Abstract
Purpose
Previous studies have indicated that platelet indices are related to the pathogenesis of cardiovascular diseases (CVD). However, it is unclear which platelet-related indicators are associated with CVD events in patients with chronic kidney disease (CKD) without dialysis.
Methods
We performed a single-center prospective cohort study involved 1391 CKD patients to explore the relationship between platelet indices and CVD events in CKD patients. A nomogram was generated to predict CVD-free survival after 3 and 5 years of follow-up in terms of the fitted Cox regression model. And the time-dependent receiver-operating characteristic (ROC) curves were applied to evaluate the prediction accuracy of platelet indices on CVD events.
Results
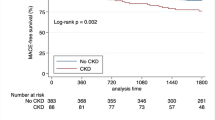
During a median follow-up of 3.41 years, 211 (15.2%) patients experienced CVD events. Results showed that platelet counts (PLT), plateletcrit (PCT), platelet-large cell ratio (P-LCR), and platelet distribution width (PDW) among 5 platelet indices were significantly lower in advanced CKD stages. Cox regression model showed that PLT, PDW, and PCT were associated with CVD events. However, after multivariable-adjusted, low level of PLT, hazard ratio (HR) 0.994 and 95% confidence interval (95% CI 0.989–1.000, p = 0.04), and PDW, HR 0.936 (95% CI 0.878–0.998, p = 0.044) predicted CVD events. The area under the ROC curve (AUC) of platelet indices assessed by time-dependent ROC curve analysis showed that only PLT and PDW were significant for predicting CVD events for 5 years.
Conclusions
We demonstrated that PLT and PDW among 5 platelet indices were independently associated with CVD events in patients with CKD.




Similar content being viewed by others

Data availability
Data can be obtained from the corresponding author on reasonable request.
References
Glassock RJ, Warnock DG, Delanaye P (2017) The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat Rev Nephrol 13:104–114. https://doi.org/10.1038/nrneph.2016.163
GBDCKD Collaboration (2020) Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet S0140–6736(20):30045–30053
Chen TK, Knicely DH, Grams ME (2019) Chronic kidney disease diagnosis and management: a review. JAMA 322:1294–1304. https://doi.org/10.1001/jama.2019.14745
Kahn MR, Robbins MJ, Kim MC, Fuster V (2013) Management of cardiovascular disease in patients with kidney disease. Nat Rev Cardiol 10:261–273. https://doi.org/10.1038/nrcardio.2013.15
Cachofeiro V, Goicochea M, de Vinuesa SG, Oubiña P, Lahera V, Luño J (2008) Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int Suppl 111:S4-9. https://doi.org/10.1038/ki.2008.516
Kim M, Kang SH, Kim JR, Park JJ, Cho YS, Youn TJ, Chae IH, Suh JW (2020) Comparison of shear stress–induced thrombotic and thrombolytic effects among 3 different antithrombotic regimens in patients with acute coronary syndrome. Clin Appl Thromb Hemost 26:1076029620912814
van der Meijden PE, Heemskerk JW (2019) Platelet biology and functions: new concepts and clinical perspectives. Nat Rev Cardiol 16:166–179. https://doi.org/10.1038/s41569-018-0110-0
Aljaroudi W, Halabi A, Harrington R (2005) Platelet inhibitor therapy for patients with cardiovascular disease: looking toward the future. Curr Hematol Rep 4:397–404
Chu S, Becker R, Berger P, Bhatt D, Eikelboom J, Konkle B, Mohler E, Reilly M, Berger J (2010) Mean platelet volume as a predictor of cardiovascular risk: a systematic review and meta-analysis. J Thromb Haemost 8:148–156. https://doi.org/10.1111/j.1538-7836.2009.03584.x
Yilmaz MB, Cihan G, Guray Y, Guray U, Kisacik HL, Sasmaz H, Korkmaz S (2008) Role of mean platelet volume in triagging acute coronary syndromes. J Thromb Thrombolysis 26:49–54. https://doi.org/10.1007/s11239-007-0078-9
Yang K, Du C, Wang X, Li F, Xu Y, Wang S, Chen S, Chen F, Shen M, Chen M, Hu M, He T, Su Y, Wang J, Zhao J (2017) Indoxyl sulfate induces platelet hyperactivity and contributes to chronic kidney disease–associated thrombosis in mice. Blood 129:2667–2679. https://doi.org/10.1182/blood-2016-10-744060
Ju HY, Kim JK, Hur SM, Woo SA, Park KA, Park MY, Choi SJ, Hwang SD (2017) Could mean platelet volume be a promising biomarker of progression of chronic kidney disease? Platelets 26:143–147. https://doi.org/10.3109/09537104.2014.890179
Yenigun EC, Aypak C, Turgut D, Piskinpasa SV, Cevher SK, Koc E, Dede F (2016) Is there a relation between mean platelet volume and chronic kidney disease stages in diabetic patients. Int J Clin Exp Med 9:330–335
Henning BF, Zidek W, Linder B, Tepel M (2002) Mean platelet volume and coronary heart disease in hemodialysis patients. Kidney Blood Press Res 25:103–108. https://doi.org/10.1159/000063516
Chi XH, Li GP, Wang QS, Qi YS, Huang K, Zhang Q, Xue YM (2017) CKD-EPI creatinine-cystatin C glomerular filtration rate estimation equation seems more suitable for Chinese patients with chronic kidney disease than other equations. BMC Nephrol 18:226. https://doi.org/10.1186/s12882-017-0637-z
Modi ZJ, Lu Y, Ji N, Kapke A, Selewski DT, Dietrich X, Abbott K, Nallamothu BK, Schaubel DE, Saran R, Gipson D (2019) Risk of cardiovascular disease and mortality in young adults with end-stage renal disease: an analysis of the US renal data system. JAMA Cardiol 4:353–362. https://doi.org/10.1001/jamacardio.2019.0375
Weiner DE, Tighiouart H, Amin MG, Stark PC, MacLeod B, Griffith JL, Salem DN, Levey AS, Sarnak MJ (2004) Chronic kidney disease as a risk factor for cardiovascular disease and all-cause mortality: a pooled analysis of community-based studies. J Am Soc Nephrol 15:1307–1315. https://doi.org/10.1097/01.asn.0000123691.46138.e2
Gansevoort RT, Correa-Rotter R, Hemmelgarn BR, Jafar TH, Heerspink HJ, Mann JF, Matsushita K, Wen CP (2013) Chronic kidney disease and cardiovascular risk: epidemiology, mechanisms, and prevention. Lancet 382:339–352. https://doi.org/10.1016/S0140-6736(13)60595-4
Ardhanari S, Alpert MA, Aggarwal K (2014) Cardiovascular disease in chronic kidney disease: risk factors, pathogenesis, and prevention. Adv Perit Dial 30:40–53
Linden MD, Jackson DE (2010) Platelets: pleiotropic roles in atherogenesis and atherothrombosis. Int J Biochem Cell Biol 42:1762–1766. https://doi.org/10.1016/j.biocel.2010.07.012
Bhatt DL, Eikelboom JW, Connolly SJ, Steg PG, Anand SS, Verma S, Branch KRH, Probstfield J, Bosch J, Shestakovska O, Szarek M, Maggioni A, Widimský P, Avezum A, Diaz R, Lewis B, Berkowitz S, Fox K, Ryden L, Yusuf S, COMPASS Steering Committee Investigators (2020) The Role of combination antiplatelet and anticoagulation therapy in diabetes and cardiovascular disease: insights from the COMPASS trial. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.120.046448
Cimmino G, Gallinoro E, Di Serafino L, De Luca N, Cirillo P (2020) Antiplatelet therapy in acute coronary syndromes. Lights and shadows of platelet function tests to guide the best therapeutic approach. Curr Vasc Pharmacol 18:262–272. https://doi.org/10.2174/1570161117666190513105859
Capodanno D, Bhatt DL, Eikelboom JW, Fox KAA, Geisler T, Michael Gibson C, Gonzalez-Juanatey JR, James S, Lopes RD, Mehran R, Montalescot G, Patel M, Steg P, Storey R, Vranckx P, Weitz J, Welsh R, Zeymer U, Angiolillo D (2020) Dual-pathway inhibition for secondary and tertiary antithrombotic prevention in cardiovascular disease. Nat Rev Cardiol 17:242–257. https://doi.org/10.1038/s41569-019-0314-y
McFadyen JD, Schaff M, Peter K (2018) Current and future antiplatelet therapies: emphasis on preserving haemostasis. Nat Rev Cardiol 15:181–191. https://doi.org/10.1038/nrcardio.2017.206
Königsbrügge O, Schmaldienst S, Auinger M, Klauser-Braun R, Lorenz M, Tabernig S, Kletzmayr J, Enzenberger B, Eigner M, Hecking M, Siller-Matula J, Pabinger I, Säemann M, Ay C (2020) Antithrombotic agents for primary and secondary prevention of cardiovascular events in patients with end-stage renal disease on chronic hemodialysis. Atherosclerosis 298:1–6. https://doi.org/10.1016/j.atherosclerosis.2020.02.011
Wang Q, Xie X, Xu G (2020) The risk of bleeding for antiplatelet agents in haemodialysis patients: a meta-analysis. BMC Nephrol 21:106. https://doi.org/10.1186/s12882-020-01757-1
Kim AJ, Lim HJ, Ro H, Ko K-P, Han SY, Chang JH, Lee HH, Chung W, Jung JY (2014) Low-dose aspirin for prevention of cardiovascular disease in patients with chronic kidney disease. PLoS ONE 9:e104179. https://doi.org/10.1371/journal.pone.0104179
Sun J, Axelsson J, Machowska A, Heimbürger O, Bárány P, Lindholm B, Lindström K, Stenvinkel P, Qureshi AR (2016) Biomarkers of cardiovascular disease and mortality risk in patients with advanced CKD. Clin J Am Soc Nephrol 11:1163–1172. https://doi.org/10.2215/CJN.10441015
Zaccardi F, Rocca B, Pitocco D, Tanese L, Rizzi A, Ghirlanda G (2015) Platelet mean volume, distribution width, and count in type 2 diabetes, impaired fasting glucose, and metabolic syndrome: a meta-analysis. Diabetes Metab Res Rev 31:402–410. https://doi.org/10.1002/dmrr.2625
Yilmaz G, Sevinc C, Ustundag S, Yavuz YC, Hacıbekiroglu T, Hatipoglu E, Baysal M (2017) The relationship between mean platelet volume and neutrophil/lymphocyte ratio with inflammation and proteinuria in chronic kidney disease. Saudi J Kidney Dis Transpl 28:90–94
Aziz AFA, Saad ATA, Bazeed MM, Allam M, Bakeer MS (2018) A comparative study of platelet parameters in chronic kidney disease, end stage renal disease patients undergoing hemodialysis and healthy individuals. Egypt J Hosp Med 71:3429–3433
Wang L, Kamijo Y, Matsumoto A, Nakajima T, Higuchi M, Kannagi R, Kyogashima M, Aoyama T, Hara A (2011) Kidney transplantation recovers the reduction level of serum sulfatide in ESRD patients via processes correlated to oxidative stress and platelet count. Glycoconj J 28:125–135. https://doi.org/10.1007/s10719-011-9329-2
Vagdatli E, Gounari E, Lazaridou E, Katsibourlia E, Tsikopoulou F, Labrianou I (2010) Platelet distribution width: a simple, practical and specific marker of activation of coagulation. Hippokratia 14:28–32
He S, Lei W, Li J, Yu K, Yu Y, Zhou L, Zhang X, He M, Guo H, Yang H, Wu T (2019) Relation of platelet parameters with incident cardiovascular disease (the dongfeng-tongji cohort study). Am J Cardiol 123:239–248. https://doi.org/10.1016/j.amjcard.2018.10.016
Tzur I, Barchel D, Izhakian S, Swarka M, Garach-Jehoshua O, Krutkina E, Plotnikov G, Gorelik O (2019) Platelet distribution width: a novel prognostic marker in an internal medicine ward. J Community Hosp Intern Med Perspect 9:464–470
Kern A, Gil RJ, Bojko K, Sienkiewicz E, Januszko-Giergielewicz B, Górny J, Bil J (2017) Platelet distribution width as the prognostic marker in coronary bifurcation treatment. Eur J Clin Invest 47:524–530. https://doi.org/10.1111/eci.12773
Boccardo P, Remuzzi G, Galbusera M (2004) Platelet dysfunction in renal failure. Semin Thromb Hemost 30:579–589. https://doi.org/10.1055/s-2004-835678
Kaw D, Malhotra D (2006) Platelet dysfunction and end-stage renal disease. Semin Dial 19:317–322. https://doi.org/10.1111/j.1525-139X.2006.00179.x
Molnar MZ, Streja E, Kovesdy CP, Budoff MJ, Nissenson AR, Krishnan M, Anker S, Norris K, Fonarow G, Kalantar-Zadeh K (2011) High platelet count as a link between renal cachexia and cardiovascular mortality in end-stage renal disease patients. Am J Clin Nutr 94:945–954. https://doi.org/10.3945/ajcn.111.014639
Peng F, Li Z, Yi C, Guo Q, Yang R, Long H, Huang F, Yu X, Yang X (2017) Platelet index levels and cardiovascular mortality in incident peritoneal dialysis patients: a cohort study. Platelets 28:576–584. https://doi.org/10.1080/09537104.2016.1246716
Zhu Y, Peng F, Chen Y, Chen W, Zhou W, Li P, Niu H, Long H (2018) Mean platelet volume/platelet count ratio and mortality in patients on peritoneal dialysis. Clin Nephrol 90:205–211. https://doi.org/10.5414/CN109329
Rechciński T, Jasińska A, Foryś J, Krzemińska-Pakuła M, Wierzbowska-Drabik K, Plewka M, Peruga J, Kasprzak J (2013) Damian prognostic value of platelet indices after acute myocardial infarction treated with primary percutaneous coronary intervention. Cardiol J 20:491–498. https://doi.org/10.5603/CJ.2013.0134
Funding
This work was supported by the Personal Training Program for Clinical Medicine Research of Army Medical University under Grant 2018XLC1007.
Author information
Authors and Affiliations
Contributions
Z.J.H. conceived the idea of the study; Y.Z.K. and X.J.C. analyzed the data; YK., H.Y.H., HT., and Y.Y.L. collected the data and interpreted the results; X.J.C. wrote the paper; all authors discussed the results and revised the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors report no conflict of interest.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted (IRB approval number 2018-006-01) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Written informed consent was obtained from all subjects before data collection.
Consent for publication
Written informed consent was obtained for publication of patient data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, Z., Xiong, J., Yang, K. et al. The association between platelet indices and cardiovascular events in chronic kidney disease patients without dialysis. Int Urol Nephrol 53, 961–971 (2021). https://doi.org/10.1007/s11255-020-02696-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-020-02696-4