Abstract
Purpose
Dynamic elasticity is a biomechanical property of the bladder in which muscle compliance can be acutely adjusted through passive stretches and reversed with active contractions. The aim of this study was to determine if manipulating dynamic elasticity using external compression could be used as a novel method to acutely increase bladder capacity and reduce bladder pressure in a porcine model.
Methods
Ex vivo experiment: bladders underwent continuous or pulsatile compression after establishing a reference pressure at bladder capacity. Bladders were then filled back to the reference pressure to determine if capacity could be acutely increased. In-vivo experiments: bladders underwent five cycles of pulsatile external compression with ultrasound confirmation. Pre and post-compression pressures were measured, and pressure was measured again 10 min post-compression.
Results
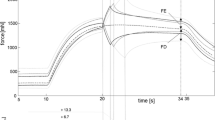
Ex vivo experiment: pulsatile compression demonstrated increased bladder capacity by 16% (p = 0.01). Continuous compression demonstrated increased capacity by 9% (p < 0.03). Comparison of pulsatile to continuous compression showed that the pulsatile method was superior (p = 0.03). In-vivo experiments: pulsatile external compression reduced bladder pressure by 19% (p < 0.00001) with a return to baseline 10 min post-compression.
Conclusions
These results suggest that regulation of bladder dynamic elasticity achieved with external compression can acutely decrease bladder pressure and increase bladder capacity. Pulsatile compression was found to be more effective as compared to continuous compression. These results highlight the clinical potential for use of non-invasive pulsatile compression as a therapeutic technique to increase bladder capacity, decrease bladder pressure, and reduce the symptoms of urinary urgency.



Similar content being viewed by others
Availability of data and material
Raw data is available by request.
References
Metcalfe PD et al (2010) Bladder outlet obstruction: progression from inflammation to fibrosis. BJU Int 106(11):1686–1694. https://doi.org/10.1111/j.1464-410X.2010.09445.x
Landau EH et al (1994) Loss of elasticity in dysfunctional bladders: urodynamic and histochemical correlation. J Urol 152(2):702–705. https://doi.org/10.1016/s0022-5347(17)32685-x
Speich JE, Borgsmiller L, Call C, Mohr R, Ratz PH (2005) ROK-induced cross-link formation stiffens passive muscle: reversible strain-induced stress softening in rabbit detrusor. Am J Physiol Physiol 289(1):C12–C21. https://doi.org/10.1152/ajpcell.00418.2004
Ratz PH, Speich JE (2010) Evidence that actomyosin cross bridges contribute to ‘passive’ tension in detrusor smooth muscle. Am J Physiol Physiol 298(6):F1424–F1435. https://doi.org/10.1152/ajprenal.00635.2009
Colhoun AF et al (2017) A pilot study to measure dynamic elasticity of the bladder during urodynamics. Neurourol Urodyn 36(4):1086–1090. https://doi.org/10.1002/nau.23043
Cullingsworth ZE et al (2020) Comparative-fill urodynamics in individuals with and without detrusor overactivity supports a conceptual model for dynamic elasticity regulation. Neurourol Urodyn 39(2):707–714. https://doi.org/10.1002/nau.24255
Jiang H, Liao D, Zhao J, Wang G, Gregersen H (2014) Contractions reverse stress softening in rat esophagus. Ann Biomed Eng 42(8):1717–1728. https://doi.org/10.1007/s10439-014-1015-7
Balthazar A et al (2019) An external compress-release protocol induces dynamic elasticity in the porcine bladder: A novel technique for the treatment of overactive bladder? Neurourol Urodyn 38(5):1222–1228. https://doi.org/10.1002/nau.23992
Colhoun AF et al (2016) Acute length adaptation and adjustable preload in the human detrusor. Neurourol Urodyn 35(7):792–797. https://doi.org/10.1002/nau.22820
Almasri AM, Ratz PH, Bhatia H, Klausner AP, Speich JE (2010) Rhythmic contraction generates adjustable passive stiffness in rabbit detrusor. J Appl Physiol 108(3):544–553. https://doi.org/10.1152/japplphysiol.01079.2009
Speich JE, Southern JB, Henderson S, Wilson CW, Klausner AP, Ratz PH (2012) Adjustable passive stiffness in mouse bladder: regulated by Rho kinase and elevated following partial bladder outlet obstruction. Am J Physiol Physiol 302(8):F967–F976. https://doi.org/10.1152/ajprenal.00177.2011
Anele UA et al (2018) Potential vascular mechanisms in an ex vivo functional pig bladder model. Neurourol Urodyn 37(8):2425–2433. https://doi.org/10.1002/nau.23710
Vince R et al (2018) Effects of vesical and perfusion pressure on perfusate flow, and flow on vesical pressure, in the isolated perfused working pig bladder reveal a potential mechanism for the regulation of detrusor compliance. Neurourol Urodyn 37(2):642–649. https://doi.org/10.1002/nau.23362
Wickramaratne N et al (2019) Acute resuscitation with polyethylene glycol-20k: a thromboelastographic analysis. J Trauma Acute Care Surg 87(2):322–330. https://doi.org/10.1097/TA.0000000000002332
Kanai A, Andersson K-E (2010) Bladder afferent signaling: recent findings. J Urol 183(4):1288–1295. https://doi.org/10.1016/j.juro.2009.12.060
Habteyes FG et al (2017) Modeling the influence of acute changes in bladder elasticity on pressure and wall tension during filling. J Mech Behav Biomed Mater 71:192–200. https://doi.org/10.1016/j.jmbbm.2017.02.020
Lentle RG, Reynolds GW, Janssen PWM, Hulls CM, King QM, Chambers JP (2015) Characterisation of the contractile dynamics of the resting ex vivo urinary bladder of the pig. BJU Int 116(6):973–983. https://doi.org/10.1111/bju.13132
Parsons BA, Drake MJ, Gammie A, Fry CH, Vahabi B (2012) The validation of a functional, isolated pig bladder model for physiological experimentation. Front Pharmacol. https://doi.org/10.3389/fphar.2012.00052
Adams SR, Dessie SG, Dodge LE, McKinney JL, Hacker MR, Elkadry EA (2015) Pelvic floor physical therapy as primary treatment of pelvic floor disorders with urinary urgency and frequency-predominant symptoms. Female Pelvic Med Reconstr Surg 21(5):252–256. https://doi.org/10.1097/SPV.0000000000000195
Kasman A, Stave C, Elliott CS (2019) Combination therapy in overactive bladder-untapped research opportunities: a systematic review of the literature. Neurourol Urodyn 38(8):2083–2092. https://doi.org/10.1002/nau.24158
Arnouk A, De E, Rehfuss A, Cappadocia C, Dickson S, Lian F (2017) Physical, complementary, and alternative medicine in the treatment of pelvic floor disorders. Curr Urol Rep 18(6):1–13. https://doi.org/10.1007/s11934-017-0694-7
Sugaya K, Nishizawa O, Satoh T, Hatano T (2000) Bladder-pumping therapy for the treatment of low-capacity or low-compliance bladders. Neurourol Urodyn 28:19–28
Sugaya K, Nishijima S, Hatano T, Ogawa Y, Kudo T, Nishizawa O (2002) Biochemical and morphological effects of bladder pumping on the urinary bladder in rats. Neurourol Urodyn 21(5):511–515. https://doi.org/10.1002/nau.10048
Mangera A et al (2014) An updated systematic review and statistical comparison of standardised mean outcomes for the use of botulinum toxin in the management of lower urinary tract disorders. Eur Urol 65(5):981–990. https://doi.org/10.1016/j.eururo.2013.10.033
Chen JL, Kuo HC (2020) Clinical application of intravesical botulinum toxin type a for overactive bladder and interstitial cystitis. Investig Clin Urol 61(Suppl 1):S33–S42. https://doi.org/10.4111/icu.2020.61.S1.S33
Cameron AP (2016) Medical management of neurogenic bladder with oral therapy. Transl Androl Urol 5(1):51–62. https://doi.org/10.3978/j.issn.2223-4683.2015.12.07
Speich JE, Southern JB, Henderson S, Wilson CW, Klausner AP, Ratz PH (2012) Adjustable passive stiffness in mouse bladder: regulated by Rho kinase and elevated following partial bladder outlet obstruction. Am J Physiol Ren Physiol. https://doi.org/10.1152/ajprenal.00177.2011
Funding
Funded by NIH Grant R01-DK101719.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No disclosures or conflicts of interest.
Ethics
All experiments in this study were approved by the institution’s animal care and use committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Duval, D.L.M., Weprin, S., Nandanan, N. et al. Regulation of bladder dynamic elasticity: a novel method to increase bladder capacity and reduce pressure using pulsatile external compressive exercises in a porcine model. Int Urol Nephrol 53, 1819–1825 (2021). https://doi.org/10.1007/s11255-021-02863-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-021-02863-1