Abstract
Diabetic nephropathy is one of the leading causes of end-stage renal disease (ESRD). There is some evidence that differences in extracellular fluid volume and capillary permeability do exist between diabetic and non-diabetic patients. This may have an impact on the gentamicin volume of distribution and clearance and thus dosing regimen. The pharmacokinetic parameters of gentamicin [volume of distribution (Vd), clearance (CL), elimination rate constant (K), and half life of elimination (t½)] were studied before hemodialysis in 20 non-diabetic patients (controls) and 20 diabetic patients, in addition to its hemodialysis clearance.
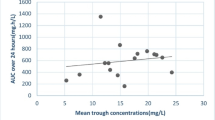
There were no statistically significant differences in Vd, CL, K, and t½ of gentamicin between the control and diabetic group. Therefore, a composite Vd of 0.25 l/kg and clearance of 0.063 ml/min/kg are suggested for dose calculation for both groups of patients. The mean hemodialysis clearance of gentamicin was higher in the diabetic (87.3 ml/min) than the control group (68.5 ml/min), comparing means by the unpaired t-test (P = 0.018).
In conclusion, the same method can be used to calculate the loading dose of gentamicin in ESRD patients between dialysis sessions in both controls and diabetics, whereas, diabetics are expected to receive a higher replacement dose after the end of dialysis.
Similar content being viewed by others
References
Curtis JR, McDonalds SJ, Weston JH (1967) Parenteral administration of Gentamicin in renal failure: patients undergoing intermittent hemodialysis. BMJ 2:537–539
De Broe ME, Yourassowsky E (1973) Gentamicin and hemodialysis. Acta Clin Belg 28:104–107
Christopher TG, Korn D, Blair AD, Forrey AW, O’Neill MA, Cutler RE (1974) Gentamicin pharmacokinetics during hemodialysis. Kidney Int 6:38–44. doi:10.1038/ki.1974.75
Halpren BA, Axline SG, Coplon NS, Brown DM (1976) Clearance of Gentamicin during hemodialysis: comparison of four artificial kidneys. J Infect Dis 133:627–636
Danish M, Schultz R, Jusko WJ (1974) Pharmacokinetics of Gentamicin and kanamycin during hemodialysis. Antimicrob Agents Chemother 6:841–847
Létourneau-Saheb L, Lapierre L, Daigneault R, Prud’Homme M, St-Louis G, Serois G (1977) Gentamicin pharmacokinetics during hemodialysis in patients suffering from chronic renal failure. Int J Clin Pharmacol Biopharm 15:116–120
Goetz DR, Pancorbo S, Hoag S, Bloom P (1980) Prediction of serum Gentamicin concentration in patients undergoing hemodialysis. Am J Hosp Pharm 37(8):1077–1083
Matzke GR, Halstenson CE, Keane WF (1984) Hemodialysis elimination rates and clearance of Gentamicin and tobramycin. Antimicrob Agents Chemother 25:128–130
Matzke GR (1994) Pharmacotherapeutic consequences of recent advances in hemodialysis therapy. Ann Pharmacother 28:512–514
Matzke GR, Millikin SP (1992) Influence of renal function and dialysis on drug disposition. In: Evans , Schentag , Jusko (eds) Applied pharmacokinetics: principles of therapeutic drug monitoring, 3rd edn. Applied Therapeutics, Inc, Vancouver, WA
Agarwal R, Toto RD (1993) Gentamicin clearance during hemodialysis: a comparison of high-efficiency cuprammonium rayon and conventional cellulose ester hemodialyzers. Am J Kidney Dis 22:296–299
Amin NB, Padhi ID, Touchette MA, Patel RV, Dunfee TP, Anandan JV (1999) Characterization of Gentamicin pharmacokinetics in patient hemodialyzed with high-flux Polysulffone membranes. Am J Kidney Dis 34:222–227. doi:10.1016/S0272-6386(99)70347-1
Akmal M (2001) Hemodialysis in diabetic patients. Am J Kidney Dis 38(Suppl 1):S195–S199. doi:10.1053/ajkd.2001.27443
Romagnoli GF, Di Landro D, Catalano C, Goepel V, Milan Manosi S, Ruffatti AM et al (1998) Short-term outcome of diabetic patients in renal replacement therapy. Nephrol Dial Transplant 13(Suppl 8):30–34. doi:10.1093/ndt/13.suppl_8.30
Oomen PH, Jager J, Hoogenberg K, Dullaart RP, Reitsma WD, Smit AJ (1999) Capillary permeability is increased in normo- and microalbuminuric type 1 diabetic patients: amelioration by ACE-inhibition. Eur J Clin Invest 29:1035–1040. doi:10.1046/j.1365-2362.1999.00568.x
Belcaro G, Nicolaides AN, Volteas N, Leon M (1992) Skin flow and the venoarterial response and capillary filtration in diabetics: a 3 years follow-up. Angiology 43:490–495. doi:10.1177/000331979204300606
Ubels FL, Muntinga JH, Links TP, Hoogenberg K, Dullaart RP, Reitsma WD et al (2001) Redistribution of blood volume in type 1 diabetes. Diabetologia 44:429–432. doi:10.1007/s001250051639
Kaufmann A, Molnar B, Craciun C, Itcus A (1980) Diabetic lymphangiopathy: an optical and electron microscopic study. Lymphology 13:202–206
Valensi P, Attali JR, Behar A, Sebaoun J (1987) Isotopic test of capillary permeability to albumin in diabetic patients: effects of hypertension, microangiopathy, and duration of diabetes. Metabolism 36:834–839. doi:10.1016/0026-0495(87)90090-4
Scholey JW (2003) Angiotensin II and the glomerulous: focus on diabetic kidney disease. Curr Hypertens Rep 5:172–180. doi:10.1007/s11906-003-0075-0
Matzke GR, Comstock TJ (2006) Influence of renal function and dialysis on drug disposition. In: Burton Shaw, Shentag Evans (eds) Applied Pharmacokinetics and pharmacodynamics: principles of therapeutic drug monitoring, 4th edn. Lippincott Williams & Wilkins, Philadelphia, USA
Vercaigne LM, Ariano RE, Zacharias JM (2004) Bayesian pharmacokinetics of Gentamicin in a haemodialysis population. Clin Pharmacokinet 43:205–210. doi:10.2165/00003088-200443030-00004
Dager WE, King JH (2006) Aminoglycosides in intermittent hemodialysis: pharmacokinetics with individual dosing. Ann Pharmacother 40:9–14. doi:10.1345/aph.1G064
Teigen MM, Duffull S, Dang L, Johnson DW (2006) Dosing of Gentamicin in patients with end-stage renal disease receiving hemodialysis. J Clin Pharmacol 46:1259–1267. doi:10.1177/0091270006292987
Dang L, Duffull S (2006) Development of a semi mechanistic model to describe the pharmacokinetics of Gentamicin in patients receiving hemodialysis. J Clin Pharmacol 46:662–673. doi:10.1177/0091270006286902
Sombolos K, Tsitamidou Z, Kyrianzis G, Karagianni A, Kantaropoulou M, Progia E (1997) Clinical evaluation of four different high-flux hemodialyzers under conventional conditions in vivo. Am J Nephrol 17:406–412
Bauer LA (2006) Clinical pharmacokinetics handbook. McGraw Hill, New York, USA
Acknowledgment
This research was supported by grant LGP-9-40 from King Abdulaziz City for Science and Technology, General Directorate of Research Grant Programs, Riyadh, Saudi Arabia. The expert technical assistance of Mr. Riyadh Alessa is greatly appreciated. The authors extend their thanks to the nursing staff at Asir Kidney Unit for facilitation of the research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by KACST grant LGP-9-40.
Rights and permissions
About this article
Cite this article
Al-Homrany, M.A., Irshaid, Y.M., El Sherif, A.K. et al. Pharmacokinetics of gentamicin in hemodialysis patients: a comparative study between diabetic and non-diabetic patients. Int Urol Nephrol 41, 663–669 (2009). https://doi.org/10.1007/s11255-008-9456-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-008-9456-2