Abstract
Background
At present, only one exchange of an icodextrin-based solution is recommended to increase peritoneal ultrafiltration (UF) during long-dwell exchanges in peritoneal dialysis (PD) patients with impaired UF.
Aim
To review our experience with two icodextrin exchanges per day on net UF and body weight in PD patients with poor UF.
Methods
Data were analyzed on nine patients with poor UF on chronic PD who were given two icodextrin exchanges per day for 6 months and had various clinical and biochemical parameters assessed monthly.
Results
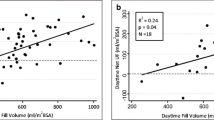
Administration of icodextrin twice daily reduced the body weight in six of nine patients by an average of 2.9 ± 1.2 kg, a reduction that was maintained throughout the study; two patients gained 0.5 kg; and, in one patient, the measurements were inadequate. Mean blood pressure was reduced. Mean serum creatinine increased slightly. Serum sodium levels decreased from a mean baseline level of 134 ± 3 to 132 ± 4 mmol/L at three and six months. Among the diabetics in this group, average daily insulin requirements were 44 ± 35 units/day at baseline and 40 ± 23 units/day after 6 months. Hb1Ac levels remained stable throughout the study period.
Conclusion
The use of two icodextrin exchanges per day reduced body weight in six of the nine patients and appeared to be safe. Long-term prospective studies are needed to assess the contribution of twice-daily icodextrin to the management of peritoneal dialysis patients with ultrafiltration failure and its long-term safety.

Similar content being viewed by others
References
Li PK, Chow KM (2007) Maximizing the success of peritoneal dialysis in high transporters. Perit Dial Int 27(Suppl 2):S148–S152
De Vriese AS, Mortier S, Lameire NH (2001) Glucotoxicity of the peritoneal membrane: the case for VEGF. Nephrol Dial Transplant 16:2299–2302
Davies SJ, Phillips L, Naish PF, Russell GI (2001) Peritoneal glucose exposure and changes in membrane solute transport with time on peritoneal dialysis. J Am Soc Nephrol 12:1046–1051
Burkart J (2004) Metabolic consequences of peritoneal dialysis. Semin Dial 17:498–504
Jolly S, Chatatalsingh C, Bargman J, Vas S, Chu M, Oreopoulos DG (2001) Excessive weight gain during peritoneal dialysis. Int J Artif Organs 24:197–202
Rippe B, Levin L (2000) Computer simulations of ultrafiltration profiles for an icodextrin-based peritoneal fluid in CAPD. Kidney Int 57:2546–2556
Finkelstein F, Healy H, Abu-Alfa A, Ahmad S, Brown F, Gehr T, Nash K, Sorkin M, Mujais S, The Icodextrin High Transport Trial Group (2005) Superiority of icodextrin compared with 4.25% dextrose for peritoneal ultrafiltration. J Am Soc Nephrol 16:546–554
Davies DS (1994) Kinetics of icodextrin. Perit Dial Int 14(Suppl 2):S45–S50
Posthuma N, ter Wee PM et al (2000) Assessment of the effectiveness, safety, and biocompatibility of icodextrin in automated peritoneal dialysis. The dextrin in APD in Amsterdam (DIANA) group. Perit Dial Int 20(S2):S106–S113
Liakopoulos V, Zarogiannis S, Eleftheriadis T, Dovas S, Stefanidis I (2007) Significant intra- and interpatient variation in ultrafiltration achieved by icodextrin in APD patients. Perit Dial Int 27(1):97–98
Ahmad M, Jeloka T, Pliakogiannis T, Tapiawala S, Zhong H, Bargman JM, Oreopoulos D (2008) Icodextrin produces higher ultrafiltration in diabetics that in non-diabetic patients on continous cyclic peritoneal dialysis. Int Urol Nephrol 40(1):219–223
Jeloka TK, Ersoy FF, Yavuz M, Sahu KM, Camsari T, Utah C et al (2006) What is the optimal dwell time for maximizing ultrafiltration with icodextrin exchange in automated peritoneal dialysis patients? Perit Dial Int 26(3):336–340
ISPD ed hoc committee on ultrafiltration management in peritoneal dialysis (2000) Perit Dial Int 20 (Suppl 4): S3–S4
Wolfson M, Ogrinc F, Mujais S (2002) Review of clinical trial experience with icodextrin. Kidney Int Suppl(81):S46–S52
Wilkie ME, Plant MJ, Edwards L, Brown CB (1997) Icodextrin 7.5% dialysate solution (glucose polymer) in patients with ultrafiltration failure: extension of CAPD technical survival. Perit Dial Int 17:84–87
Gokal R, Moberly J, Lindholm B, Mujais S (2002) Metabolic and laboratory effects of icodextrin. Kidney Int Suppl (81):S62–S71
Babazono T, Nakamoto H, Kasai K, Kuriyama S, Sugimoto T, Nakayama M, Hamada C, Furuya R, Hasegawa H, Kasahara M, Moriishi M, Tomo T, Miyazaki M, Sato M, Yorioka N, Kawaguchi Y, Japanese Extraneal Collaborated Study Group (2007) Effects of icodextrin on glycemic and lipid profiles in diabetic patients undergoing peritoneal dialysis. Am J Nephrol 27(4):409–415
Paniagua R, Ventura MD, Avila-Diaz M, Cisneros A, Vicente-Martinez M et al (2009) Icodextrin improves metabolic and fluid management in high and high average transport diabetic patients. Perit Dial Int 29(4):422–432
Hithaisi C, Lobbedez T, Padmanabhan S, Pineda ME, Oreopoulos DG (2004) No beneficial effect of icodextrin on blood glucose control. Perit Dial Int 24(2):199–200
Gokal R, Mistry CD, Peers EM (1995) Peritonitis occurrence in a multicenter study of icodextrin and glucose in CAPD. MIDAS study group. Multicenter investigation of icodextrin in ambulatory dialysis. Perit Dial Int 15:226–230
Posthuma N, ter Weel PM, Donker AJ et al (1998) Icodextrin use in CCPD patients during peritonitis: ultrafiltration and serum disaccharide concentrations. Nephrol Dial Transplant 13(9):2341–2344
Ahmad M, Shah H, Pliakogiannis T, Oreopoulos DG (2007) Prevention of membrane damage in patient on peritoneal dialysis with new peritoneal dialysis solutions. Int Urol Nephrol 39(1):299–312
Oreopoulos D, Thodis E, Paraskevas KI (2008) The promising future of long-term peritoneal dialysis. Int Urol Nephrol 40(2):405–410
Kössi J, Grönlund S et al (2009) The effect of 4% icodextrin solution on adhesiolysis surgery time at the Hartmann’s reversal: a pilot, multicentre, randomized control trial vs lactated Ringer’s solution. Colorectal Dis 11(2):168–172
Brown EA, Davies SJ, Rutherford P, Meeus F, Borras M, Riegel W, Divino Filho JC, Vonesh E, Van Bree M, EAPOS Group (2003) Survival of functionally anuric patients on automated peritoneal dialysis: the European APD outcome study. J Am Soc Nephrol 14(11):2948–2957
Gobin J, Fernando S, Santacroce S, Finkelstein FO (2008) The utility of two daytime icodextrin exchanges to reduce dextrose exposure in automated peritoneal dialysis patients: a pilot study of nine patients. Blood Purif 26(3):279–283
Sav T, Oymak O, Inanc MT, Dogan A, Tokgoz B, Utas C (2009) Effects of twice-daily icodextrin administration on blood pressure and left ventricular mass in patients on continous ambulatory peritoneal dialysis. Perit Dial Int 29(4):443–449
Mistry CD, Gokal R, Peers E, the MIDAS Study Group (1994) A randomized multicenter clinical trial comparing isosmolar icodextrin with hyperosmolar glucose solution in CAPD. Kidney Int 46:496–503
Plum J, Gentile S, Verger C, Brunkhorst R, Bahner U, Faller B et al (2002) Efficacy and safety of a 7.5% icodextrin peritoneal dialysis solution in patients treated with automated peritoneal dialysis. Am J Kidney Dis 39:862–871
Chan TM, Yung S (2007) Studying the effects of new peritoneal dialysis solutions on the peritoneum. Perit Dial Int 27(S2):S87–S93
Martikainen TA, Teppo AM, Gronhagen-Riska C, Ekstrand AV (2005) Glucose-free dialysis solutions:inductors of inflammation or preservers of peritoneal membrane? Perit Dial Int 25:453–460
Moriishi M, Kawanishi H (2008) Icodextrin and intraperitoneal inflammation. Perit Dial Int 28(S3):S96–S100
Acknowledgments
We thank Dr. Bengt Lindholm of Baxter Novum and Renal Medicine, Karolinska Institute, Stockholm, Sweden, for his critical review of the final manuscript and his important suggestion and Sandra Medeiros for her secretarial assistance in the preparation of this document.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dousdampanis, P., Trigka, K., Chu, M. et al. Two icodextrin exchanges per day in peritoneal dialysis patients with ultrafiltration failure: one center’s experience and review of the literature. Int Urol Nephrol 43, 203–209 (2011). https://doi.org/10.1007/s11255-010-9716-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-010-9716-9