Abstract
Purpose
To evaluate the effect of potassium citrate administration on the composition of encrusted material on the ureteral stent after Double-J insertion.
Methods
We designed a randomized clinical trial for our study; 65 patients that underwent transurethral lithotripsy and Double-J stent insertion were included in the study after informed consent and divided into two groups. In the first group (33 patients) potassium citrate was prescribed after surgery till stent removal and the second group (32 patients) followed without prescribing this medication. After stent removal, encrusted materials on removed stents were analyzed then the type and composition of encrusted material compared with the primary stone that was removed.
Results
Our results revealed that the type and composition of primary stone and encrusted stone were similar in patients that do not receive potassium citrate (p-value of 0.073, 0.251 and 0.944 for calcium oxalate, uric acid, and calcium phosphate respectively). In patients that taking potassium citrate rate of calcium oxalate (p-value < 0.001) and uric acid (p-value < 0.001) material on encrusted stent significantly decreased compared with the non-intervention group.
Conclusion
Results of this study revealed that taking of potassium citrate after ureteral stent insertion significantly decreases the formation of calcium oxalate and uric acid encrusted material on Double-J stent so it could be recommended for prevention of stent encrustation in patients that primary stone analysis are calcium oxalate and uric acid stone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With the advance in endourologic procedure need to stent placement is increasing. The consensus for ureteral stent insertion is compromised renal function, mucosal ureteral wall injuries or perforation, and significant residual stone. Placement of ureteral stent is not free of morbidity, and is associated with Irritative urinary symptoms, urinary tract infection, stent encrustation, and sometimes rare situations like stent migration, fragmentation, and retained/forgotten stent [1,2,3].
Among these morbidities, encrustation is sometimes problematic as could prevent easy removal of the stent [4,5,6].The most important factor related to stent encrustation is the duration of stent placement as this time increase by more than 3 months, the rate of encrustation will have increased [7,8,9,10,11]. The other risk factors are urinary tract infections and pregnancy [12]. Using of potassium citrate in urolithiasis is well-established especially in hypocitraturic patients and recurrent calcium stone formers. The potential mechanisms are inhibition of the nucleation, aggregation of calcium and oxalate crystals, and alkalinization of urine [13]. Also, the effect of potassium citrate on the dissolution of uric acid confirmed as strongly recommended in these stones [14,15,16]. With these well-known effects of potassium citrate, we designed a study if it could change the type and composition of encrusted material on the stent in patients that underwent transurethral lithotripsy and Double-J stent insertion.
Materials and methods
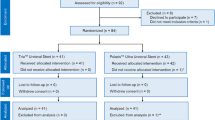
In a randomized clinical trial study from January 2020 to April 2020, seventy patients that undergone transurethral lithotripsy and Double-J stent insertion for ureteral stone were enrolled in our study after obtaining written informed consent. This study was approved by the ethics committee of Tehran University of Medical Sciences (IRCT20190624043991N6). The exclusion criteria were patients that taking drugs such as digoxin and potassium sparing-diuretics, pregnancy, chronic kidney disease, and history of hyperkalemia. Patients were divided randomly to separate groups; the first group (35 patients) received the extended-release oral tablet of potassium citrate; 10 meq three times a day till stent removal, and the second group (35 patients) do not receive this medication. The same dietary recommendation was given to the control and intervention groups in written instruction. To kept the urine pH of the intervention groups between 6.8 and 7.2, we measured their urine pH every 2 weeks. The dosage of potassium citrate increased to 40 meq per day if urine pH was lower than 6.8 and if it was higher than 7.2, we decreased the dosage of potassium citrate to 20 meq per day. Also, to prevent hyperkalemia, we controlled the electrolytes of the patients of the intervention group every two weeks. If patients had severe urinary symptoms or fever that suggestive of urinary tract infection, a urine sample was sent for culture. We used Double-J 4.8 Fr stent for all patients (Gohar Shafa, Polyurethane type, 4.8 Fr Double-J stent). Our indications for stent insertion were significant bolus edema, ureteral stricture, ureteral perforation, and significant hydronephrosis. After removal of the Double-J stent, we evaluated the proximal tail of double-j stent (4–5 cm) for any difference of stent encrustation in two groups (due to more problematic stent removal if encrustation formed in the proximal tail), also we had analyzed stents in our laboratory for the difference in type and composition of encrusted material on stent from primary stone in two groups. The primary stone analysis was performed in our laboratory by an expert biologist with a qualitative infrared spectroscopy method. After stent removal, the encrusted material on the stent was removed mechanically and analyzed in the same way as primary stone analysis by the infrared spectroscopy method. Demographic data, location of ureteral stones, type of primary stone, the composition of encrusted materials, stent-related symptoms evaluated in two intervention and control groups. All statistical analysis was performed by independent t-test, Chi-Square test, Fisher-exact test, paired t-test, and by using SPSS 22.0 software for windows. A flowchart of our study is depicted in “Appendix”.
Results
At the end of the study, three patients of the control group and two patients of the intervention group lost the follow-up so excluded from the study (“Appendix”). The control group (32 patients) and intervention group (33 patients) matched for the location of ureteral stone and duration of the indwelling double-j stent. There were no significant differences between the 2 groups in terms of demographic characteristics such as age, sex, and body mass index (Table 1). The hospital stay time and duration of stent-related symptoms were not significantly different between the two groups. There was no significant difference between groups regard to primary stone composition. As mentioned earlier our indications for stent insertion were significant bolus edema, ureteral stricture, ureteral perforation, and significant hydronephrosis; in these conditions, we routinely remove DJ stent after 4–6 weeks but due to the beginning of COVID-19 pandemic and fear of hospital admission many of them had several days delay in returning for DJ removal. There were not any significant differences between the two groups regarding the indications for stent placement.
In patients that had not received potassium citrate, type of primary stone and encrusted material on stent was not significantly different (p-value of 0.073, 0.251, and 0.944 for calcium oxalate, uric acid, and calcium phosphate respectively) but in patients taking potassium citrate by comparing the pre-stenting primary stone composition and post-stenting encrustation materials; results revealed a decrease in calcium oxalate and uric acid encrustation on stent surface (p-value < 0.001). An image of the effect of potassium citrate on decreasing encrustation represents in Fig. 1. The results are mentioned in detail in Table 2.
The mean duration of double-j stent placement in the control group and intervention group was 50.59 ± 17.01 and 50.03 ± 15.07 days respectively (due to the beginning of COVID-19 pandemic and fear of hospital admission many of them had several days delay in returning for DJ removal), in this period stent-related complication including hematuria, Irritative symptoms, urinary tract infection, and rare complications such as stent migration and fracture compared between two groups (Table 3). There were no significant differences between mentioned items in the two groups especially the rate of urinary tract infection. We didn’t have Clavien-Dindo complication of grade 3or 4, and most complications were categorized as grade 2 complications.
Discussion
With increasing the routine use of ureteral stent in the endourologic procedure, stent-related complications will be more common. One of the complex problems of Double-J stent insertion is stent encrustation [17]. Stent encrustation could result in difficulty in stent removal and occasionally due to obstruction of ureteral lumen result in renal failure in case of forgotten stents [18]. The most significant factor related to stent encrustation is the time of stent placement [11, 19, 20]. In an interesting study by Sighinolfi et al., they analyzed variables that related to stent encrustation and found encrustation in the proximal tail of double-j stent-related to the metabolic factors such as hypercalciuria and calcium oxalate was the most common composition on proximal tail whereas encrustation at the distal tail related to the urinary tract infection and age of patients; maybe aging could predispose to urinary tract infection [21]. In the study by Kawahara et al., they mentioned that stent encrustation is not related to the duration of stent placement, but interestingly caliber of stent-related to stent encrustation as larger caliber stent (> 7 Fr) demonstrated a lower rate of encrustation compared with smaller stents (< 6 Fr) [22]. The stent encrustation is a complex phenomenon and several theories are suggested in this regard; as stent encrustation increased by duration of stent placement, the role of urinary tract infection could not be ignored, especially urease-producing bacteria can predispose to stone formation by increasing urinary pH (especially in urine pH more than 7.2). Also, bacteria could have formed a biofilm that accelerates the aggregation of crystals on biofilm [23,24,25,26]. In recent years many inventions in stent material have been made to reduce the chance of stent encrustation such as using silicone stents instead of conventional polyurethane stents; in the study by Bouzidi et al. rate of stent encrustation and biofilm formation were lower in silicone stents compared with polyurethane stents [27]. The other novelty in ureteral stent design to reduce stent encrustation were drug-eluting stents(antibiotic-eluting) and heparin-coated stents (anti-adhesive effect) that both of which had incongruous results [28,29,30].
In the study by Scarneciu et al., they evaluated 138 removed ureteral stents for the composition of encrustation material on the Double-J stent in a different part of the stent and found the most common composition is calcium oxalate; they concluded that the mechanism of stent encrustation and urolithiasis is similar [31]. Ilie et al. [32]cited encrustation mainly occurred in renal and bladder end of the stent and ureteric segment of the stent is less prone to encrustation due to peristaltic movement of the ureter, but on the contrary study by Baltitude et al. [33]mentioned only proximal tail of double-j stent predispose to encrustation because peristalsis has an inhibitory effect on encrustation on the body and distal end of the double-j stent with the washing of debris. Several studies confirmed that encrustation in the proximal tail of double-j stent is more problematic for stent removal and predict the need for multiple sessions of surgery for stent removal [19, 34]. The role of potassium citrate as an inhibitor of calcium oxalate stones is well known for the inhibition of nucleation and aggregation of crystals. Also since the most frequent composition of encrusted material is calcium oxalate it is the rationale that encrustation and stone formation have the same pathophysiology. A study by Pak et al. evaluated 1392 patients with urolithiasis and mentioned hypocitraturia as the most common metabolic finding in their report (44.3%) [35]. In a retrospective study by Idweini they compared two group of patients that underwent Shock Wave Lithotripsy(SWL) and double-j stent insertion;82 patients underwent SWL and double-j stent insertion without potassium citrate administration and 132 patients underwent SWL and stent insertion and taking potassium citrate due to metabolic reasons, stent encrustation reduced significantly in patients used potassium citrate (7 vs 33% p-value < 0.05) [36]. Concerning these known effects of potassium citrate and prevalence of hypocitraturia in stone formers, we have conducted a randomized trial study to evaluate its impact on changing composition of encrusted material on stent especially proximal tail because removal of double-j with proximal tail encrustation is more difficult than encrustation in body and distal tail of stent. As far as we know, this study is the first clinical trial that evaluated the effect of potassium citrate on the composition of encrusted material on double-j stents. The results revealed that in patients who had a Double-J stent and primary stone analysis is calcium oxalate and uric acid, type and composition of encrusted material on stent significantly change by taking of potassium citrate compared to control group that does not receive this medication, so this may result in easier stent removal in this group. We don’t find serious complications with this medication so it could be recommended as a low-cost and feasible way to reduce stent encrustation in daily practice. One of the concerns about potassium citrate administration is that if alkalinization of urine exceeds more than 7.2, the risk of stone formation increased so we measured their urine pH every two weeks to adjust the dose of potassium citrate. Our study had some shortcomings and limitations; the first one is that some patients lost the follow-up, the second one is that some patients had a delay in their DJ removal due to fear of hospital admission during the COVID-19 pandemic but this factor matched in the two groups in the final analysis. The third one is that our study was a single-center study with a limited number of patients so we recommend a similar study by multicenter collaboration with evaluating a large number of patients. Another study that compares recurrent and first stone former for evaluating the impact of potassium citrate on stent encrustation in patients with a metabolic change in 24-h urine analysis could be interesting. This study revealed that in patients who underwent transurethral lithotripsy and double-j stent insertion and primary stone analysis is calcium oxalate and uric acid, potassium citrate administration has a preventive effect on stent encrustation, but this effect is not evident in patients with other types of stones (calcium phosphate and struvite stones).
Conclusion
Results of this study revealed that taking of potassium citrate after ureteral stent insertion significantly decreases the formation of calcium oxalate and uric acid encrusted material on Double-J stent so it could be recommended for prevention of stent encrustation in patients that primary stone analysis are calcium oxalate and uric acid stone.
Abbreviations
- BMI:
-
Body mass index
- UVJ:
-
Ureterovesical junction
- Ca. ox:
-
Calcium oxalate
- Ca.H.po4:
-
Calcium hydrogen phosphate
- Fr:
-
French
- DJ:
-
Double-J
- SWL:
-
Shock wave lithotripsy
- COVID-19:
-
Coronavirus disease 2019
References
Joshi HB, Stainthorpe A, MacDonagh RP, Keeley FX Jr, Timoney AG, Barry MJ (2003) Indwelling ureteral stents: evaluation of symptoms, quality of life and utility. J Urol 169(3):1065–1069. https://doi.org/10.1097/01.ju.0000048980.33855.90 (discussion 9)
Damiano R, Oliva A, Esposito C, De Sio M, Autorino R, D’Armiento M (2002) Early and late complications of double pigtail ureteral stent. Urol Int 69(2):136–140. https://doi.org/10.1159/000065563
Memon NA, Talpur AA, Memon JM (2007) Indications and complications of indwelling ureteral stenting at NMCH, Nawabshah. Pak J Surg 23(3):187–191
Dyer RB, Chen MY, Zagoria RJ, Regan JD, Hood CG, Kavanagh PV (2002) Complications of ureteral stent placement. Radiographics 22(5):1005–1022. https://doi.org/10.1148/radiographics.22.5.g02se081005
Borboroglu PG, Kane CJ (2000) Current management of severely encrusted ureteral stents with a large associated stone burden. J Urol 164(3 Pt 1):648–650. https://doi.org/10.1097/00005392-200009010-00007
Singh I, Gupta NP, Hemal AK, Aron M, Seth A, Dogra PN (2001) Severely encrusted polyurethane ureteral stents: management and analysis of potential risk factors. Urology 58(4):526–531. https://doi.org/10.1016/s0090-4295(01)01317-6
El-Faqih SR, Shamsuddin AB, Chakrabarti A, Atassi R, Kardar AH, Osman MK et al (1991) Polyurethane internal ureteral stents in treatment of stone patients: morbidity related to indwelling times. J Urol 146(6):1487–1491. https://doi.org/10.1016/s0022-5347(17)38146-6
Bibby LM, Wiseman OJ (2021) Double JJ ureteral stenting: encrustation and tolerability. Eur Urol Focus 7(1):7–8. https://doi.org/10.1016/j.euf.2020.08.014
Cauda V, Chiodoni A, Laurenti M, Canavese G, Tommasi T (2017) Ureteral double-J stents performances toward encrustation after long-term indwelling in a dynamic in vitro model. J Biomed Mater Res Part B Appl Biomater 105(8):2244–2253. https://doi.org/10.1002/jbm.b.33756
Kadihasanoglu M, Kilciler M, Atahan O (2017) Luminal obstruction of double j stents due to encrustation depends on indwelling time: a pilot study. Aktuelle Urol 48(3):248–251. https://doi.org/10.1055/s-0042-118050
Polat H, Yucel MO, Utangac MM, Benlioglu C, Gok A, Cift A et al (2017) Management of forgotten ureteral stents: relationship between indwelling time and required treatment approaches. Balk Med J 34(4):301–307. https://doi.org/10.4274/balkanmedj.2015.1562
Canales BK, Higgins L, Markowski T, Anderson L, Li QA, Monga M (2009) Presence of five conditioning film proteins are highly associated with early stent encrustation. J Endourol 23(9):1437–1442. https://doi.org/10.1089/end.2009.0389
Pak CY, Fuller C, Sakhaee K, Preminger GM, Britton F (1985) Long-term treatment of calcium nephrolithiasis with potassium citrate. J Urol 134(1):11–19. https://doi.org/10.1016/s0022-5347(17)46962-x
Trinchieri A, Esposito N, Castelnuovo C (2009) Dissolution of radiolucent renal stones by oral alkalinization with potassium citrate/potassium bicarbonate. Arch Ital Urol Androl 81(3):188–191. http://www.ncbi.nlm.nih.gov/pubmed/19911683
Honda M, Yamamoto K, Momohara C, Komori K, Takada T, Fujioka H (2003) Oral chemolysis of uric acid stones. Hinyokika kiyo Acta urologica Japonica. 49(6):307–10. http://www.ncbi.nlm.nih.gov/pubmed/12894725
Heilberg IP (2016) Treatment of patients with uric acid stones. Urolithiasis 44(1):57–63. https://doi.org/10.1007/s00240-015-0843-8
Ahallal Y, Khallouk A, El Fassi MJ, Farih MH (2010) Risk factor analysis and management of ureteral double-j stent complications. Rev Urol 12(2–3):e147–51. http://www.ncbi.nlm.nih.gov/pubmed/20811552
Ulker V, Celik O (2019) Endoscopic, single-session management of encrusted, forgotten ureteral stents. Medicina (Kaunas) 55(3):58. https://doi.org/10.3390/medicina55030058
Kartal IG, Baylan B, Gok A, Sagnak AL, Karakoyunlu N, Cakici MC et al (2018) The association of encrustation and ureteral stent indwelling time in urolithiasis and KUB grading system. Urol J 15(6):323–328. https://doi.org/10.22037/uj.v0i0.4592
Legrand F, Saussez T, Ruffion A, Celia A, Djouhri F, Musi G et al (2021) Double loop ureteral stent encrustation according to indwelling time: results of a European multicentric study. J Endourol 35(1):84–90. https://doi.org/10.1089/end.2020.0254
Sighinolfi MC, Sighinolfi GP, Galli E, Micali S, Ferrari N, Mofferdin A et al (2015) Chemical and mineralogical analysis of ureteral stent encrustation and associated risk factors. Urology 86(4):703–706. https://doi.org/10.1016/j.urology.2015.05.015
Kawahara T, Ito H, Terao H, Yoshida M, Matsuzaki J (2012) Ureteral stent encrustation, incrustation, and coloring: morbidity related to indwelling times. J Endourol 26(2):178–182. https://doi.org/10.1089/end.2011.0385
Gleeson MJ, Glueck JA, Feldman L, Griffith DP, Noon GP (1989) Comparative in vitro encrustation studies of biomaterials in human urine. ASAIO Trans 35(3):495–498. https://doi.org/10.1097/00002480-198907000-00104
Mosayyebi A, Manes C, Carugo D, Somani BK (2018) Advances in ureteral stent design and materials. Curr Urol Rep 19(5):35. https://doi.org/10.1007/s11934-018-0779-y
Scotland KB, Lo J, Grgic T, Lange D (2019) Ureteral stent-associated infection and sepsis: pathogenesis and prevention: a review. Biofouling 35(1):117–127. https://doi.org/10.1080/08927014.2018.1562549
Tsukanov AY, Akhmetov DS, Blesman AI, Rogachev EA (2018) The impact of ureteral stent surface on encrustation and biofilm formation. Urologiia (Moscow, Russia: 1999). http://www.ncbi.nlm.nih.gov/pubmed/29901293(2):40-5
Bouzidi H, Traxer O, Doré B, Amiel J, Hadjadj H, Conort P et al (2008) Caractéristiques des incrustations des endoprothèses urétérales chez les patients lithiasiques. Prog Urol 18(4):230–237
De Grazia A, Somani BK, Soria F, Carugo D, Mosayyebi A (2019) Latest advancements in ureteral stent technology. Transl Androl Urol 8(Suppl 4):S436–S441. https://doi.org/10.21037/tau.2019.08.16
Forbes C, Scotland KB, Lange D, Chew BH (2019) Innovations in ureteral stent technology. Urol Clinics N Am 46(2):245–255. https://doi.org/10.1016/j.ucl.2018.12.013
Mosayyebi A, Vijayakumar A, Yue QY, Bres-Niewada E, Manes C, Carugo D et al (2017) Engineering solutions to ureteral stents: material, coating and design. Central Eur J Urol 70(3):270–274. https://doi.org/10.5173/ceju.2017.1520
Scarneciu I, Bratu OG, Cobelschi CP, Neculoiu CD, Scarneciu CC, Lupu S et al (2018) The risk factors and chemical composition of encrustation of ureteral double J stents in patients with urolithiasis. Rev Chim (Bucharest) 69(12):3406–3409
Ilie VG, Ilie VI (2017) Ureteric stent use-part of the solution and part of the problem. Curr Urol 11(3):126–130
Bultitude MF, Tiptaft RC, Glass JM, Dasgupta P (2003) Management of encrusted ureteral stents impacted in upper tract. Urology 62(4):622–626
Weedin JW, Coburn M, Link RE (2011) The impact of proximal stone burden on the management of encrusted and retained ureteral stents. J Urol 185(2):542–547. https://doi.org/10.1016/j.juro.2010.09.085
Pak CY, Poindexter JR, Adams-Huet B, Pearle MS (2003) Predictive value of kidney stone composition in the detection of metabolic abnormalities. Am J Med 115(1):26–32. https://doi.org/10.1016/s0002-9343(03)00201-8
Idweini Y (2016) Alkaline citrate medication to prevent stent encrustation in urolithiasis therapy. J Urol Nephrol 2:1
Acknowledgements
We would like to acknowledge Nooria Darman Pasargad for providing the Alitoral tablets (Alithoral 10).
Funding
This study did not receive any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Nothing to declare.
Ethical approval
This study was approved by the ethics committee of Tehran University of Medical Sciences (IRCT20190624043991N6).
Informed consent
Informed consent was obtained from all patients included in our study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix

Rights and permissions
About this article
Cite this article
Mohammadi, A., Rakebi, M.M., gholamnezhad, M. et al. Does potassium citrate administration change the type and composition of encrusted material on Double-J stent compared to primary stone?. Int Urol Nephrol 53, 1797–1803 (2021). https://doi.org/10.1007/s11255-021-02891-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-021-02891-x