Abstract
Urbanization has increased wealth disparity within the United States, impacting the urban landscape and species interactions. In particular, the interactions between street trees and the arthropod communities that live among them may be modified by both human population densities across urban to suburban locations, as well as income levels within these areas. We examined the effect of land use type (urban vs. suburban) and median household income on variation in leaf damage and arthropod abundance of red maples (Acer rubrum) in the District of Columbia metropolitan region. We compared these levels of leaf damage to rates observed in a nearby temperate deciduous forest. We predicted leaf damage would be positively correlated with urbanization (forested < suburban < urban) and negatively correlated with neighborhood income level (low > medium > high). Instead, we observed higher levels of leaf damage on trees in the forest environment compared to the urban and suburban areas. Leaves from urban medium and high-income areas were less likely to exhibit herbivore damage than those from suburban areas. Of the leaves with damage, those in urban high-income and suburban low-income areas exhibited the most leaf area missing. These trends may be related to specific factors associated with urbanization and income level, such as impervious surface coverage and tree coverage. This study highlights differences in biotic interactions across individual neighborhoods and the importance of including socio-economic variables when examining species interactions in urban environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Street trees are key components of many urban ecosystems. In particular, street trees provide valuable ecosystem services to their surrounding environment through environmental, economic, and social benefits for their city’s residents (Beecham and Lucke 2015; Donovan and Butry 2010; Landry and Chakraborty 2009; Mullaney et al. 2015; Soares et al. 2011; Turner-Skoff and Cavender 2019; Wolf et al. 2020). Additionally, urban street trees can serve as habitats for various species, increasing species biodiversity in urban ecosystems (Pataki et al. 2021). While tree health is impacted by many abiotic and biotic factors (Hilbert et al. 2018), herbivory in particular can have large consequences for tree growth, success, and survival (Crawley 1989). On average, plants suffer greater effects from invertebrates than from vertebrate herbivores (Bigger and Marvier 1998). While most damage from an individual herbivorous insect may be minor, collectively they can have a substantial impact, even at low densities (Kozlov and Zvereva 2018). Foliar herbivory directly removes tissue, which decreases leaf area and limits photosynthetic capabilities, alters growth, causes leaf abscission, can induce costly defenses, and can make leaves vulnerable to pathogens (Björkman et al. 2008; Kozlov et al. 2012, 2014; Marquis and Whelan 1994; Nabity et al. 2009; Welter 1989; Zangerl et al. 2002). This damage is further propelled as decreased photosynthesis can lead to reduced bud development and production of smaller leaves, creating a positive feedback loop by providing less support to subsequent buds, resulting in shorter shoots the following growing season (Zvereva et al. 2012). Thus, understanding the effects of foliar insect herbivory on urban street trees is essential for promoting their health and long-term maintenance.
Previous research shows conflicting patterns related to herbivore abundance and herbivory rates on trees in urban settings, with some finding that urban trees experienced higher levels of insect herbivory relative to rural habitats (Cuevas-Reyes et al. 2013; Turrini et al. 2016), while others found decreased herbivory in urban environments (Bode and Gilbert 2015; Herrmann et al. 2012; Kozlov et al. 2017; Moreira et al. 2019). Thus, there is little evidence for a universal increase in herbivore abundance or species richness along urbanization gradients, although guild-specific trends have been identified (Raupp et al. 2010). For example, Raupp et al. (2010) found that small arthropods with sucking mouthparts, limited mobility, and multiple generations on the same host plant generally increase along an urbanization gradient. Although urbanization’s effect on street trees’ herbivory have been studied across several cities (Moreira et al. 2019), no studies have considered how economic factors within urban areas may impact herbivory.
While President Franklin D. Roosevelt’s 1932 New Deal program attempted to boost the U.S. economy during the Great Depression, it resulted in lasting effects on the racial and socioeconomic makeup of communities; redlining and restricting people of a certain race or class to specific neighborhoods became common (Hillier 2003, Appel and Nickerson 2016). Due to historic redlining, communities within cities can vary significantly in features and characteristics, including impervious surface cover, green space, and the presence and community composition of street trees (Burghardt et al. 2023; Chakraborty et al. 2019; Hoffman et al. 2020; Hsu et al. 2021; Nardone et al. 2021). These environmental differences across income gradients within a city suggest that generalizations of plant-herbivore interactions across an entire city may not accurately reflect interactions within specific neighborhoods, and that a more nuanced approach to examining these dynamics across neighborhoods with different income levels may be important.
Red maple (Acer rubrum) is native to and ubiquitous across the eastern United States (Walters and Yawney 1990). Red maple is a popular street tree species planted for its generalist nature and ability to grow in a range of soil conditions (National Wildlife Federation 2021), and as such research on red maples in an urban context is widespread. For example, red maples in North Carolina are negatively impacted by urban warming (Dale and Frank 2014a, b) due to increased fecundity, abundance, and survival of the gloomy scale (Melanaspis tenebricosa) (Dale and Frank 2017). However, few studies have examined damage from leaf chewing insect herbivores on red maple in urban environments, and none in the context of variation in neighborhood income.
In this study, we examined 1800 red maple leaves to determine whether herbivory varies along an urbanization gradient and across income levels within urbanization types in the D.C. metropolitan area. We compared herbivorous insect chewing damage on leaves of red maple in the urban District of Columbia and suburban Montgomery County, Maryland, with the temperate deciduous forest of the Smithsonian Environmental Research Center (SERC) in Edgewater, Maryland as a natural reference site. We predicted that leaf damage would be positively correlated with urbanization (urban > suburban > forested). We also examined whether income level within the urban and suburban areas relates to leaf damage or arthropod abundances. We predicted a negative relationship between arthropods abundance and neighborhood income levels, with arthropod abundance and herbivory being inversely related to income (low > medium > high income).
Methods
Study sites
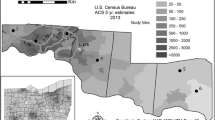
The D.C. Metropolitan area encompasses the District of Columbia (D.C.) and surrounding Maryland and Virginia suburban areas. Here we focus on sites in the District of Columbia, Montgomery County, Maryland and Anne Arundel County, Maryland. We selected study sites based on red maple presence and then categorized these sites based on green space, percent pavement cover, and income (Table 1) as urban (District of Columbia), suburban (Montgomery County), and the temperate deciduous forest on SERC’s campus (Anne Arundel County). D.C. features many common metropolitan characteristics such as a dense road network and many large buildings. Using this anecdotal knowledge, we then quantified the difference between area types (urban versus suburban) using impervious surface and tree coverage (extracted from the National Land Cover Database, 2019). Excluding SERC, our study sites with more than 50% impervious surface coverage and less than 50% tree coverage were classified as urban, and vice versa for suburban areas. Within the urban and suburban locations, we chose three neighborhoods to span median household income levels (Fig. 1; Table 1). We used census data (2022) to determine the median household income of each census block. We then assigned neighborhoods as high, middle, or low-income based on census-designated income brackets typical for each region (Montgomery County, MD and D.C.) (Table 1). Within D.C. (urban), we included the Anacostia (low-income), Brightwood (middle-income), and Georgetown (high-income) neighborhoods. In Montgomery County (suburban), we chose the White Oak (low-income), Rockville (middle-income), and Travilah (high-income) neighborhoods. We used three sites within the 2650-acre forested grounds of the Smithsonian Environmental Research Center (Anne Arundel County, Maryland) as a natural forest (low-human impact) reference (Fig. 1).
Map of the greater Washington, DC area with neighborhood study sites colored based to income level (yellow, low; medium, orange; high, navy blue). Urban sites were all within the Washington, DC boundaries, depicted by the black square line. Suburban sites were in the surrounding area and the temperate deciduous forest in Edgewater, Maryland is depicted with a star
Study species
We chose red maple as a study species, as it is abundant across land-use types in the study region and the Northeast United States. In July of 2021, we sampled 10 red maple trees at each of our 9 study sites (N = 90 trees across all sites). We selected trees haphazardly, ensuring no directly neighboring trees were sampled. Due to differences in land use history at each site, there was some variation in tree size and age both within and among sites. We attempted to control for this by choosing healthy, mature trees with no signs of signs of disease and fully developed canopies, that were at least 10 cm in diameter at breast height (DBH) but with lower canopies no more than 8 m from the ground. At the rural site, we were more limited in tree selection as trees were either very large with inaccessible canopies, or smaller than most of the trees sampled in urban and suburban sites. Because of this constraint, more of the trees sampled at rural sites fell closer to the lower size limit than in urban and suburban sites. For each sample tree, we visually divided the canopy into four quadrants for sampling the arthropod community (urban and suburban sites) and herbivorous insect foliar damage (all sites).
Arthropod collection
Within the urban and suburban sites, we used a 91.44 × 91.44 cm beat sheet to collect arthropods from each of the four quadrants of each sample tree. We used a 30-foot pole to beat the branches in each quadrant for 15 s (1 min total per tree). Sampling in quadrants allowed us to ensure that the beat sheet was under the area of tree being beaten, without disturbing other areas of the canopy before they could be sampled. Arthropods that fell from the tree were collected from the beat sheet using an aspirator and placed into labeled collection tubes. Total arthropod abundance per tree was based on the summing of abundances found in each quadrant. Arthropods were not sampled from the forest sites due to the general smaller size of these trees, as we could not collect a full sample of each quadrant without disturbing the entire tree.
We sorted arthropods into leaf chewing vs. piercing herbivores and predators based on broad taxonomic groupings using protocols developed by the Herbivory Variability Network (The Herbivory Variability Network et al. 2023). Chewing herbivores identified included: grasshoppers/crickets/katydids (Orthoptera), caterpillars (Lepidoptera), and herbivorous beetles (Coleoptera: Chrysomelidae, Elateridae). Piercing herbivores identified included: hoppers (Hemiptera: Cicadellidae, Membracidae), aphids (Hemiptera: Aphidae), thrips (Thysanoptera), mirids (Hempitera: Miridae), whiteflies (Hemiptera: Aleyrodidae), mealybugs (Hemiptera: Pseudococcidae), and scale insects (Hemiptera: Coccoidea). Predators identified included: wasps (Hymenoptera), spiders (Araneae), assassin bug (Hemiptera: Reduviidae), and predatory beetles (Coleoptera: Coccinellidae, Staphylinidae).
Leaf collection
We collected twenty leaves from each sample tree (N = 1800 leaves in total), with five leaves collected within each of the four quadrants of each tree. We used a 30-foot extendable pole pruner, to randomly select leaves from branches at various heights and different distances from the tree’s trunk to provide a comprehensive sample of each entire tree. To ensure the leaves suffered no additional damage after collection, we placed all collected leaves in bags labeled by the site/tree number, stored in a cooler for transport to the lab, and kept refrigerated until processing.
We recorded percent damage due to herbivory for all sampled leaves using the LeafByte application (Getman-Pickering et al. 2020). We took photos of all leaves within a week of collection and uploaded them to LeafByte. In some cases, when large sections of the leaf were missing, we made an educated guess of the original outlines of the leaf, and the percent missing was then calculated with this supplemented information. In addition, we took great care to ensure only chewing herbivory was considered. We excluded instances of tissue loss due to fungal infection, tearing, wind, or sun damage from the total percentage using the exclude feature in the LeafByte application.
Statistical analysis
We completed all statistics in R version 4.1.3 (R Core Team 2022). Leaf damage data were continuous, over-dispersed, and highly zero-inflated. As such, we conducted two-stage mixed-effects models using the lme4 package (Bates et al. 2015) to examine the effects of (a) land use type, including all data from forest, suburban, and urban sites, and (b) interactive effects of land use and income for the suburban and urban sites only. These two-stage models consisted of first examining the probability of observing zero values for percent leaf damage using logistic regression with a binomial distribution (Campbell 2021; Young et al. 2022) using the lme4::glmer() function, and including either (a) land use for models that did not include income (comparing urban, suburban, and forest) or (b) land use by income interactions for models that did include income (comparing urban and suburban neighborhoods) as fixed factors and tree identity as a random effect. Then we examined the non-zero continuous data using a second model using the lme4::lmer() function with log-transformed percent leaf damage as the dependent variable, land use or land use by income interactions (as described above), as appropriate, as fixed factors, and tree identity as a random effect. Damage data were log-transformed to improve normality of the non-zero values.
Chewing herbivore, piercing herbivore, and predator count data were over-dispersed and zero-inflated. We analyzed these data for the suburban and urban sites only using the glmmTMB::glmmTMB() function (Brooks et al. 2017), including land use by income interactions as fixed factors, tree identity as a random effect, either chewing herbivore, piercing herbivore, or predator abundance as a dependent variable, and using a single zero-inflation parameter and a negative binomial distribution (Payne et al. 2018).
Results
Leaf damage by area type and income
Roughly 35% (635 of 1800 leaves) of leaves sampled experienced no damage. Land use significantly affected the incidence of leaf herbivory, with a significantly higher probability of no damage on urban leaves than suburban leaves and the lowest probability of no damage on forest leaves (Fig. 2; x2 = 227.24, df = 2, p < 0.001). Of the leaves with any damage, most damage values were low, with 45% (526 leaves) of damaged leaves having less than 1% damage and an additional 53% (614 leaves) of damaged leaves having between 1 and 20% damage. When considering leaves with damage, urban and suburban leaves had significantly lower average percent damage than forest leaves (Fig. 2; x2 = 8.07, df = 2, p = 0.018; means ± standard errors were as follows, Urban: 0.89% ± 0.12%, Suburban: 0.93% ± 0.09%, Forest: 1.23% ± 0.12%). Despite these average trends, urban and suburban leaves were more likely to experience very high levels of leaf damage (Figs. 2 and 8 urban leaves and 13 suburban leaves experienced greater than 20% damage, compared to only 4 forest leaves with such high damage values). However, the small sample size of leaves with extreme damage makes this trend difficult to statistically compare.
Distribution of leaf damage values observed across land use types. Lowercase letters along the bottoms of the distributions represent significant differences in the probability of occurrence of zero leaf damage among neighborhoods, while uppercase letters along the top represent significant differences in the mean percent damage of leaves where damage was observed. N = 600 leaves per land use type
When considering only the urban and suburban sites, income significantly interacted with land use to impact percent leaf damage (Fig. 3). Specifically, a significantly higher probability of no damage was observed for urban leaves in medium and high-income neighborhoods than urban low-income neighborhoods, and suburban low and high-income neighborhoods were less likely to have no damage leaves than urban neighborhoods of any income level (Fig. 3; x2 = 10.66, df = 2, p = 0.005). When considering leaves with damage, leaves from suburban high-income and urban low-income neighborhoods had significantly greater damage than leaves from all other neighborhoods (Fig. 3; x2 = 37.41, df = 2, p < 0.001; means ± standard errors were as follows, Suburban Low: 0.78% ± 0.13%, Suburban Medium: 0.59% ± 0.10%, Suburban High: 1.56% ± 0.25%, Urban Low: 1.83% ± 0.35%, Urban Medium: 0.70% ± 0.16%, Urban High: 0.54% ± 0.13%).
Distribution of leaf damage values observed across land use types stratified by neighborhood income level. Lowercase letters along the bottoms of the distributions represent significant differences in the probability of occurrence of zero leaf damage among neighborhoods, while uppercase letters along the tops represent significant differences in the mean percent damage of leaves where damage was observed. N = 200 leaves per category. See Table 1 for median household income for each neighborhood
Arthropods abundance by area type and income
Across the urban and suburban sites, there was a significant interactive effect of land use and neighborhood income on invertebrate chewing herbivore (Χ2 = 19.32, df = 2, p < 0.001), invertebrate piercing herbivore (Χ2 = 217.00, df = 2, p < 0.001), and predator abundances (Χ2 = 124.69, df = 2, p < 0.001). Specifically, chewing herbivore abundances were lowest in the urban areas, intermediate in the suburban low and medium-income neighborhoods, and highest in the suburban high-income neighborhood (Fig. 4). Piercing herbivore abundances were lowest in the urban and suburban low-income neighborhoods, intermediate in the urban medium and high-income neighborhoods, and highest in the suburban medium and high-income neighborhoods (Fig. 4). Predator abundances were lowest in the urban low-income neighborhood, intermediate in the urban medium and high and suburban low and medium-income neighborhoods, and highest in the suburban high-income neighborhood (Fig. 4).
Distribution of (left) chewing herbivore, (center) piercing herbivore, and (right) predator abundance values observed across land use types stratified by neighborhood income level. Lowercase letters represent significant differences in the insect abundances across neighborhoods. Note the differences in scale of the y-axis across panels. N = 10 trees per category
Discussion
Overall, we found higher abundances of arthropods, including chewing and piercing herbivores and predators, in suburban locations than in urban areas, indicating that urbanization might decrease arthropod abundance. One might say there are more “bugs in the ‘burbs” (in the United States, burbs is a colloquial term for suburban areas and by bugs we mean specifically chewing herbivores). Our research adds to the literature, which demonstrates conflicting patterns of insect abundances across urbanization gradients, with guild specific trends (Raupp et al. 2010). One factor that may have impacted herbivore abundance in our study is the difference in habitat availability between urban and suburban locations. Our suburban sites had almost two times the tree coverage compared to the selected urban areas (Table 1). The lower tree coverage in the urban sites may have resulted in decreased habitat availability, and potentially overcrowding of arthropods on individual trees (Mullu 2016). Similarly, habitat fragmentation and physical barriers (buildings) may make it difficult for many arthropod species to migrate among trees in urban environments (Fenoglio et al. 2021). For example, Penone et al. (2013) found that mobile Orthopterans were more sensitive to increased impervious surface cover (barriers between habitat fragments) compared to sedentary species. These factors could result in increased competition between urban-dwelling arthropods if fewer habitats are present and accessible (Harrison and Bruna 1999; Nee and May 1992; Wilson et al. 2016), while a higher number and density of trees in the suburbs could promote arthropod survival and abundance.
In addition to reduced habitat availability, habitat quality may also be lower in urban areas compared to the suburbs. Streets and sidewalks confine urban street trees into small regulated grids that reduce nutrient and water uptake by limiting space and increasing soil compaction (Dale and Frank 2014b). This nutrient and water stress impacts street tree health, including reduced gas exchange rates, increased xylem embolism, and subsequent cavitation within vascular tissue (Savi et al. 2015; Sperry and Tyree 1988). Changes in tree quality may in turn impact the herbivorous insects feeding on them. For example, armored-scale insect survival and abundance declines with increasing water stress on street trees (Cockfield and Potter 1986; Hanks and Denno 1993). Altogether the increased number of herbivorous insects on street trees in suburban areas compared to urban areas in our study might be due to underlying drivers of habitat quantity (impervious surface coverage and tree cover) and quality.
We found that the incidence of leaf damage on street trees was significantly lower than in the nearby temperate deciduous forest, with more non-damaged leaves in urban > suburban > forest areas. This result is consistent with recent research (Bode and Gilbert 2015; Herrmann et al. 2012; Kozlov et al. 2017; Moreira et al. 2019; Schueller et al. 2019), which has found decreased insect herbivory with increasing urbanization, thus contributing to a growing body of literature that contradicts past findings that herbivory increases with urbanization (Cuevas-Reyes et al. 2013; Dreistadt et al. 1990; Raupp et al. 2010). Further, the lower incidence of leaf damage in urban areas compared to suburban areas in our study correlates with our observed decreases in chewing herbivores in our urban sites compared to our suburban sites.
The low levels of herbivory in urban areas in our study might be attributed to bottom-up controlling factors common to urban environments. Irrigation and pruning result in foliage with increased nitrogen and decreased secondary defense compounds (Raupp et al. 2010), while increased carbon dioxide and nitrogen levels are associated with lower defense levels (Moreira et al. 2019). Fossil fuel pollution from transportation, runoff due to high impervious surface coverage (e.g., middle-income urban site and high-income urban site with around 60% impervious surface cover), or direct fertilizer application all lead to increased nitrogen content of plants (Nikula et al. 2010; Searle et al. 2011, 2012). Counterintuitively, this increased leaf quality in urban areas may lead to lower levels of herbivory, as herbivores can each consume less per capita and still fulfill their nutritional needs (Kozlov et al. 2017; La Pierre and Smith 2016).
Our study adds a layer of complexity to the existing literature by investigating the impacts of neighborhood income on leaf damage and arthropod abundances. In addition to differences across the urbanization gradient, we observed significant interactive effects of neighborhood income with urbanization for both insect abundance and leaf damage. Specifically, we found the fewest chewing and piercing herbivores in low-income areas (urban and suburban), followed by urban medium- and high-income areas, and finally suburban medium- and high-income areas had the highest herbivore abundances. Similarly, the fewest predators were found in urban low-income areas and the most in suburban high-income areas. While we found more non-damaged leaves in the urban medium and high-income areas, when excluding zero-damage leaves, we surprisingly found similar average amounts of damage across the income spectrum: suburban high-income and urban low-income areas both had leaves with the greatest herbivore damage.
Interestingly, we found the same level of herbivory in the urban low-income neighborhood as in the suburban high-income neighborhood. With the highest tree coverage (63%) and lowest impervious surface coverage (7%; Table 1), the high-income suburban site has environmental conditions that are similar to those experienced by temperate deciduous forest trees, especially considering that wooded areas surround the neighborhood. High herbivory rates may stem from a “spillover effect” of herbivorous insects from nearby forested areas (Blitzer et al. 2012). Although residents in the urban low-income site have the lowest median household income in our study, their neighborhood has the highest tree coverage and lowest impervious surface of the three urban D.C. sites (Table 1). The urban low-income site in our study has multiple parks within and adjacent to the neighborhood, making it similar to the suburban high-income site. This green space could explain the similarity in rates of herbivory on street trees between the urban low-income and suburban high-income site, as the same mechanism of spillover could be responsible.
Street trees play important roles in urban and suburban areas and variations in herbivory can impact the ecosystem services street trees provide humans. Urbanization results in more buildings and paved roads, contributing to the urban island heat effect (Arnfield 2003; Freedman 1995; Oke and Maxwell 1975; Price 1979) and increased runoff of pollutants (Haughton and Hunter 2004). Vegetated areas with plants and street trees help lower temperatures (Armson et al. 2012) and absorb runoff (Bolund and Hunhammar 1999). Considering all the benefits urban street trees provide residents, especially to those in low-income neighborhoods, it is important to understand the ecology of street trees in suburban and urban ecosystems. While the scope of our study and results are restricted to the D.C. metropolitan area, our work contributes to a growing body of literature demonstrating that urbanization, neighborhood income, and related factors (e.g., impervious surface and tree canopy coverage) may influence plant-insect interactions. Future research should expand on this work and consider how the unique landscape characteristics of cities that are driven by income disparities impact arthropod abundance and herbivory. Understanding the specific mechanisms driving differences in plant-herbivore interactions across income levels in urban ecosystem can help better inform street tree management in the future.
Data availability
Statistical data that support the findings of this study are posted in a GitHub repository.
References
Appel I and Nickerson J (2016) Pockets of Poverty: The Long-Term Effects of Redlining SSRN Electronic Journal 10.2139/ssrn.2852856
Armson D, Stringer P, Ennos AR (2012) The effect of tree shade and grass on surface and globe temperatures in an urban area. Urban Forestry Urban Green 11(3):245–255. https://doi.org/10.1016/j.ufug.2012.05.002
Arnfield AJ (2003) Two decades of urban climate research: a review of turbulence, exchanges of energy and water, and the urban heat island. Int J Climatol 23(1):1–26. https://doi.org/10.1002/joc.859
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting Linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Beecham S, Lucke T (2015) Street Trees in Paved Urban Environments: The Benefits and Challenges. Proceeding of the 16th National Street Tree Symposium
Bigger DS, Marvier MA (1998) How different would a world without herbivory be? A search for generality in ecology. Integr Biology: Issues News Reviews 1(2):60–67. https://doi.org/10.1002/(sici)1520-6602(1998)1:2%3C60::aid-inbi4%3E3.0.co;2-z
Björkman C, Dalin P, Ahrné K (2008) Leaf trichome responses to herbivory in willows: induction, relaxation and costs. New Phytol 179(1):176–184. https://doi.org/10.1111/j.1469-8137.2008.02442.x
Blitzer EJ, Dormann CF, Holzschuh A, Klein A-M, Rand TA, Tscharntke T (2012) Spillover of functionally important organisms between managed and natural habitats. Agric Ecosyst Environ 146(1):34–43. https://doi.org/10.1016/j.agee.2011.09.005
Bode RF, Gilbert AB (2015) Seed predators, not herbivores, exert natural selection on Solidago spp. in an urban Archipelago. Environ Entomol 45(1):150–154. https://doi.org/10.1093/ee/nvv158
Bolund P, Hunhammar S (1999) Ecosystem services in urban areas. Ecol Econ 29(2):293–301. https://doi.org/10.1016/S0921-8009(99)00013-0
Brooks ME, Kristensen K, Van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9(2):378–400
Burghardt KT, Avolio ML, Locke DH, Grove JM, Sonti NF, Swan CM (2023) Current street tree communities reflect race-based housing policy and modern attempts to remedy environmental injustice. Ecology 104(2). https://doi.org/10.1002/ecy.3881
Campbell H (2021) The consequences of checking for zero-inflation and overdispersion in the analysis of count data. Methods Ecol Evol 12(4):665–680. https://doi.org/10.1111/2041-210x.13559
U.S. Census Bureau (2022) American Community Survey 5-year estimates (https://data.census.gov/map/
Chakraborty T, Hsu A, Manya D, Sheriff G (2019) Disproportionately higher exposure to urban heat in lower-income neighborhoods: a multi-city perspective. Environ Res Lett 14(10):105003. https://doi.org/10.1088/1748-9326/ab3b99
Cockfield SD, Potter DA (1986) Interaction of Euonymus scale (Homoptera: Diaspididae) feeding damage and severe water stress on leaf abscission and growth of Euonymus fortunei. Oecologia 71(1):41–46. https://doi.org/10.1007/BF00377318
Crawley MJ (1989) Insect herbivores and Plant Population dynamics. Ann Rev Entomol 34(1):531–562. https://doi.org/10.1146/annurev.en.34.010189.002531
Cuevas-Reyes P, Gilberti L, González-Rodríguez A, Fernandes GW (2013) Patterns of herbivory and fluctuating asymmetry in Solanum lycocarpum St. Hill (Solanaceae) along an urban gradient in Brazil. Ecol Ind 24:557–561. https://doi.org/10.1016/j.ecolind.2012.08.011
Dale AG, Frank SD (2014a) Urban warming trumps natural enemy regulation of herbivorous pests. Ecol Appl 24(7):1596–1607. https://doi.org/10.1890/13-1961.1
Dale AG, Frank SD (2014b) The effects of urban warming on Herbivore abundance and Street Tree Condition. PLoS ONE 9(7):e102996. https://doi.org/10.1371/journal.pone.0102996
Dale AG, Frank SD (2017) Warming and drought combine to increase pest insect fitness on urban trees. PLoS ONE 12(3):e0173844. https://doi.org/10.1371/journal.pone.0173844
Dewitz J, U.S. Geological Survey (2021) &. National Land Cover Database (NLCD) 2019 Products (ver. 3.0, February 2024) (https://doi.org/10.5066/P9KZCM54
Donovan GH, Butry DT (2010) Trees in the city: valuing street trees in Portland, Oregon. Landsc Urban Plann 94(2):77–83. https://doi.org/10.1016/j.landurbplan.2009.07.019
Dreistadt SH, Dahlsten DL, Frankie GW (1990) Urban Forests and Insect Ecology. Bioscience 40(3):192–198. https://doi.org/10.2307/1311364
Fenoglio MS, Calviño A, González E, Salvo A, Videla M (2021) Urbanisation drivers and underlying mechanisms of terrestrial insect diversity loss in cities. Ecol Entomol 46(4):757–771. https://doi.org/10.1111/een.13041
Freedman B (1995) 1 - THE ECOLOGICAL EFFECTS OF POLLUTION, DISTURBANCE, AND OTHER STRESSES. In B. Freedman (Ed.), Environmental Ecology (Second Edition) (pp. 1–10). Academic Press. https://doi.org/10.1016/B978-0-08-050577-0.50006-6
Getman-Pickering ZL, Campbell A, Aflitto N, Grele A, Davis JK, Ugine TA (2020) LeafByte: a mobile application that measures leaf area and herbivory quickly and accurately. Methods Ecol Evol 11(2):215–221. https://doi.org/10.1111/2041-210X.13340
Hanks LM, Denno RF (1993) Natural Enemies and Plant Water Relations Influence the Distribution of an armored scale insect. Ecology 74(4):1081–1091. https://doi.org/10.2307/1940478
Harrison S, Bruna E (1999) Habitat fragmentation and large-scale conservation: what do we know for sure? Ecography. 22(3):225–232
Haughton G, Hunter C (2004) Sustainable cities. Routledge
Herrmann DL, Pearse IS, Baty JH (2012) Drivers of specialist herbivore diversity across 10 cities. Landsc Urban Plann 108(2):123–130. https://doi.org/10.1016/j.landurbplan.2012.08.007
Hillier, Amy E.(2003) Redlining and the Home Owners’ Loan Corporation Journal of Urban History 29(4) 394-420 10.1177/0096144203029004002
Hilbert D, Roman L, Koeser A, Vogt J, Van Doorn N (2018) Urban Tree Mortality: A literature review. https://doi.org/10.13140/RG.2.2.25953.15204
Hoffman JS, Shandas V, Pendleton N (2020) The effects of historical housing policies on resident exposure to intra-urban heat: a study of 108 US urban areas. Climate 8(1):12
Hsu A, Sheriff G, Chakraborty T, Manya D (2021) Disproportionate exposure to urban heat island intensity across major US cities. Nat Commun 12(1):2721. https://doi.org/10.1038/s41467-021-22799-5
Kozlov MV, Zvereva EL (2018) Background Insect Herbivory: Impacts, Patterns and Methodology. In F. M. Cánovas, U. Lüttge, & R. Matyssek (Eds.), Progress in Botany Vol. 79 (pp. 313–355). Springer International Publishing. https://doi.org/10.1007/124_2017_4
Kozlov MV, Lanta V, x11b, ch, Zverev VE, Zvereva EL (2012) Delayed local responses of downy birch to damage by leafminers and leafrollers. Oikos, 121(3), 428–434. http://www.jstor.org/stable/41428987
Kozlov MV, Zverev V, Zvereva EL (2014) Confirmation bias leads to overestimation of losses of woody plant foliage to insect herbivores in tropical regions. PeerJ, 2, e709. https://doi.org/10.7717/peerj.709
Kozlov MV, Lanta V, Zverev V, Rainio K, Kunavin MA, Zvereva EL (2017) Decreased losses of woody plant foliage to insects in large urban areas are explained by bird predation. Glob Change Biol 23(10):4354–4364. https://doi.org/10.1111/gcb.13692
La Pierre KJ, Smith MD (2016) Soil nutrient additions increase invertebrate herbivore abundances, but not herbivory, across three grassland systems. Oecologia 180(2):485–497. https://doi.org/10.1007/s00442-015-3471-7
Landry SM, Chakraborty J (2009) Street Trees and Equity: evaluating the spatial distribution of an urban amenity. Environ Plann A: Econ Space 41(11):2651–2670. https://doi.org/10.1068/a41236
Marquis RJ, Whelan CJ (1994) Insectivorous birds increase growth of White Oak through Consumption of Leaf-Chewing insects. Ecology 75(7):2007–2014. https://doi.org/10.2307/1941605
Moreira X, Abdala-Roberts L, Mier y Teran B, Covelo JC, de la Mata F, Francisco R, Hardwick M, Pires B, Roslin RM, Schigel T, ten Hoopen DS, Timmermans JPJG, van Dijk BGH, Castagneyrol LJA, B., Tack AJM (2019) Impacts of urbanization on insect herbivory and plant defences in oak trees. Oikos 128(1):113–123. https://doi.org/10.1111/oik.05497
Mullaney J, Lucke T, Trueman SJ (2015) A review of benefits and challenges in growing street trees in paved urban environments. Landsc Urban Plann 134:157–166. https://doi.org/10.1016/j.landurbplan.2014.10.013
Mullu D (2016) A review on the effect of habitat fragmentation on ecosystem. J Nat Sci Res 6(15):1–15
Nabity PD, Zavala JA, Delucia EH (2009) Indirect suppression of photosynthesis on individual leaves by arthropod herbivory. Ann Botany 103(4):655–663. https://doi.org/10.1093/aob/mcn127
Nardone A, Rudolph KE, Morello-Frosch R, Casey JA (2021) Redlines and Greenspace: the relationship between historical redlining and 2010 Greenspace across the United States. Environ Health Perspect 129(1):017006. https://doi.org/10.1289/EHP7495
National Wildlife Federation (2021) Red Maple. https://www.nwf.org/Home/Educational-Resources/Wildlife-Guide/Plants-and-Fungi/Red-Maple
Nee S, May RM (1992) Dynamics of metapopulations: Habitat Destruction and competitive coexistence. J Anim Ecol 61(1):37–40. https://doi.org/10.2307/5506
Nikula S, Vapaavuori E, Manninen S (2010) Urbanization-related changes in European aspen (Populus tremula L.): Leaf traits and litter decomposition. Environ Pollut 158(6):2132–2142. https://doi.org/10.1016/j.envpol.2010.02.025
Oke TR, Maxwell GB (1975) Urban heat island dynamics in Montreal and Vancouver. Atmospheric Environment (1967), 9(2), 191–200. https://doi.org/10.1016/0004-6981(75)90067-0
Pataki DE, Alberti M, Cadenasso ML, Felson AJ, McDonnell MJ, Pincetl S, Pouyat RV, Setälä H, Whitlow TH (2021) The Benefits and Limits of Urban Tree Planting for Environmental and Human Health. Frontiers in Ecology and Evolution, 9. https://doi.org/10.3389/fevo.2021.603757
Payne EH, Gebregziabher M, Hardin JW, Ramakrishnan V, Egede LE (2018) An empirical approach to determine a threshold for assessing overdispersion in Poisson and negative binomial models for count data. Commun Stat Simul Comput 47(6):1722–1738. https://doi.org/10.1080/03610918.2017.1323223
Penone C, Kerbiriou C, Julien J-F, Julliard R, Machon N, Le Viol I (2013) Urbanisation effect on Orthoptera: which scale matters? Insect Conserv Divers 6(3):319–327. https://doi.org/10.1111/j.1752-4598.2012.00217.x
Price JC (1979) Assessment of the Urban Heat Island Effect through the Use of Satellite Data. Mon Weather Rev 107(11):1554–1557. https://doi.org/10.1175/1520-0493(1979)107%3C1554:AOTUHI%3E2.0.CO;2
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing. In https://www.R-project.org/
Raupp MJ, Shrewsbury PM, Herms DA (2010) Ecology of Herbivorous arthropods in Urban landscapes. Ann Rev Entomol 55(1):19–38. https://doi.org/10.1146/annurev-ento-112408-085351
Savi T, Bertuzzi S, Branca S, Tretiach M, Nardini A (2015) Drought-induced xylem cavitation and hydraulic deterioration: risk factors for urban trees under climate change? New Phytol 205(3):1106–1116. https://doi.org/10.1111/nph.13112
Schueller SK, Paul S, Payer N, Schultze R, Vikas M (2019) Urbanization decreases the extent and variety of leaf herbivory for native canopy tree species Quercus rubra, Quercus alba, and Acer saccharum. Urban Ecosyst 22(5):907–916. https://doi.org/10.1007/s11252-019-00866-6
Searle SY, Bitterman DS, Thomas S, Griffin KL, Atkin OK, Turnbull MH (2011) Respiratory alternative oxidase responds to both low- and high-temperature stress in Quercus rubra leaves along an urban–rural gradient in New York. Funct Ecol 25(5):1007–1017. https://doi.org/10.1111/j.1365-2435.2011.01875.x
Searle SY, Turnbull MH, Boelman NT, Schuster WSF, Yakir D, Griffin KL (2012) Urban environment of New York City promotes growth in northern red oak seedlings. Tree Physiol 32(4):389–400. https://doi.org/10.1093/treephys/tps027
Soares AL, Rego FC, McPherson EG, Simpson JR, Peper PJ, Xiao Q (2011) Benefits and costs of street trees in Lisbon, Portugal. Urban Forestry Urban Green 10(2):69–78. https://doi.org/10.1016/j.ufug.2010.12.001
Sperry JS, Tyree MT (1988) Mechanism of water stress-Induced Xylem Embolism 1. Plant Physiol 88(3):581–587. https://doi.org/10.1104/pp.88.3.581
The Herbivory Variability Network, Robinson ML, Hahn PG, Inouye BD, Underwood N, Whitehead SR, Abbott KC, Bruna EM, Cacho NI, Dyer LA, Abdala-Roberts L, Allen WJ, Andrade JF, Angulo DF, Anjos D, Anstett DN, Bagchi R, Bagchi S, Barbosa M, Wetzel WC (2023) Plant size, latitude, and phylogeny explain within-population variability in herbivory. Science 382(6671):679–683. https://doi.org/10.1126/science.adh8830
Turner-Skoff JB, Cavender N (2019) The benefits of trees for livable and sustainable communities. PLANTS PEOPLE PLANET 1(4):323–335. https://doi.org/10.1002/ppp3.39
Turrini T, Sanders D, Knop E (2016) Effects of urbanization on direct and indirect interactions in a tri-trophic system. Ecol Appl 26(3):664–675. https://doi.org/10.1890/14-1787
Walters RS, Yawney HW (1990) Acer rubrum L. Red Maple. Silvics North Am 2:60–69
Welter SC (1989) Arthropod Impact on Plant Gas Exchange. CRC, pp 135–164. https://doi.org/10.1201/9780429290916-5
Wilson MC, Chen X-Y, Corlett RT, Didham RK, Ding P, Holt RD, Holyoak M, Hu G, Hughes AC, Jiang L, Laurance WF, Liu J, Pimm SL, Robinson SK, Russo SE, Si X, Wilcove DS, Wu J, Yu M (2016) Habitat fragmentation and biodiversity conservation: key findings and future challenges. Landscape Ecol 31(2):219–227. https://doi.org/10.1007/s10980-015-0312-3
Wolf KL, Lam ST, McKeen JK, Richardson GRA, van den Bosch M, Bardekjian AC (2020) Urban Trees and Human Health: a scoping review. Int J Environ Res Public Health 17(12):4371. https://www.mdpi.com/1660-4601/17/12/4371
Young DS, Roemmele ES, Yeh P (2022) Zero-inflated modeling part I: traditional zero-inflated count regression models, their applications, and computational tools. WIRE Comput Stat 14(1):e1541. https://doi.org/10.1002/wics.1541
Zangerl AR, Hamilton JG, Miller TJ, Crofts AR, Oxborough K, Berenbaum MR, De Lucia EH (2002) Impact of folivory on photosynthesis is greater than the sum of its holes. Proc Natl Acad Sci 99(2):1088–1091. https://doi.org/10.1073/pnas.022647099
Zvereva EL, Zverev V, Kozlov MV (2012) Little strokes fell great oaks: minor but chronic herbivory substantially reduces birch growth. Oikos 121(12):2036–2043. http://www.jstor.org/stable/41686695
Acknowledgements
Funding was provided by the National Science Foundation Grant #NSF DBI 1950656 to A. Cawood and J. Parker for SERC’s Research Experience for Undergraduates program for E. Blake’s REU experience to conduct this research.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. EB, SB, and AH conducted the field research. EB, AH, and KK conducted the statistical analyses. EB wrote the first manuscript draft, with revisions from all authors. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Blake, E., Bennett, S., Hruska, A. et al. Insect herbivory on Acer rubrum varies across income and urbanization gradients in the D.C. metropolitan area. Urban Ecosyst (2024). https://doi.org/10.1007/s11252-024-01584-4
Accepted:
Published:
DOI: https://doi.org/10.1007/s11252-024-01584-4