Abstract
Objectives
To investigate the relationship between the number of valvular insufficiency (VI) and emergency hospitalization or mortality in maintenance hemodialysis (HD) patients.
Methods
The maintenance HD patients with cardiac ultrasonography were included. According to the number of VI ≥ 2 or not, the patients were divided into two groups. The difference of emergency hospitalized for acute heart failure, arrhythmia, acute coronary syndrome (ACS) or stroke, cardiovascular mortality, and all-cause mortality between the two groups were compared.
Results
Among 217 maintenance HD patients, 81.57% had VI. 121 (55.76%) patients had two or more VI, and 96 (44.24%) with one VI or not. The study subjects were followed up for a median of 47 (3–107) months. At the end of the follow up, 95 patients died (43.78%), of whom 47 (21.66%) patients died because of cardiovascular disease. Age (HR 1.033, 95% CI 1.007–1.061, P = 0.013), number of VI ≥ 2 (HR 2.035, 95% CI 1.083–3.821, P = 0.027) and albumin (HR 0.935, 95% CI 0.881–0.992, P = 0.027) were independent risk factors for cardiovascular mortality. The three parameters were also independent risk factors for all-cause mortality. The patients with number of VI ≥ 2 were more likely to be emergency hospitalized for acute heart failure (56 [46.28%] vs 11 [11.46%], P = 0.001). On the contrary, the number of VI was not associated with emergency hospitalized for arrhythmia, ACS or stroke. Survival analysis results showed that probability of survival was statistically different in the two groups (P < 0.05), no matter based on cardiovascular mortality or all-cause mortality. Based on age, number of VI ≥ 2 and albumin, nomogram models for 5-year cardiovascular and all-cause mortality were built.
Conclusions
In maintenance HD patients, the prevalence of VI is prominently high. The number of VI ≥ 2 is associated with emergency hospitalized for acute heart failure, cardiovascular and all-cause mortality. Combining age, number of VI ≥ 2, and albumin can predict cardiovascular and all-cause mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic kidney disease (CKD) is a growing disease burden globally, which is predicted to become the fifth global cause of death by 2040 [1]. In 2017, there were 132·3 million [95% UI 121·8 to 143·7] cases of CKD in China, counting for 18.97% of global [2]. It means that China has become the country with the heaviest burden of CKD in the world. Among them, dialysis expenses account for the main CKD medical expenses. Hemodialysis (HD) is the most common form of renal replacement therapy in the world, accounting for approximately 89% of all dialysis and 69% of all renal replacement therapy [3]. According to the data from Chinese national renal data system (CNRDS), by December 31, 2020, there were 806,759 dialysis patients nationwide, including 692,736 HD patients, which accounting for 85.87%.
Cardiovascular disease (CVD) is one of the leading cause of death among maintenance HD patients [4], which is found in more than half of the cases [5]. Common cardiovascular diseases include coronary artery disease (CAD), arrhythmias, and sudden cardiac death (SCD). Except that, valvular heart disease (VHD) is also the common concomitant disease in dialysis [6]. Rheumatic heart disease and degenerative with age are the main cause of VHD [7]. Over the past 50 years, with the wide use of antibiotics and the development of medicine, the epidemiology of VHD has changed markedly in worldwide. Rheumatic heart disease has significantly decreased. On the contrary, as the population is aging, degenerative valve disease has been gradually increasing [8]. Except that, CKD is the independent risk factor of VHD [9].
Both rheumatic heart disease and degenerative changes may cause valvular insufficiency (VI). With the decline of renal function, the prevalence of VI is increasing, prominently in mitral valve [5]. Except that, valve calcification (VC) is common in CKD. It was reported that 80–99% CKD stage 5 patients had VC [10]. The prevalence of VC is eight times higher in maintenance HD patients than in the general population [11]. While several medical literature have reported that VC was associated with the mortality of maintenance HD patients [12, 13], less attention has been paid to VI. Now, in this study, we aimed to investigate the relationship between the number of VI and the mortality in maintenance HD patients.
Materials and methods
Patient selection
The new entry maintenance HD patients in the Blood Purification Department of Wuhan Fourth Hospital from April 1, 2012, to July 15, 2019, were selected. The inclusion criteria: (1) age ≥ 18 years; (2) cardiac ultrasonography was carried out within a week; (3) patients accepted standardized quality control management and regular hemodialysis until December 31, 2020 or died. The primary outcome measures were emergency hospitalized for acute heart failure, arrhythmia, stroke or new onset acute coronary syndrome (ACS), containing angina pectoris or acute myocardial infarction (AMI). The secondary outcome measures were cardiovascular or all-cause mortality. The exclusion criteria: (1) patients had a history of malignancy; (2) patients leave the dialysis center midway because of changing to peritoneal dialysis or kidney transplant; (3) patients lack of complete medical records, and (4) patients had a history of valve intervention or congenital heart disease.
Data collection
The basic data of patients were collected, including age, gender, using angiotensin converting enzyme inhibitors (ACEI) or angiotensin receptor blocker (ARB), the history of diabetes mellitus (DM), hypertension and CVD, containing acute myocardial infarction and angina. After obtaining the consent from the patient, blood specimens for biochemical tests were collected from the vascular access before dialysis. The baseline clinical data included hemoglobin, serum albumin (ALB), calcium, phosphorus, parathyroid hormone (PTH) and serum uric acid.
Cardiac ultrasonography is a specific and sensitive method for the detection of VI. All cardiac ultrasonography measurements were performed by two sonographers unaware of biochemical results. VI was defined according to guideline recommendations [14].
Statistics
Data analysis was performed with Statistical Package for Social Analysis (SPSS for Windows, IBM Corp, USA) version 20.0. The study population characteristics were presented as the mean ± standard deviation (SD), or percentage. According to the number of VI ≥ 2 or not, the studies were categorized into two groups. Two-sample t-test was used to compare the differences between the two groups for continuous variables. Differences for the nominal variables were compared by Chi-square test. To estimate survival probabilities, the Kaplan–Meier method was used and log-rank test for comparing the differences between the two groups. Cox regression analysis was used to explore the risk factors for new onset ACS, cardiovascular and all-cause mortality. Multivariate logistic regression models were used to test the associations of variables and number of VI ≥ 2. Statistical significance was defined as P < 0.05. GraphPad Prism 8 was used in graphic production. Nomogram models were performed with R version 4.1.3.
Results
Baseline data comparison
A total of 262 patients were selected, in them 15 cases with malignancy, 16 cases changed to peritoneal dialysis, 7 cases received a kidney transplant, 7 cases without complete medical records, were excluded. At last, 217 patients were enrolled in this study. 133 (61.29%) males and 84 (38.71%) females were contained. The mean age was 59.64 ± 14.52 years. There were 177 (81.57%) patients with VI, and 40 (18.43%) patients without. The distribution of VI in the patients are shown in Table 1. There were 121 (55.76%) patients with two or more VI. Based on the number of VI ≥ 2 or not, the studies were divided into two groups. The clinical characteristics of the study patients are shown in Table 1.
Emergency hospitalization and mortality
By December 31, 2020, the study subjects were followed up for a median of 47 (3–107) months. During the follow up, the patients of two groups emergency hospitalized for acute heart failure, arrhythmia, acute coronary syndrome (ACS) or stroke were shown in Table 2. In general, emergency hospital admission rate of VI ≥ 2 group was higher (90.91% vs 59.38%, P = 0.001). This difference was mainly due to heart failure hospitalization (Table 2).
At the end of the follow up, 95 patients died (43.78%); of whom 47 (21.66%) patients died because of CVD. In the number of VI ≥ 2 group, 59 (48.76%) patients died, including 32 (26.45%) cases because of cardiovascular events. In the number of VI < 2 group, 36 (37.50%) patients died, containing 15 (15.63%) CVD cases.
Risk factors of cardiovascular mortality, all-cause mortality and new onset ACS
To explore the independent risk factors of cardiovascular and all-cause mortality for the study patients, COX regression models were analyzed. The results showed that any single valve insufficiency was not related to cardiovascular or all-cause mortality. But age, number of VI ≥ 2 and ALB were all the independent risk factors of cardiovascular and all-cause mortality (Tables 3 and 4). Compared with number of VI < 2 group, number of VI ≥ 2 had higher cardiovascular mortality (HR 2.035, 95% CI 1.083–3.821, P = 0.027) and all-cause mortality (HR 1.711, 95% CI 1.102–2.656, P = 0.017).
To explore the relationship of VI and new onset ACS, COX regression models were analyzed. Unfortunately, none of VI or number of VI ≥ 2 was found to be correlated with new onset ACS. But we found that age (HR 1.023, 95% CI 1.004–1.043, P = 0.019), DM history (HR 1.686, 95% CI 1.040–2.734, P = 0.034) and CVD history (HR 2.271, 95% CI 1.395–3.697, P = 0.001) were independently associated with new onset ACS, as shown in Table 5.
To further find the influencing factors of VI ≥ 2, multivariate Logistic regression was used. The analysis results showed that after adjusting for multiple factors, only hemoglobin was related to VI ≥ 2 (OR 0.982, 95% CI 0.967–0.996, P = 0.015), as shown in Table 6.
Kaplan–Meier analyses
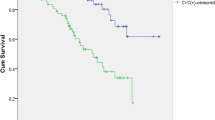
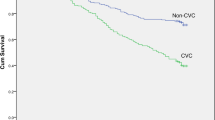
To compare the difference in survival between the two groups, Kaplan–Meier analyses were performed. Kaplan–Meier analysis of cardiovascular mortality results showed that number of VI ≥ 2 group had a significantly lower probability of survival than number of VI < 2 group (χ2 = 6.839, P = 0.009), as shown in Fig. 1. Figure 2 showed similar results in all-cause mortality (χ2 = 5.604, P = 0.018).
Nomogram models for 5-year cardiovascular and all-cause mortality
According to the results of multivariate COX regression analysis, we built the nomogram models to predict 5-year cardiovascular and all-cause mortality. Three variables from the independent risk factors were included: age, number of VI ≥ 2 and ALB. Based on the individual scores calculated using the nomogram, a total score was determined (Figs. 3, 4). The C-index of 5-year cardiovascular mortality model was 0.698 and the C-index for 5-year all-cause mortality model was 0.703. The calibration curves of two nomogram models are shown in Figs. 5, 6.
Discussion
Our study showed that the morbidity of VI was common in maintenance HD patients, accounting for 81.57%. And 64.98% patients had mitral insufficiency, which was the most common. Mitral insufficiency was also the first valve disease in the general population [15]. A retrospective analysis of 1326 dialysis patients found 42.8% cases had mitral regurgitation, in which 30.4% mild, 9.7% moderate, 2.6% severe [5]. In our study, the morbidity of tricuspid insufficiency was 53.46%, which was second. Tricuspid regurgitation was also a common valvular disease with high morbidity and mortality in general population [16, 17]. Unfortunately, no large-scale studies have reported the incidence of tricuspid regurgitation in dialysis patients. In general, VI can be classified as primary or secondary. Primary causes are structural or degenerative abnormalities at any part of the valve apparatus, containing leaflets, chordae tendineae, papillary muscles or annulus. Secondary causes are associated with heart geometrical alterations, usually from cardiac dysfunction without organic valvular diseases. Unfortunately, our study did not further explore the causes of VI in the research subjects.
In our study, 49.47% death cases were because CVD. Over the past decade, with the development of medical technology and medical insurance, the quality of life and survival rate of maintenance HD patients have been significantly improved. Ultra-pure dialysis water, dialyzers with high biocompatibility and clearance rate, individualized dialysis programs, focusing on nutrition and complication control, all make important contributions. Despite this, CVD remains the most common cause of mortality in maintenance HD patients. Evidence showed that the risk of premature death due to CVD in maintenance HD patients was 20 times higher than in the general population [18].
Samad et al.’s research showed that 5-year survival in patients decreased as mitral regurgitation severity increasing [5]. Green et al. reported mitral regurgitation was the only parameters predictive of sudden cardiac death [19]. Based on the data analysis of 533 patients admitted for first systolic heart failure, Hsiao SH et al. also discovered that systolic pulmonary regurgitation was associated with cardiovascular death [20]. Nevertheless, we found that insufficiency of any one valve was not the independent risk factor for cardiovascular mortality or all-cause mortality, but number of VI ≥ 2 was correlated to both. It means in our future work, we need to pay more attention to the number of valvular lesions in dialysis patients. In addition, we also found that age and albumin were independent risk factors for cardiovascular and all-cause death in maintenance HD patients. A meta-analysis for patients undergoing hemodialysis also found that age and lower albumin were the risk factors of cardiac death [21]. Based on our results, we build the nomogram models to predict 5-year cardiovascular and all-cause mortality.
Some researches revealed DM and previous CVD were also correlated to cardiovascular and all-cause mortality [21, 22]. But in our results, neither history of DM nor CVD was associated with cardiovascular or all-cause mortality. Banshodani et al. analyzed 260 dialysis patients and also discovered that DM was not an independent risk factor for cardiovascular or all-cause mortality [23]. This difference may be related to the sample size.
In addition, no research has reported the relationship between the number of VI and acute heart failure. For the first time, we found that maintenance HD patients with number of VI ≥ 2 were more likely to be emergency hospitalized for acute heart failure. On the contrary, the number of VI was not associated with emergency hospitalized for arrhythmia, ACS or stroke. Mitral regurgitation can leads to heart failure by inducing volume overload of the heart [24]. Systolic pulmonary regurgitation was also considered to correlate with heart failure rehospitalization [20]. But a study of 639 hospitalized patients with acute heart failure found that moderate or severe tricuspid regurgitation was not associated with readmission for heart failure [25].
It is well known that ischemic mitral regurgitation is frequently associated with AMI [26]. On the contrary, whether patients with VI are more likely to develop ACS has not been reported. In our study, we found neither any VI nor number of VI ≥ 2 was the independent risk of new onset ACS in maintenance HD patients.
Anemia is a common complication in maintenance HD patients. Our study found that hemoglobin was associated with more than two valvular lesions, but the specific mechanism needed further study. Tigen et al.’ study proved anemia (hemoglobin levels less than 12.5 mg/dL) was an independent predictor of moderate or severe functional mitral regurgitation in non-ischemic dilated cardiomyopathy patients with normal renal function [27].
There are several limitations in our study: (1) This is a retrospective study with a single-center. The sample size is small, which may lead to selective bias. (2) Some covariates that might affect cardiovascular and all-cause mortality in maintenance HD patients have not been included, such as spKt/V homocysteine dialysis vintage. (3) Severity of valvular insufficiency has not been subdivided, which may have more value for cardiovascular and all-cause mortality.
In conclusion, the prevalence of VI in maintenance HD patients is very high. The number of VI ≥ 2 is associated with emergency hospitalized for acute heart failure, cardiovascular, and all-cause mortality in maintenance HD patients. In addition, combining age, number of VI ≥ 2 and albumin can predict 5-year cardiovascular and all-cause mortality in maintenance HD patients.
Data availability
All data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Foreman KJ, Marquez N, Dolgert A et al (2018) Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: reference and alternative scenarios for 2016–40 for 195 countries and territories. Lancet 392:2052–2090
GBD Chronic Kidney Disease Collaboration (2020) Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the global burden of disease study 2017. Lancet 395(10225):709–733
Bello AK, Okpechi IG, Osman MA et al (2022) Epidemiology of haemodialysis outcomes. Nat Rev Nephrol 18(6):378–395
McCullough PA, Chan CT, Weinhandl ED, Burkart JM et al (2016) Intensive hemodialysis, left ventricular hypertrophy, and cardiovascular disease. Am J Kidney Dis 68(51):S5–S14
Samad Z, Sivak JA, Phelan M et al (2017) Prevalence and outcomes of left-sided valvular heart disease associated with chronic kidney disease. J Am Heart Assoc 6(10):e006044
Hoevelmann J, Mahfoud F, Lauder L et al (2021) Valvular heart disease in patients with chronic kidney disease. Herz 46(3):228–233
Urena-Torres P, D’Marco L, Raggi P et al (2020) Valvular heart disease and calcification in CKD: more common than appreciated. Nephrol Dial Transpl 35(12):2046–2053
Chlabicz M, Chlabicz S, Marcinowicz L et al (2014) Outpatient use of systemic antibiotics in Poland: 2004–2008. Przegl Epidemiol 68(3):435–547
Kuźma Ł, Małyszko J, Bachórzewska-Gajewska H et al (2020) Impact of chronic kidney disease on long-term outcome of patients with valvular heart defects. Int Urol Nephrol 52(11):2161–2170
Rong S, Qiu X, Jin X et al (2018) Risk factors for heart valve calcification in chronic kidney disease. Medicine (Baltimore) 97(5):9804
Moshar S, Bayesh S, Mohsenikia M et al (2016) The association of calcium-phosphorus product with the severity of cardiac valves failure in patients under chronic hemodialysis. Cardiol Res 7(2):80–83
Chen XN, Chen ZJ, Ma XB et al (2015) Aortic artery and cardiac valve calcification are associated with mortality in Chinese hemodialysis patients: a 35 years follow-up. Chin Med J 128(20):2764–2771
Li M, Ye ZC, Li CM et al (2020) The influence of cardiac valvular calcification on all-cause and cardiovascular mortality in maintenance hemodialysis patients. Int Urol Nephrol 52(5):943–951
Zoghbi WA, Adams D, Bonow RO et al (2017) Recommendations for noninvasive evaluation of native valvular regurgitation: a report from the American society of echocardiography developed in collaboration with the society for cardiovascular magnetic resonance. J Am Soc Echocardiogr 30(4):303–371
Nkomo VT, Gardin JM, Skelton TN et al (2006) Burden of valvular heart diseases: a population based study. Lancet 368(9540):1005–1011
Topilsky Y, Maltais S, Medina Inojosa J et al (2019) Burden of tricuspid regurgitation in patients diagnosed in the community setting. J Am Coll Cardiol Img 12(3):433–442
Wang N, Fulcher J, Abeysuriya N et al (2019) Tricuspid regurgitation is associated with increased mortality independent of pulmonary pressures and right heart failure: a systematic review and meta-analysis. Eur Heart J 40(5):476–484
Cozzolino M, Mangano M, Stucchi A et al (2018) Cardiovascular disease in dialysis patients. Nephrol Dial Transpl 33(suppl_3):iii28–iii34
Green D, Ritchie JP, Abidin N et al (2014) The association of ECG and echocardiographic abnormalities with sudden cardiac death in a dialysis patient cohort. J Nephrol 27(1):81–86
Hsiao SH, Hsiao CS, Chiou KR (2021) Systolic pulmonary regurgitation affects the outcome of patients with severe systolic heart failure. ESC Heart Fail 8(6):5121–5131
Ma L, Zhao S (2017) Risk factors for mortality in patients undergoing hemodialysis: a systematic review and meta-analysis. Int J Cardiol 238:151–158
Ku E, McCulloch CE, Ahearn P et al (2020) Trends in cardiovascular mortality among a cohort of children and young adults starting dialysis in 1995 to 2015. JAMA Netw Open 3(9):e2016197
Banshodani M, Kawanishi H, Moriishi M et al (2020) Association between dialysis modality and cardiovascular diseases: a comparison between peritoneal dialysis and hemodialysis. Blood Purif 49(3):302–309
Watanabe S, Bikou O, Hajjar RJ et al (2018) Swine model of mitral regurgitation induced heart failure. Methods Mol Biol 1816:327–335
Mutlak D, Lessick J, Khalil S et al (2018) Tricuspid regurgitation in acute heart failure: is there any incremental risk? Eur Heart J Cardiovasc Imaging 19(9):993–1001
Levine RA, Hung J (2003) Ischemic mitral regurgitation, the dynamic lesion: clues to the cure. J Am Coll Cardiol 42(11):1929–1932
Tigen K, Karaahmet T, Kirma C et al (2010) The association of functional mitral regurgitation and anemia in patients with non-ischemic dilated cardiomyopathy. Cardiol J 17(3):274–280
Acknowledgements
This study was funded by the Wuhan Municipal Health Commission with no. WX18Q31 and WX20C04.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Ethical approval
This study was approved by the Ethics Committee of Wuhan Fourth Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wei, H., Liu, S., Tian, M. et al. The number of valvular insufficiency is a strong predictor of cardiovascular and all-cause mortality in hemodialysis patients. Int Urol Nephrol 55, 2915–2924 (2023). https://doi.org/10.1007/s11255-023-03576-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-023-03576-3