Abstract
Background
Fibroblast growth factor-23 (FGF23) is a circulating factor that regulates renal reabsorption of inorganic phosphate. Serum FGF23 level is increased in chronic kidney disease (CKD) patients as a compensatory mechanism to hyperphosphataemia. FGF23 directly signals in the parathyroid glands and can be used to predict future secondary hyperparathyroidism in dialysis patients. We examined the relationship between FGF23 and serum calcium, phosphate, 1,25(OH)2D3, and PTH levels in haemodialysis patients.
Methods
FGF23 and the above mentioned characteristics were measured in 50 chronic haemodialysis patients. We analysed the correlation between FGF23 and the other characteristics by using the Pearson correlation coefficient and multiple regression analysis.
Results
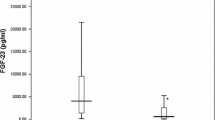
FGF23 was significantly increased in haemodialysis patients compared with healthy controls (1525 ± 373 vs. 37 ± 9 pg/ml, P < 0.0001). There was a significant negative correlation between log FGF23 and 1,25(OH)2D3 (R = −0.375, P = 0.009) and a significant positive correlation between log FGF23 and log PTH values (R = 0.287, P = 0.041). In multiple regression analysis log PTH and 1,25(OH)2D3 values were independent predictors of log FGF23 (P = 0.037 and 0.009, respectively).
Conclusions
Our results revealed a marked increase in FGF23 levels in haemodialysis patients. PTH and vitamin D3 were independent predictors of FGF23 in the study group. Serum phosphate did not correlate with or predict FGF23 level despite the high prevalence of hyperphosphataemia in the study group.



Similar content being viewed by others
References
Itoh N, Ornitz DM (2004) Evolution of FGF and FGF gene families. Trends Genet 20:563–569. doi:10.1016/j.tig.2004.08.007
Shimada T, Mizutani S, Muto T et al (2001) Cloning and characterization of FGF23 as a causative factor of tumor induced osteomalacia. Proc Natl Acad Sci USA 98:6500–6505. doi:10.1073/pnas.101545198
Yamashita T, Yoshioka M, Itoh N (2000) Identification of a novel fibroblast growth factor, FGF23, preferentially expressed in the ventrolateral thalamic nucleus of the brain. Biochem Biophys Res Commun 277:494–498. doi:10.1006/bbrc.2000.3696
Liu S, Guo R, Simpson LG et al (2003) Regulation of fibroblast growth factor 23 expression but not degradation by PHEX. J Biol Chem 278:37419–37426. doi:10.1074/jbc.M304544200
Liu S, Zhou J, Tang W et al (2006) Pathogenic role of FGF23 in Hyp mice. Am J Physiol Endocrinol Metab 291:E38–E49. doi:10.1152/ajpendo.00008.2006
Liu S, Quarles D (2007) How fibroblast growth factor 23 works. J Am Soc Nephrol 18:1637–1647. doi:10.1681/ASN.2007010068
Ornitz DM (2000) FGFs, heparan sulphate and FGFRs: complex interactions essential for development. Bioessays 22:108–112. doi :10.1002/(SICI)1521-1878(200002)22:2<108::AID-BIES2>3.0.CO;2-M
Urakawa I, Yamazaki Y, Shimada T et al (2006) Klotho converts canonical FGF receptor into a specific receptor for FGF23. Nature 444:770–774. doi:10.1038/nature05315
Shimada T, Kakitani M, Yamazaki Y et al (2004) Targeted ablation of FGF23 demonstrates an essential physiological role of FGF23 in phosphate and vitamin D metabolism. J Clin Invest 113:561–568
Kuro-o M, Matsumura Y, Aizawa H et al (1997) Mutation of the mouse Klotho gene leads to a syndrome resembling aging. Nature 390:45–51. doi:10.1038/36285
Liu S, Tang W, Zhou J et al (2006) Fibroblast growth factor 23 is a counter-regulatory phosphaturic hormone for vitamin D. J Am Soc Nephrol 17:1305–1315. doi:10.1681/ASN.2005111185
Peters BS, Moyses RM, Jorgetti V et al (2007) Effects of Parathyroidectomy on bone remodelling markers and vitamin D status in patients with chronic kidney disease-mineral and bone disorder. Int Urol Nephrol 39(4):1251–1256. doi:10.1007/s11255-007-9254-2
Saito H, Maeda A, Ohtomo S et al (2005) Circulating FGF23 is regulated by 1 alpha, 25 dihydroxyvitamin D3 and phosphorus in vivo. J Biol Chem 280:2543–2549. doi:10.1074/jbc.M408903200
Yamazaki Y, Okazaki R, Shibata M et al (2002) Increased circulatory level of biologically active full length FGF23 in patients with hypophosphatemic rickets/osteomalacia. J Clin Endocrinol Metab 87:4957–4960. doi:10.1210/jc.2002-021105
K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification and stratification (2002) Am J Kidney Dis 39: S1–S266
Larsson T, Nisbeth U, Ljunggren O et al (2003) Circulating concentration of FGF23 increases as renal function declines in patients with chronic kidney disease, but does not change in response to variation in phosphate intake in healthy volunteers. Kidney Int 64:2272–2279. doi:10.1046/j.1523-1755.2003.00328.x
Westerberg P, Linde T, Wikstrom B et al (2007) Regulation of fibroblast growth factor-23 in chronic kidney disease. Nephrol Dial Transplant 22:3202–3207. doi:10.1093/ndt/gfm347
Shigematsu T, Kazama JJ, Yamashita T et al (2004) Possible involvement of circulating fibroblast growth factor 23 in the development of secondary hyperparathyroidism associated with renal insufficiency. Am J Kidney Dis 44:250–256. doi:10.1053/j.ajkd.2004.04.029
Imanishi Y, Inaba M, Nakatsuka K et al (2004) FGF23 in patients with end-stage renal disease on hemodialysis. Kidney Int 65(5):1943–1946. doi:10.1111/j.1523-1755.2004.00604.x
Okada N, Hayashi D, Tomida K et al (2004) Role of FGF23 in secondary hyperparathyroidism in patients with mild chronic kidney disease. J Am Soc Nephrol 15:282A. doi:10.1097/01.ASN.0000136132.20189.95
Schiavi SC (2006) Fibroblast growth factor 23: the making of a hormone. Kidney Int 69:425–427. doi:10.1038/sj.ki.5000168
Perward F, Azam N, Zhang MYH et al (2004) Dietary phosphorus regulates serum FGF23 concentrations and 1,25 (OH)2D metabolism in mice. J Bone Miner Res 19:S251
Gupta A, Winer K, Econs MJ et al (2004) FGF23 is elevated by chronic hyperphosphataemia. J Clin Endocrinol Metab 89:4489–4492. doi:10.1210/jc.2004-0724
Urena-Torres P, Friedlander G, de Vernejoul MC et al (2008) Bone mass does not correlate with the serum fibroblast growth factor 23 in haemodialysis patients. Kidney Int 73:102–107. doi:10.1038/sj.ki.5002622
Jonsson K (2005) The role of fibroblast growth factor 23 in renal disease. Nephrol Dial Transplant 20:479–482. doi:10.1093/ndt/gfh701
Nakanishi S, Kazama JJ, Nii-Kono T et al (2005) Serum fibroblast growth factor-23 levels predict the future refractory hyperparathyroidism in dialysis patients. Kidney Int 67:1171–1178. doi:10.1111/j.1523-1755.2005.00184.x
Kazama JJ, Sato F, Omori K et al (2004) Pre-treatment serum FGF23 levels predict the efficacy of calcitriol therapy in dialysis patients with secondary hyperparathyroidism. J Am Soc Nephrol 15:282A
Wei M, Taskapan H, Esbaei K et al (2006) K/DOQI guidelines requirements for calcium, phosphate, calcium phosphate product, and parathyroid hormone control in dialysis patients: can we achieve them? Int Urol Nephrol 38(2):739–743. doi:10.1007/s11255-006-0082-6
Locatelli F, Pisoni RL, Combe C et al (2004) Anemia in hemodialysis patients of five European countries: association with morbidity and mortality in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Nephrol Dial Transplant 19:121–132. doi:10.1093/ndt/gfg458
Taskapan H, Wei M, Oreopoulos DG (2006) 25 (OH) vitamin D3 in patients with chronic kidney disease and those on dialysis: rediscovering its importance. Int Urol Nephrol 38(2):317–322. doi:10.1007/s11255-006-0081-7
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ibrahim, S., Rashed, L. Serum fibroblast growth factor-23 levels in chronic haemodialysis patients. Int Urol Nephrol 41, 163–169 (2009). https://doi.org/10.1007/s11255-008-9466-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-008-9466-0