Abstract
Purpose
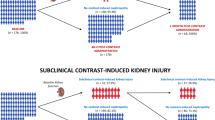
We performed a pilot study to evaluate the feasibility of future research about the presence of subclinical kidney damage after Gadolinium-based contrast media exposure. The future study aims to understand which are the behaviors of two markers of kidney damage, such as urinary NephroCheck (NC) and/or neutrophil gelatinase-associated lipocalin (NGAL). Specifically, after GBCM exposure, NC urinary detection should identify proximal tubule damage while NGAL urinary detection should be related to distal tubule damage.
Methods
We performed a pilot study in patients who had Gadolinium exposure. The feasibility of future study is reached when at least 90% of candidates completed the pilot study. In each patient, we tested urinary NC and NGAL levels 24 h before magnetic resonance imaging (MRI) and 12–24 h after the exposure. Furthermore, we evaluated the administration of other nephrotoxic agents, the presence of comorbidity, and kidney function by S-creatinine and urine protein before the MRI.
Results
We enrolled 35 candidates of whom 33 patients completed all study procedures. Our population had a mean age of 60.7 ± 14.8 years with normal kidney function with a median S-creatinine equal to 0.7 mg/dl (Interquartile range [IQR] 0.6–0.91). Urinary NC levels increased from 0.21 ng2/ml2 (IQR 0.11–0.4) before MRI to 0.34 ng2/ml2 (IQR 0.16–0.86) (p = 0.005). Conversely, we did not appreciate any significant modification in urinary NGAL (p = 0.53).
Conclusion
Our pilot study seems adequate in terms of feasibility and encourages us to focus our future research on renal proximal tubule, as the principal site of subclinical kidney damage after Gadolinium exposure.


Similar content being viewed by others
References
Elmståhl B, Leander P, Grant D, Doughty RW, Chai C-M, Björk J et al (2007) Histomorphological changes after renal X-ray arteriography using iodine and gadolinium contrast media in an ischemic porcine model. Acta Radiol. https://doi.org/10.1080/02841850701663442
Akgun H, Gonlusen G, Cartwright J Jr, Suki WN, Truong LD (2006) Are gadolinium-based contrast media nephrotoxic? Arch Pathol Lab Med. https://doi.org/10.1043/1543-165(2006)130[1354:AGCMNA]2.0.CO;2
Cacheris WP, Quay SC, Rocklage SM (1990) The relationship between thermodynamics and the toxicity of gadolinium complexes. Magn Reson Imaging. https://doi.org/10.1016/0730-725X(90)90055-7
Schmidt-Lauber C, Bossaller L, Abujudeh HH, Vladimer GI, Christ A, Fitzgerald KA et al (2015) Gadolinium-based compounds induce NLRP3-dependent IL-1β production and peritoneal inflammation. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2013-204900
Stutz A, Golenbock DT, Latz E (2009) Inflammasomes: too big to miss. J Clin Invest. https://doi.org/10.1172/JCI40599
Mawad H, Laurin LP, Naud JF, Leblond FA, Henley N, Vallée M et al (2016) Changes in urinary and serum levels of novel biomarkers after administration of gadolinium-based contrast agents. Biomark Insights. https://doi.org/10.4137/BMI.S39199
Spasojević-Dimitrijeva B, Kotur-Stevuljević J, Đukić M, Paripović D, Miloševski-Lomić G, Spasojević-Kalimanovska V et al (2017) Serum neutrophil gelatinase-associated lipocalin and urinary kidney injury molecule-1 as potential biomarkers of subclinical nephrotoxicity after gadolinium-based and iodinated-based contrast media exposure in pediatric patients with normal kidney function. Med Sci Monit. https://doi.org/10.12659/MSM.903255
Kashani K, Al-Khafaji A, Ardiles T, Artigas A, Bagshaw SM, Bell M et al (2013) Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit Care. https://doi.org/10.1186/cc12503
Price PN, Safirstein RL, Megyesi J (2009) The cell cycle and acute kidney injury. Kidney Int. https://doi.org/10.1038/ki.2009.224
McCullough PA, Shaw AD, Haase M, Bouchard J, Waikar SS, Siew ED et al (2013) Diagnosis of acute kidney injury using functional and injury biomarkers: workgroup statements from the tenth Acute Dialysis Quality Initiative Consensus Conference. Contrib Nephrol. https://doi.org/10.1159/000349963
U.S. Food and Drug Administration. Evaluation of Automatic Class III Designation for NephroCheckSystem® Test System Decision Summary. http://www.accessdata.fda.gov/cdrh_docs/reviews/den130031.pdf
Shang W, Wang Z (2017) The update of NGAL in acute kidney injury. Curr Protein Pept Sci. https://doi.org/10.2174/1389203717666160909125004
Schmidt-Ott KM, Mori K, Kalandadze A, Li JY, Paragas N, Nicholas T et al (2006) Neutrophil gelatinase-associated lipocalin-mediated iron traffic in kidney epithelia. Curr Opiom Nephrol Hypertens. https://doi.org/10.1097/01.mnh.0000232886.81142.58
Schmidt-Ott KM (2011) Neutrophil gelatinase-associated lipocalin as biomarker of acute kidney injury—where do we stand today? Nephrol Dial Transpalnt. https://doi.org/10.1093/ndt/gfr006
Andreucci M, Faga T, Pisani A, Perticone M, Michael A (2017) The ischemic/nephrotoxic acute kidney injury and the use of renal biomarkers in clinical practice. Eur J Intern Med. https://doi.org/10.1016/j.ejim.2016.12.001
Hertzog MA (2008) Considerations in determining sample size for pilot studies. Res Nurs Health. https://doi.org/10.1002/nur.20247
Ortega LM, Heung M (2018) The use of cell cycle arrest biomarkers in the early detection of acute kidney injury. Is this the new renal troponin? Nefrología. 38(4):361–367.https://doi.org/10.1016/j.nefro.2017.11.013
Devireddy LR, Hart DO, Goetz DH, Green MR (2010) A mammalian siderophore synthesized by an enzyme with a bacterial homolog involved in enterobactin production. Cell. https://doi.org/10.1016/j.cell.2010.04.040
Bao G, Clifton M, Hoette TM, Mori K, Deng S et al (2010) Iron traffics in circulation bound to a siderocalin (ngal)-catechol complex. J Nat Chem Biol. https://doi.org/10.1038/nchembio.402
Paragas N, Qiu A, Zhang Q, Samstein B, Deng SX, Schmidt-Ott KM et al (2011) The Ngal reporter mouse detects the response of the kidney to injury in real time. Nat Med. https://doi.org/10.1038/nm.2290
Schmidt-Ott KM, Mori K, Li JY, Kalandadze A, Cohen DJ, Devarajan P et al (2007) Dual action of neutrophil gelatinase-associated lipocalin. J Am Soc Nephrol. https://doi.org/10.1681/ASN.2006080882
Acknowledgements
The authors wish to thank the physicians and nurses at the Neuroradiology and Radiology Department for their work. Without their support, this work would not have been possible.
Funding
The study did not receive any kind of funding.
Author information
Authors and Affiliations
Contributions
All the authors have read and approved the final manuscript. Specifically, the authors’ contributions are: guarantors of the integrity of the entire study, FKM and CR; study concepts/study design or data acquisition, all the authors; manuscript drafting or manuscript revision for important intellectual content, all the authors; literature research/statistical analysis/data analysis/interpretation, FKM and GA; manuscript editing, all the authors.
Corresponding author
Ethics declarations
Conflict of interest
CR received compensation and consulted for Astute Medical, OCD, Asahi Medical, Baxter, and Toray Medical. All the other authors have no competing interests.
Ethical approval
The study was approved by the local institutional ethics committee (Resolution number 1449, 25 September 2019). The clinical investigation was conducted according to the principles expressed in the Declaration of Helsinki.
Consent to participate
All the enrolled patients signed informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Martino, F., Amici, G., Godi, I. et al. Gadolinium-based contrast media exposure and the possible risk of subclinical kidney damage: a pilot study. Int Urol Nephrol 53, 1883–1889 (2021). https://doi.org/10.1007/s11255-020-02779-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-020-02779-2