condition changes. We used an epiphytic orchid with only one leaf produced every growing season to examine whether and how determinate growth species adapt to changing environments after their preexisting leaves mature. Leaf photosynthesis and anatomy of Pleione aurita were investigated at full expansion and at 40 days after the fully expanded leaves were transferred from high to low light or from low to high light. Leaves show large physiological and morphological plasticity to light gradients at full expansion and the transferred leaves exhibited multiple physiological modifications, including reallocation of nitrogen between light harvesting and carbon fixation, and enhancement of thermal dissipation in their new environments, to optimize carbon assimilation and avoid photoinhibition. Irrespective of the various changes either to shade or sun, the sole preexisting leaf could not fully acclimate to new light environments due to the mesophyll thickness constraint. This leads to the consequence that only plants exposed to high light throughout the experiment had a positive annual biomass gain. Our results highlighted the importance of new leaf production in the carbon accumulation during photosynthetic light acclimation, and contribute new insights of epiphytes physiological responses to their highly dynamic arboreal habitat.




Similar content being viewed by others
References
Anderson JM, Chow WS, Park YI (1995) The grand design of photosynthesis: acclimation of the photosynthetic apparatus to environmental cues. Photosynth Res 46:129–139. doi:10.1007/bf00020423
Avalos G, Mulkey SS (1999) Photosynthetic acclimation of the liana Stigmaphyllon lindenianum to light changes in a tropical dry forest canopy. Oecologia 120:475–484. doi:10.1007/s004420050880
Baker NR, Rosenqvist E (2004) Applications of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities. J Exp Bot 55:1607–1621. doi:10.1093/Jxb/Erh196
Benzing DH (1998) Vulnerabilities of tropical forests to climate change: the significance of resident epiphytes. Clim Change 39:519–540. doi:10.1023/A:1005312307709
Boardman N (1977) Comparative photosynthesis of sun and shade plants. Annu Rev Plant Phys 28:355–377
Brooks JR, Hinckley TM, Sprugel DG (1994) Acclimation responses of mature Abies amabilis sun foliage to shading. Oecologia 100:316–324. doi:10.1007/bf00316960
Cabral JS, Petter G, Mendieta-Leiva G, Wagner K, Zotz G, Kreft H (2015) Branchfall as a demographic filter for epiphyte communities: lessons from forest floor-based sampling. PLoS ONE 10(6):e0128019. doi:10.1371/journal.pone.0128019
Cervantes SE, Graham EA, Andrade JL (2005) Light microhabitats, growth and photosynthesis of an epiphytic bromeliad in a tropical dry forest. Plant Ecol 179:107–118. doi:10.1007/s11258-004-5802-3
Chen X, Cribb PJ, Gale SW (2009) Pleione D. Don. In: Wu ZY, Raven PH, Hong DY (eds) Flora of China, vol 25. Missouri Botanical Garden Press, St.Louis, pp 325–333
Cribb P, Butterfield I (1999) The Genus Pleione, 2nd edn. Royal Botanic Gardens, Kew, Richmond
Einzmann HJR, Beyschlag J, Hofhansl F, Wanek W, Zotz G (2015) Host tree phenology affects vascular epiphytes at the physiological, demographic and community level. Aob Plants 7. doi:10.1093/aobpla/plu073
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Hietz P (1997) Population dynamics of epiphytes in a Mexican humid montane forest. J Ecol 85:767–775. doi:10.2307/2960600
Hikosaka K, Terashima I (1995) A model of the acclimation of photosynthesis in the leaves of C3 plants to sun and shade with respect to nitrogen use. Plant Cell Environ 18:605–618. doi:10.1111/j.1365-3040.1995.tb00562.x
Ida TY, Kudo G (2010) Seasonal patterns of carbon assimilation and allocation of a summer-green forest herb, Parasenecio auriculata (Senecioneae; Asteraceae). Plant Ecol 210:181–193. doi:10.1007/s11258-010-9748-3
Inskeep WP, Bloom PR (1985) Extinction coefficients of chlorophyll a and b in N,N-dimethylformamide and 80% acetone. Plant Physiol 77:483–485. doi:10.1104/Pp.77.2.483
Ishii H, Ohsugi Y (2011) Light acclimation potential and carry-over effects vary among three evergreen tree species with contrasting patterns of leaf emergence and maturation. Tree Physiol 31:819–830. doi:10.1093/treephys/tpr079
Jahns P, Holzwarth AR (2012) The role of the xanthophyll cycle and of lutein in photoprotection of photosystem II. Biochim Biophys Acta 1817:182–193. doi:10.1016/j.bbabio.2011.04.012
Kamaluddin M, Grace J (1993) Growth and photosynthesis of tropical forest tree seedlings (Bischofia javanica Blume) as influenced by a change in light availability. Tree Physiol 13:189–201
Kursar TA, Coley PD (1999) Contrasting modes of light acclimation in two species of the rainforest understory. Oecologia 121:489–498. doi:10.1007/s004420050955
Laube S, Zotz G (2003) Which abiotic factors limit vegetative growth in a vascular epiphyte? Funct Ecol 17:598–604. doi:10.1046/j.1365-2435.2003.00760.x
Lawlor DW, Tezara W (2009) Causes of decreased photosynthetic rate and metabolic capacity in water-deficient leaf cells: a critical evaluation of mechanisms and integration of processes. Ann Bot-Lond 103:561–579. doi:10.1093/aob/mcn244
Matelson TJ, Nadkarni NM, Longino JT (1993) Longevity of fallen epiphytes in a neotropical montane forest. Ecology 74:265–269. doi:10.2307/1939523
Matsubara S, Krause GH, Aranda J, Virgo A, Beisel KG, Jahns P, Winter K (2009) Sun-shade patterns of leaf carotenoid composition in 86 species of neotropical forest plants. Funct Plant Biol 36:20–36. doi:10.1071/FP08214
Maxwell C, Griffiths H, Young AJ (1994) Photosynthetic acclimation to light regime and water stress by the C3-CAM epiphyte Guzmania monostachia: gas-exchange characteristics, photochemical efficiency and the xanthophyll cycle. Funct Ecol 8:746–754. doi:10.2307/2390234
Maxwell K, Marrison JL, Leech RM, Griffiths H, Horton P (1999) Chloroplast acclimation in leaves of Guzmania monostachia in response to high light. Plant Physiol 121:89–95. doi:10.1104/pp.121.1.89
Mondragón D, Valverde T, Hernández-Apolinar M (2015) Population ecology of epiphytic angiosperms: a review. Trop Ecol 56:1–39
Niinemets U, Cescatti A, Rodeghiero M, Tosens T (2006) Complex adjustments of photosynthetic potentials and internal diffusion conductance to current and previous light availabilities and leaf age in Mediterranean evergreen species Quercus ilex. Plant Cell Environ 29:1159–1178. doi:10.1111/j.1365-3040.2006.01499.x
Oguchi R, Hikosaka K, Hirose T (2003) Does the photosynthetic light-acclimation need change in leaf anatomy? Plant Cell Environ 26:505–512. doi:10.1046/j.1365-3040.2003.00981.x
Oguchi R, Hikosaka K, Hirose T (2005) Leaf anatomy as a constraint for photosynthetic acclimation: differential responses in leaf anatomy to increasing growth irradiance among three deciduous trees. Plant Cell Environ 28:916–927. doi:10.1111/j.1365-3040.2005.01344.x
Oguchi R, Hikosaka K, Hiura T, Hirose T (2006) Leaf anatomy and light acclimation in woody seedlings after gap formation in a cool-temperate deciduous forest. Oecologia 149:571–582. doi:10.1007/s00442-006-0485-1
Padmawathe R, Qureshi Q, Rawat GS (2004) Effects of selective logging on vascular epiphyte diversity in a moist lowland forest of Eastern Himalaya, India. Biol Conserv 119:81–92. doi:10.1016/j.biocon.2003.10.024
Pires MV, de Almeida AAF, Abreu PP, Silva DD (2012) Does shading explain variation in morphophysiological traits of tropical epiphytic orchids grown in artificial conditions? Acta Physiol Plant 34:2155–2164. doi:10.1007/s11738-012-1016-9
Schmidt G, Zotz G (2001) Ecophysiological consequences of differences in plant size: in situ carbon gain and water relations of the epiphytic bromeliad, Vriesea sanguinolenta. Plant Cell Environ 24:101–111. doi:10.1046/j.1365-3040.2001.00658.x
Shimizu M, Ishida A, Tange T, Yagi H (2006) Leaf turnover and growth responses of shade-grown saplings of four Shorea rain forest species to a sudden increase in light. Tree Physiol 26:449–457
Sims DA, Pearcy RW (1992) Response of leaf anatomy and photosynthetic capacity in Alocasia macrorrhiza (Araceae) to a transfer from low to high light. Am J Bot 79:449–455. doi:10.2307/2445158
Wagner K, Bogusch W, Zotz G (2013) The role of the regeneration niche for the vertical stratification of vascular epiphytes. J Trop Ecol 29:277–290. doi:10.1017/S0266467413000291
Walker DA (1989) Automated measurement of leaf photosynthetic O2 evolution as a function of photon flux density. Philos Trans R Soc B 323:313–326. doi:10.1098/rstb.1989.0013
Walters RG (2005) Towards an understanding of photosynthetic acclimation. J Exp Bot 56:435–447. doi:10.1093/jxb/eri060
Werner FA, Gradstein SR (2008) Seedling establishment of vascular epiphytes on isolated and enclosed forest trees in an Andean landscape, Ecuador. Biodivers Conserv 17:3195–3207. doi:10.1007/s10531-008-9421-5
Winkler M, Hulber K, Hietz P (2007) Population dynamics of epiphytic bromeliads: life strategies and the role of host branches. Basic Appl Ecol 8:183–196. doi:10.1016/j.baae.2006.05.003
Yamashita N, Ishida A, Kushima H, Tanaka N (2000) Acclimation to sudden increase in light favoring an invasive over native trees in subtropical islands, Japan. Oecologia 125:412–419. doi:10.1007/s004420000475
Yang SJ, Sun M, Zhang YJ, Cochard H, Cao KF (2014) Strong leaf morphological, anatomical, and physiological responses of a subtropical woody bamboo (Sinarundinaria nitida) to contrasting light environments. Plant Ecol 215:97–109. doi:10.1007/s11258-013-0281-z
Zotz G (1998) Demography of the epiphytic orchid, Dimerandra emarginata. J Trop Ecol 14:725–741. doi:10.1017/S0266467498000534
Zotz G (2013) The systematic distribution of vascular epiphytes-a critical update. Bot J Linn Soc 171:453–481. doi:10.1111/Boj.12010
Zotz G, Tyree MT (1996) Water stress in the epiphytic orchid, Dimerandra emarginata (G. Meyer) Hoehne. Oecologia 107:151–159. doi:10.1007/Bf00327898
Zotz G, Hietz P, Schmidt G (2001) Small plants, large plants: the importance of plant size for the physiological ecology of vascular epiphytes. J Exp Bot 52:2051–2056. doi:10.1093/jexbot/52.363.2051
Zotz G, Laube S, Schmidt G (2005) Long-term population dynamics of the epiphytic bromeliad, Werauhia sanguinolenta. Ecography 28:806–814. doi:10.1111/j.2005.0906-7590.04292.x
Zotz G, Bogusch W, Hietz P, Ketteler N (2010) Growth of epiphytic bromeliads in a changing world: the effects of CO2, water and nutrient supply. Acta Oecol 36:659–665. doi:10.1016/j.actao.2010.10.003
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (31670342, 31370362, 31170315), the Natural Science Foundation of Yunnan Province (2013FA044), and the National Key Project of the Ministry of Science and Technology of China (2015BAD10B03).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure S1
Determinate growth habit in family Orchidaceae. (a) Laelia anceps; (b) Flickingeria albopurpurea; (c) Bulbophyllum odoratissimum;(d) Cattleya sp.; (e) Coelogyne ovalis; (f) Epigeneium amplum. For a, b, and c, only one leaf is produced every year; for d, e, and f, two leaves expand simultaneously each year. Bar = 2 cm TIFF 3762 kb)
Rights and permissions
About this article
Cite this article
Zhang, W., Huang, W. & Zhang, SB. The study of a determinate growth orchid highlights the role of new leaf production in photosynthetic light acclimation. Plant Ecol 218, 997–1008 (2017). https://doi.org/10.1007/s11258-017-0747-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-017-0747-5