Abstract
The fragmentation of habitats associated with urbanisation poses a significant threat to already vulnerable and endangered mammal species. While the county of Dorset has been identified as one of the UK’s biodiversity hotspots, it is characterised by large-scale urbanisation and intensive agriculture. Focusing on the largest urban area of Dorset—the conurbation of Bournemouth, Christchurch, and Poole—this study aims to investigate the importance of urban areas in supporting vulnerable and endangered mammals by comparing it to four other major land cover categories in the county: arable & horticulture, grassland, woodland, and heathland. The study utilises data collected by the county’s environmental records department through mainly volunteer-contributed ad-hoc sighting data of 10 872 georeferenced mammal records collected between 2000 and 2018. Our findings reveal that, despite having significantly different mammal communities to all other land covers, urban land cover was shown to host four out of five of the vulnerable and endangered species recorded in this study. These included the European rabbit (vulnerable on the IUCN red list), hazel dormouse (vulnerable on the UK red list), West European hedgehog (vulnerable on the UK red list) and European water vole (endangered on the UK red list). Our findings highlight the significance of urban areas as important habitats for mammal biodiversity, presenting an opportunity for the conservation of specific vulnerable and endangered species. Despite limitations in ad-hoc sighting data, our analysis indicates an overrepresentation of certain 'charismatic' species, like foxes and hedgehogs, in urban samples, likely due to their high intrinsic value to the population. While the recognition of urban habitats for 'red list' species is growing, it is also important to acknowledge the relative importance of charismatic species to urban human communities in conservation and management strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation is a complex socio-economic process of increasing population concentration in towns and cities (De Barros Ruas et al. 2022). In 2018, over half of the global population resided in urban areas, compared with the 30% recorded in 1950 (United Nations 2018). In the European Union, urbanisation has shown a constant increase between 1961 to 2018, with urban areas now home to around two thirds of its population (European Commission 2020). Such rapid expansion of urban areas is considered as one of the major threats to biodiversity worldwide, as growing cities consume habitats and disrupt regional ecological processes by impacting biogeochemical and energetic flows (Antrop 2004; Hansen et al. 2005; Grimm et al. 2008; Seto et al. 2011; Grabowski et al. 2023). This is likely because certain landscape features reflect key aspects of urbanisation—e.g. an increased number and density of buildings can alter native vegetation, introduce disturbances from humans and pets, and increase noise and light pollution. Roads can contribute to pollution from traffic and direct mortality from collisions (Van der Ree et al. 2009; Seress et al. 2014). It is expected that due to urbanisation, the extent of built-up areas in the world will triple between 2000 and 2050 (Liu et al. 2016), leading to further degradation or fragmentation of critical habitats into patches too small to support complex ecological communities (Thompson and Jones 1999; Czech et al. 2000).
This loss of natural habitats in rural areas can increases the risk of human-wildlife conflict (HWC; Soulsbury and White 2015; Basak et al. 2023). While some urban species have been shown to thrive in these fragmented urban environments due to the loss of predatory species, as well as increased food availability (often household waste; Baker and Harris 2007), HWC can have negative consequences for both humans and wildlife. These may include zoonotic disease transmission (Hegglin et al. 2015), wildlife-vehicle collisions (Schell et al. 2020), and attacks by wildlife on humans, their pets or domesticated animals (Marvin 2000; Mayer 2013; Ikeda et al. 2019), to name a few.
Conversely, existence of green areas in urban environments and the associated human-wildlife interactions can also yield positive outcomes, known as ecosystem services (Mace et al. 2012), which enhance the economic, social, and environmental value of these regions (Gómez-Baggethun and Barton 2013; Collins et al. 2017). Urban biodiversity serves as both an ecosystem process regulator and a valuable service in its own right (Mace et al. 2012). Urban green spaces like parks, canals, and ponds offer various benefits (Kabisch et al. 2015; Botzat et al. 2016; Biernacka et al. 2020), including improving education by immersing locals in nature and fostering environmental learning (Neill 1997), increasing property values in the area (Jim and Chen 2006), and enhancing residents' mental and physical well-being by promoting outdoor social interactions and physical activity (Smith et al. 1997; Maas et al. 2006). Because of this, the provision of ecosystem services depends upon the protection of biodiversity, and the concept of managing it for ecosystem services has been well discussed (Palumbi et al. 2009; Mori et al. 2017).
Although urban environments are not commonly considered as biodiversity hotspots, they have been shown to provide important habitats for certain endangered species (Alvey 2006). In Australia, 30% of threatened plant and animal species occur in cities (Ives et al. 2016). Similar findings have been reported in Europe, suggesting that even heavily urbanised areas can offer suitable habitats for endangered local flora and fauna (Jokimäki et al. 2018, Kowarik and von der Lippe 2018). For example, urban green areas in Sweden contained two thirds of the country’s red-listed species and were of particular importance to mammal species (Colding 2013).
Urban mammals, often labelled as commensal and synanthropic species, thrive in human landscapes, deriving benefits from human food and waste without relying solely on human-derived food (Baker and Harris 2007). However, the topic of urban mammal biodiversity has received relatively little research attention, especially when compared to other urban animal groups such as birds (Rega-Brodsky et al. 2022). Existing research on urban mammals has demonstrated that their populations can thrive in urban areas that have a good ecological management plan (Mahan and O’Connell 2005), and in some cases, the abundance of certain endangered mammal species have been shown to be higher in urban areas than natural habitats (Maclagan et al. 2018).
In Great Britain, 26% of terrestrial mammals are at risk of extinction, with urbanisation accounting for a greater impact on species than any other habitat conversion (Burns et al. 2016). However, studies focused on mammal diversity have shown that mammals in the UK are also able to occupy relatively novel environments. Britain’s road verges provide habitats to mice and voles (Bellamy et al. 2000), and residential gardens are utilised by bats, foxes, squirrels, hedgehogs, mice and voles (Baker and Harris 2007). Rivers and streams within parks, sports grounds, and urban reserves are important habitats for water voles in urban areas in the UK (Leivesley et al. 2021). Additionally, research on West European hedgehogs has shown that their numbers have declined markedly in the UK, but that they are still commonly present in areas of human habitation (Williams et al. 2018; Gazzard et al. 2021). The sustained construction of high-density, low-cost housing (Baker and Harris 2007) as part of the British Government’s recent affordable housing strategy (Rolnik 2019), is expected to continue to alter habitats and affect urban mammals in the UK, highlighting the importance of researching urban mammal diversity in the country. Therefore, the primary aim of this study is to assess the importance of urban areas in supporting endangered and vulnerable mammals in South-East Dorset, UK and to then identify measures which can be taken to protect any important species. By using volunteer-contributed sighting data for analysis, differences in mammal communities in urban (and suburban) regions compared to other land cover types will be identified, including those on the IUCN Red List and the UK Red List.
Methods
Study area
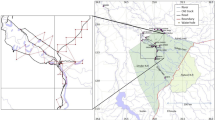
This study focuses on South-east Dorset (UK), home to Dorset's largest urban area—the unitary authority of Bournemouth, Christchurch, and Poole (BCP, pop. 400,300). Dorset is a predominantly rural maritime county (2,653 km2) in the south of England, with a population of approximately 780,000 people (ONS 2021 Census). The county hosts many diverse rural landscapes (Jiang et al. 2013), with 139 Sites of Special Scientific Interest, as designated by Natural England (Dorset Council 2022). It has been identified as one of the UK’s biodiversity hotspots (Prendergast et al. 1993) due to its internationally important wildlife species (Loth and Newton 2018). However, urbanisation and intensive agriculture have occurred on a large scale in Dorset (Hooftman and Bullock 2012; Jiang et al. 2013), causing a substantial decline in overall biodiversity in the area (Loth and Newton 2018). The unitary authority of Bournemouth, Christchurch and Poole has experienced an urbanisation rate of 5.7% between 2011 and 2021, comparatively higher than the rest of Dorset (4.0%) or e.g. the nearby city of Southampton (5.1%; ONS 2021 Census). Despite being highly urbanised, the BCP has been shown to provide important habitats to the red fox (Vulpes vulpes), where it hosts the highest concentration of this species for all urban areas in the UK (Scott et al. 2018). However, few studies have specifically focused on mammal biodiversity in urban areas in the county.
Biological data collection
Mammal records from 27 species were obtained by the Dorset Environmental Records Centre (DERC) and contained 11,400 georeferenced records in the study area (Fig. 1). Data was collected between 2000 to 2018 by 53 different local wildlife recording organisations and conservation organisations. Out of the 11,400, 528 records (4.6%) were discarded as they were not georeferenced. The vast majority of the records collected were ad-hoc citizen science records, with about 50% of the data coming from Dorset Records portal directly. Records from Peoples Trust for Endangered Species, Living Record and iRecord represented a further 30% of the survey. Some systematic surveys were also included in these records, such as the Cyril Diver project (see Carroll et al. 2023 for an overview), but these records made up only ~ 2% of the records and were based on surveys in heathlands and grasslands, and not urban areas. About 50% of the records were direct sightings of live animals, with around 20% being direct sightings of dead animals (mainly on the road). The remainder were indirect sightings, including bones in bird pellets, mole hills, scats and pellets. While the year of the record was provided, no details on time/month or associated weather conditions, which might influence observation were provided.
Data collection through citizen science recordings does create bias, with more sightings found in urban areas overall, and fewer sightings in other areas. Below, we explain how we overcome these biases in the data. However, citizen science records also have bias in what species participants choose to report. In this dataset, there were more records of hedgehogs and badgers than of the grey squirrel, and almost 7 times more recordings of hedgehogs and badgers than the black rat. Such biases in the data cannot be overcome but are discussed and interpreted in line with the overall aims of the study.
Geographical data collection
The collected data points were located within the ST, SU, SY and SZ grid references of the British National Grid (Ordnance Survey 2023). Subsequently, this area was split into smaller, 5 km x 5 km squares and only those squares where mammal records occurred were used in further analysis. The total size of the study area was 121,211 hectares. The initial 18 land cover classes were acquired from the 2020 UK Land cover map (UK Centre for Ecology and Hydrology 2021) and subsequently aggregated into five main land cover classes based on their similarity—arable and horticulture, grasslands (including improved grasslands, neutral grasslands, calcareous grasslands and acid grasslands), woodland (including broadleaved and coniferous woodland), urban and suburban (as a single category), heather (heather and heather grassland) and other (including all remaining UK LCM2020 land cover categories) (Table S1). Urban areas encompass densely populated zones, like town and city centres, often lacking vegetation, as well as areas like dock sides, car parks, and industrial estates. Suburban areas represent a blend of urban and vegetation spectral signatures, with suburban and urban zones forming a continuum (UKCEH 2021). Consequently, urban and suburban land covers are used as a single category in this paper. The UKCEH land cover classes are based on the UK Biodiversity Action Plan (BAP) Broad Habitats (Jackson 2000). The data on land cover category for each mammal record was derived using ArcMap 10.6 software and its Spatial Join function.
Statistical analysis
The majority of data used in this study was submitted ad-hoc by local wildlife recorders and conservation organisations. Such data has clear analytical challenges (as described by Robinson et al. 2018, and listed below). Data processing was used to try to eliminate as many of these data issues as possible. (1) Within each habitat the number of records varied markedly, and it is necessary to check that appropriate records exist to accurately estimate biodiversity within the habitat (i.e. sample size can affect estimates of diversity, as discussed in Rosenzweig 1995). To determine if sufficient records were available, rarefaction curves and sample completeness were calculated and visualised using the iNEXT package (Hsieh et al. 2016). (2) Community analysis such as PERMANOVA and Principal Coordinate Analysis (PCoA) require replicate samples from different habitats, as well as approximately equal sampling effort (i.e. similar numbers of records) in each area. This was not the case with the data obtained. To address this, we randomly selected 100 individual records from each habitat type to form a representative sample of the habitat, with probability of selection based on proportion of each species in the records for that habitat. We repeated this process 30 times for each habitat to create pseudo-replicates for analysis (following Hui et al. 2022). This process standardised the sample size between habitats, and also provided a proxy measure of the number of individuals seen (initial recordings were generally of presence of a species, not the number seen). Following this, multivariate analysis (PERMANOVA and Principal Coordinate Analysis—PCoA) was conducted using the Vegan package in R (Oksanen et al. 2008) on the replicated and standardised effort data. The use of visual ordination methods such as PCoA also normally calculate community structures based on relative abundance of key species, rather than being overly influenced by rare species, hence beginning to overcome the issue of presence only data (if rare species were not present, not recorded, or only recorded in small numbers, then they would be little difference to the overall community structure). However, it was not possible to overcome recorder bias for species (i.e. only species the observers consider to be of interest are submitted), and the implications of this are discussed later in the paper.
Results
A total of 11,400 georeferenced records from 27 species were obtained. The number of sightings was 1885 for arable and horticulture land, 3229 for grassland, 680 for heather, 3121 for urban and 1918 for woodland. The number of sightings varied greatly between species and land covers (Fig. 2), with urban land cover containing reported observations of west European hedgehogs, European rabbit, hazel dormouse and European water vole, which were listed as ‘vulnerable’ or ‘endangered’ (Table S3).
As the number of sightings was highly variable between land covers, rarefaction curves were plotted to assess sampling completeness between different land covers (Fig. 3). Sample completeness per land cover were found to be 100% for arable – horticulture, 100% for grassland, 99.7% for heather, 99.9% for urban and 99.9% for woodland, which indicates that the sampling effort was adequate to capture the majority of the species.
Species distribution in Bournemouth and its surrounding areas
A similar number of species per each land cover was found (Table S2, Fig. 3). However, in terms of community structure, PERMANOVA showed that there were significant differences between land covers (F5,174 = 78.58, p < 0.001) and pairwise comparisons showed that all land covers were significantly different from each other in terms of community structure (p < 0.001 in all cases). Despite all having different communities, some land covers were more closely related than others (Fig. 4). There were relatively close similarities in communities in the woodland, heather and ‘other’ land covers, as indicated by the direct overlap of coordinates. The grassland and arable horticulture land covers also showed some similarities in terms of mammal communities, as highlighted by the closeness (although not direct overlap) of coordinates. In contrast, the notable isolation of blue coordinates showed that the urban mammal communities were relatively different from all other land covers. The west European hedgehog and red fox were the differentiating species that were the most important in discriminating between urban the other land covers.
Vulnerable and endangered species
Five species in the present study were listed as ‘vulnerable’ or ‘endangered’ on either the Mammal Society UK red list or the IUCN red list. The west European hedgehog was listed as ‘vulnerable’ by the Mammal Society UK red list and was present in all land covers but had the highest number of observations in the urban land cover (Figs. 2 and 5A). The European rabbit was listed as ‘vulnerable’ on the global IUCN red list, but not on the Mammal Society UK red list and was present in all land covers, with a relatively similar number of observations across all (Figs. 2 and 5B). The hazel dormouse was listed as ‘vulnerable’ by the Mammal Society UK red list and was present in all land covers at very low density but had the highest number of observations in the woodland land cover (Figs. 2 and 5C). The European water vole was listed as ‘endangered’ by the Mammal Society UK red list and was present in all land covers, displaying no particular preference towards a certain land cover (Figs. 2 and 5D). The Eurasian red squirrel was listed as ‘endangered’ by the Mammal Society UK red list and was non-existent or very low in all land covers except woodland, where the number of observations were relatively high (Figs. 2. and 5E).
Discussion
Bias
There is inherent bias within most ad-hoc citizen science data collection which presents numerous difficulties for robust analysis (Robinson et al. 2018). Here, we have tried to address many issues, including those of rare species / presence only data and sample size (see methods) through the analysis techniques used. However, reporting bias (the greater reporting of charismatic species, especially in urban environments) was apparent in the data, with no available mechanism to remove this bias from the data.
In this study, there were clear signs of more recorded observations of species which may be viewed as rare, and less on others that may be viewed common (as discussed by Van Strien et al. (2013) and Lehtiniemi et al. (2020)). For example, there was a much larger number of reported west European hedgehogs (N = 1097), and very few brown rats (N = 71) or house mice (N = 13) in the urban setting. This is likely not because there were many more hedgehogs in the urban land cover, but instead because citizen scientists likely viewed data on these species as more important, hence reporting their sightings more often. Although this bias likely leads to an overestimation of total populations of urban west European hedgehogs, the results are still in agreement with other studies which show that urban environments represent a stronghold habitat for the west European hedgehogs in the UK (Gazzard and Baker 2020). Additionally, the fact that certain mammals were reported frequently in urban areas (e.g. west European hedgehogs), suggested that these mammals were considered as important and/or interesting by those reporting them. Therefore, it is possible that these mammals were valuable from an ecosystems services perspective (notably cultural and provisioning services), where they may add socio-economic value to urban areas (Gómez-Baggethun and Barton 2013; Collins et al. 2017). This relative importance of charismatic species in urban human communities should be recognised in future urban conservation and management strategies.
Specialist and generalist mammals
The urban land cover had a particularly large number of observations of west European hedgehog and red fox, which were shown to be mostly low in other land covers. This could be due to the generalist nature of both the west European hedgehog and the red fox (Young et al. 2006; Dell’Arte et al. 2007). Certain mammal generalists are able to utilise unoccupied ecological niches, allowing them to colonise fragmented or human-modified habitats that other specialists cannot (Schlinkert et al. 2016). In contrast, other land covers in this study were shown to support some potentially specialist species. In certain cases, some species only occupied one or two land covers, or were present in the other land covers, but in much lower numbers. Examples of this included hazel dormouse in the woodland land cover, as well as polecat ferret and brown hare in arable horticulture and grassland land covers. These findings suggested that the species occupy a niche in the habitats where they are present, which is not available in other habitats. Zhong et al. (2016) discussed the niche partitioning of specialist mammal species and highlighted that these species are the most at risk of habitat fragmentation, as unlike generalists, they are unable to colonise (and occupy alternative niches in) other, more human-modified land covers.
General management solutions to enrich mammal biodiversity in urban areas
The primary aim of this study was to assess the importance of urban areas in supporting endangered and vulnerable mammals in South-East Dorset, and to then identify measures which can be taken to protect any important species. We found that despite having relatively different communities compared to the other studied land covers, urban areas did support populations of several vulnerable or endangered species. Existing literature has also highlighted the importance of cities and urban areas in terms of distinct assemblages of threatened and/or protected mammals (Fernández and Simonetti 2013; Ives et al. 2016; Dietz et al. 2020). Four out of five of the vulnerable and endangered species recorded in this study were present in the urban setting. Despite the challenges associated with managing urban and suburban ecosystem services and the general decline in its associated biodiversity (Faeth et al. 2011; Ibáñez‐Álamo et al. 2017). Lambert and Donihue (2020) suggested that these ecosystems are far from being ecological lost causes and their management provides a unique opportunity for protecting biodiversity (Leivesley et al. 2021).
Multiple conservation management measures and policies may be considered to conserve urban populations of the vulnerable and endangered species identified in our study. Urban mammals such as hedgehogs could be better protected through improving garden connectivity, for example by creating ‘hedgehog roads’ (Gazzard and Baker 2020). Similarly, given that habitat fragmentation has been identified as one of the key threats to urban mammals (notably the hazel dormouse), the creation of structural connectivity through urban hedgerow networks has been shown as an important tool for protecting their populations (Mortelliti et al. 2011). The establishment of these vital green spaces is important for safeguarding populations of urban mammals, especially parks, sports areas (e.g. golf courses) and small ponds/reserves (Stewart et al. 2017; Leivesley et al. 2021). Green spaces which incorporate wide water bodies and tall diverse vegetation are especially beneficial and have been shown to provide a suitable foraging and burrowing habitats for European water voles (Richards et al. 2014), the fastest declining mammal in the UK due to habitat degradation (Barreto 1998; Strachan 2004; Richards et al. 2014). Additionally, urban mammal populations may be further protected with the introduction of public conservation campaigns (McLauchlan 2019), including those encouraging drivers to be vigilant for mammals on roads surrounded by a mix of urban and grassland cover, especially in the summer months (July – August) when mammals such as hedgehogs are most active (Wright et al. 2020).
Conservation of urban biodiversity must implement ‘systems thinking’, which encourages city planners and policy makers not to just focus on the individual discrete elements within urban management, but instead consider the many complex processes occurring in these ecosystems (Hostetler et al. 2011). The provision of many urban ecosystem services is reliant on the management of greenspaces (Gaston et al. 2013). Management of these urban ecosystem services can vary greatly in their objectives and may be conflicting in certain cases (Bullock et al. 2011), for example increasing urban carbon sequestration (through planting trees) may cause increased emissions of biogenic volatile organic compounds that are harmful to human health (Leung et al. 2010). Gaston et al. (2013) argued that managing ecosystem services presents a number of substantial challenges, including the structure of green space, its temporal dynamics and the spatial constraint on ecosystem service flows. Managing ecosystem services in urban areas can also be complicated due to the high number of land managers present. Aside from land owned by government and non-government organisations, urban areas are often managed by individual homeowners or tenants. Challenges can arise as a result of this because each individual may have conflicting goals and perceptions of desirable management strategies (Hofmann et al. 2012). Consequently, large-scale urban management which targets specific environmental issues and/or ecosystem services (for example maximising green space flood mitigation or organic carbon storage potential) can be difficult to achieve (Gaston et al. 2013). Despite this, examples of multifunctional urban nature-based solutions providing biodiversity, climate adaptation and mitigation benefits alongside other cultural ecosystem services are becoming more common (Cadotte et al. 2021; Stafford et al. 2021).
Mammal populations can thrive in urban areas which have a good ecological management plan (Mahan and O’Connell 2005). Urban spaces with ‘manicured habitats’ that are surrounded by human-modified landscapes supported low mammal species richness and diversity however well managed urban areas, with parks can support mammal biodiversity as high as (and in some cases exceed) mature riparian forests (Mahan and O’Connell 2005). The utilisation of these urban green spaces by mammal species depends upon multiple factors, including quality of the urban habitats, the availability of natural and artificial food sources and the proximity (and tolerance of this proximity) to humans (Baker and Harris 2007). Mammal dynamics vary greatly within urban areas, with substantially dissimilar communities in relatively close proximities (Gallo et al. 2017). This is likely because most mammals are limited in terms of mobility and are frequently exposed to physical barriers (such as buildings, drainage systems, railway lines and roads) which reduce their ability to move between green spaces (Ordeñana et al. 2010).
To further protect urban biodiversity, a transdisciplinary framework should be used along with removing policies that hinder conservation (Gordon et al. 2009; Gaston et al. 2013; Hostetler et al. 2011). For instance, stormwater policies can be replaced with low-impact designs like rain gardens and bioswales. Incentives for using native plants and sustainable construction techniques should be offered. Ecological tools like remote sensing, landscape ecology, and ecosystem services modelling can also help (Schell et al. 2020; Gaston et al. 2013; Crossman et al. 2013; Grafius et al. 2016).
Conclusion
Our results highlighted that four out of five of the vulnerable and endangered species recorded were present in the urban settings, including the European rabbit (vulnerable on the IUCN red list), hazel dormouse (vulnerable on the UK red list), west European hedgehog (vulnerable on the UK red list) and European water vole (endangered on the UK red list). These findings suggest that urban areas can be important for supporting mammal biodiversity and highlight a unique opportunity to conserve populations of certain vulnerable and endangered species. Further research should assess particular integrated conservation tools and strategies which can be utilised by urban planners to support populations of vulnerable and endangered mammals on a county and nation-wide scale.
Data availability
The data used in the present study is available upon request from the Dorset Environmental Records Centre (https://derc.org.uk).
References
Alvey AA (2006) Promoting and preserving biodiversity in the urban forest. Urban For Urban Green 5(4):195–201
Antrop M (2004) Landscape change and the urbanization process in Europe. Landsc Urban Plan 67(1–4):9–26
Baker PJ, Harris S (2007) Urban mammals: What does the future hold? An analysis of the factors affecting patterns of use of residential gardens in Great Britain. Mammal Rev 37(4):297–315
Barreto GR (1998) The tightrope hypothesis: An explanation for plummeting water vole numbers in the Thames catchment. United Kingdom Floodplains 311–327
Basak SM, Rostovskaya E, Birks J, Wierzbowska IA (2023) Perceptions and attitudes to understand human-wildlife conflict in an urban landscape–A systematic review. Ecol Ind 151:110319
Bellamy PE, Shore RF, Ardeshir D, Treweek JR, Sparks TH (2000) Road verges as habitat for small mammals in Britain. Mammal Rev 30(2):131–139
Biernacka M, Kronenberg J, Łaszkiewicz E (2020) An integrated system of monitoring the availability, accessibility and attractiveness of urban parks and green squares. Appl Geogr 116:102152
Botzat A, Fischer LK, Kowarik I (2016) Unexploited opportunities in understanding liveable and biodiverse cities. A review on urban biodiversity perception and valuation. Glob Environ Chang 39:220–233
Bullock JM, Aronson J, Newton AC, Pywell RF, Rey-Benayas JM (2011) Restoration of ecosystem services and biodiversity: Conflicts and opportunities. Trends Ecol Evol 26(10):541–549
Burns F, Eaton MA, Barlow KE, Beckmann BC, Brereton T, Brooks DR, Brown PMJ, Al Fulaij N, Gent T, Henderson I, Noble DG, Parsons M, Powney GD, Roy HE, Stroh P, Walker K, Wilkinson JW, Wotton SR, Gregory RD (2016) Agricultural management and climatic change are the major drivers of biodiversity change in the UK. PLoS ONE 11(3):e0151595
Cadotte MW, Potgieter LJ, Wang CJ, MacIvor JS (2021) Invasion theory as a management tool for increasing native biodiversity in urban ecosystems. J Appl Ecol 58:2394–2403
Carroll T, Stafford R, Gillingham PK, Bullock JM, Brown D, Brown M, Walls RM, Diaz A (2023) Correlated biodiversity change between plant and insect assemblages resurveyed after 80 years across a dynamic habitat mosaic. Ecol Evol 13(6):e10168
Colding J (2013) Local assessment of Stockholm: Revisiting the Stockholm urban assessment. Urbanization, biodiversity and ecosystem services: Challenges and opportunities. Springer, Dordrecht, pp 313–335
Collins R, Schaafsma M, Hudson MD (2017) The value of green walls to urban biodiversity. Land Use Policy 64:114–123
Crossman ND, Burkhard B, Nedkov S, Willemen L, Petz K, Palomo I, Drakou EG, Martín-Lopez B, McPhearson T, Boyanova K (2013) A blueprint for mapping and modelling ecosystem services. Ecosyst Serv 4:4–14
Czech B, Krausman PR, Devers PK (2000) Economic associations among causes of species endangerment in the United States: Associations among causes of species endangerment in the United States reflect the integration of economic sectors, supporting the theory and evidence that economic growth pr. Bioscience 50(7):593–601
De Barros Ruas R, Costa LM, Bered F (2022) Urbanization driving changes in plant species and communities – A global view. Global Ecology and Conservation 38:e02243
Dell’Arte GL, Laaksonen T, Norrdahl K, Korpimäki E (2007) Variation in the diet composition of a generalist predator, the red fox, in relation to season and density of main prey. Acta Oecologica 31(3):276–281
Dietz M, Bögelsack K, Krannich A, Simon O (2020) Woodland fragments in urban landscapes are important bat areas: An example of the endangered Bechstein’s bat Myotis bechsteinii. Urban Ecosystems 23(6):1359–1370
Dorset Council (2022) Sites of scientific interest [online]. Available from: https://www.dorsetcouncil.gov.uk/countryside-coast-parks/countryside-management/countryside-site-designations/sites-of-special-scientific-interest#:~:text=The%20national%20wildlife%20and%20geological,currently%20139%20SSSIs%20in%20Dorset
European Commission (2020) Urbanisation in Europe. https://knowledge4policy.ec.europa.eu/foresight/topic/continuing-urbanisation/developments-and-forecasts-on-continuing-urbanisation_en
Faeth SH, Bang C, Saari S (2011) Urban biodiversity: Patterns and mechanisms. Ann N Y Acad Sci 1223(1):69–81
Fernández IC, Simonetti JA (2013) Small mammal assemblages in fragmented shrublands of urban areas of Central Chile. Urban Ecosyst 16(2):377–387
Gallo T, Fidino M, Lehrer EW, Magle SB (2017) Mammal diversity and metacommunity dynamics in urban green spaces: Implications for urban wildlife conservation. Ecol Appl 27(8):2330–2341
Gaston KJ, Ávila-Jiménez ML, Edmondson JL (2013) Managing urban ecosystems for goods and services. J Appl Ecol 50(4):830–840
Gazzard A, Baker PJ (2020) Patterns of feeding by householders affect activity of hedgehogs (Erinaceus europaeus) during the hibernation period. Animals 10(8):1344
Gazzard A, Boushall A, Brand E, Baker PJ (2021) An assessment of a conservation strategy to increase garden connectivity for hedgehogs that requires cooperation between immediate neighbours: A barrier too far? PLoS ONE 16(11):e0259537
Grabowski Z, Fairbairn AJ, Teixeira LH, Micklewright J, Fakirova E, Adeleke E, Meyer ST, Traidl-Hoffman C, Schloter M, Helmreich B (2023) Cosmopolitan conservation: The multi-scalar contributions of urban green infrastructure to biodiversity protection. Biodivers Conserv 32:3595–3606
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the ecology of cities. Science 319(5864):756–760
Gómez-Baggethun E, Barton DN (2013) Classifying and valuing ecosystem services for urban planning. Ecol Econ 86:235–245
Gordon A, Simondson D, White M, Moilanen A, Bekessy SA (2009) Integrating conservation planning and land use planning in urban landscapes. Landsc Urban Plan 91(4):183–194
Grafius DR, Corstanje R, Warren PH, Evans KL, Hancock S, Harris JA (2016) The impact of land use/land cover scale on modelling urban ecosystem services. Landscape Ecol 31(7):1509–1522
Hansen AJ, Knight RL, Marzluff JM, Powell S, Brown K, Gude PH, Jones K (2005) Effects of exurban development on biodiversity: Patterns, mechanisms, and research needs. Ecol Appl 15(6):1893–1905
Hegglin D, Bontadina F, Deplazes P (2015) Human–wildlife interactions and zoonotic transmission of Echinococcus multilocularis. Trends Parasitol 31(5):167–173
Hofmann M, Westermann JR, Kowarik I, Van der Meer E (2012) Perceptions of parks and urban derelict land by landscape planners and residents. Urban For Urban Green 11(3):303–312
Hooftman DAP, Bullock JM (2012) Mapping to inform conservation: A case study of changes in semi-natural habitats and their connectivity over 70 years. Biol Cons 145(1):30–38
Hostetler M, Allen W, Meurk C (2011) Conserving urban biodiversity? Creating green infrastructure is only the first step. Landsc Urban Plan 100(4):369–371
Hsieh TC, Ma KH, Chao A (2016) iNEXT: An R package for rarefaction and extrapolation of species diversity (Hill numbers). Methods Ecol Evol 7:1451–1456
Hui E, Stafford R, Matthews IM, Smith VA (2022) Bayesian Networks as a novel tool to enhance interpretability and predictive power of ecological models. Ecol Inform 68:101539
Ibáñez-Álamo JD, Rubio E, Benedetti Y, Morelli F (2017) Global loss of avian evolutionary uniqueness in urban areas. Glob Change Biol 23(8):2990–2998
Ikeda T, Kuninaga N, Suzuki T, Ikushima S, Suzuki M (2019) Tourist-wild boar (Sus scrofa) interactions in urban wildlife management. Global Ecology and Conservation 18:e00617
Ives CD, Lentini PE, Threlfall CG, Ikin K, Shanahan DF, Garrard GE, Bekessy SA, Fuller RA, Mumaw L, Rayner L (2016) Cities are hotspots for threatened species. Glob Ecol Biogeogr 25(1):117–126
Jackson DL (2000) JNCC Report No. 307. Guidance on the interpretation of the Biodiversity Broad habitat Classification (terrestrial and freshwater types): Definitions and the relationships with other habitat classifications. Joint Nature Conservation Committee, Peterborough
Jiang M, Bullock JM, Hooftman DAP (2013) Mapping ecosystem service and biodiversity changes over 70 years in a rural E nglish county. J Appl Ecol 50(4):841–850
Jim CY, Chen WY (2006) Impacts of urban environmental elements on residential housing prices in Guangzhou (China). Landsc Urban Plan 78(4):422–434
Jokimäki J, Suhonen J, Kaisanlahti-Jokimäki M-L (2018) Urban core areas are important for species conservation: A European-level analysis of breeding bird species. Landsc Urban Plan 178:73–81
Kabisch N, Qureshi S, Haase D (2015) Human–environment interactions in urban green spaces—A systematic review of contemporary issues and prospects for future research. Environ Impact Assess Rev 50:25–34
Kowarik I, von der Lippe M (2018) Plant population success across urban ecosystems: A framework to inform biodiversity conservation in cities. J Appl Ecol 55(5):2354–2361
Lambert MR, Donihue CM (2020) Urban biodiversity management using evolutionary tools. Nat Ecol Evol 4(7):903–910
Lehtiniemi M, Outinen O, Puntila-Dodd R (2020) Citizen science provides added value in the monitoring for coastal non-indigenous species. J Environ Manag 267:110608
Leivesley JA, Stewart RA, Paterson V, McCafferty DJ (2021) Potential importance of urban areas for water voles: Arvicola amphibius. Eur J Wildl Res 67(1):1–4
Leung DYC, Wong P, Cheung BKH, Guenther A (2010) Improved land cover and emission factors for modeling biogenic volatile organic compounds emissions from Hong Kong. Atmos Environ 44(11):1456–1468
Liu Z, He C, Wu J (2016) The relationship between habitat loss and fragmentation during urbanization: An empirical evaluation from 16 world cities. PLoS ONE 11(4):e0154613
Loth AF, Newton AC (2018) Rewilding as a restoration strategy for lowland agricultural landscapes: Stakeholder-assisted multi-criteria analysis in Dorset, UK. J Nat Conserv 46:110–120
Maas J, Verheij RA, Groenewegen PP, De Vries S, Spreeuwenberg P (2006) Green space, urbanity, and health: How strong is the relation? J Epidemiol Commun Health 60(7):587–592
Mace GM, Norris K, Fitter AH (2012) Biodiversity and ecosystem services: A multilayered relationship. Trends Ecol Evol 27(1):19–26
Maclagan SJ, Coates T, Ritchie EG (2018) Don’t judge habitat on its novelty: Assessing the value of novel habitats for an endangered mammal in a peri-urban landscape. Biol Cons 223:11–18
Mahan CG, O’Connell TJ (2005) Small mammal use of suburban and urban parks in central Pennsylvania. Northeast Nat 12(3):307–314
Marvin G (2000) The problem of foxes: Legitimate and illegitimate killing. Natural enemies: People-wildlife conflicts in anthropological perspective
Mathews F, Harrower C (2020) IUCN–compliant Red List for Britain’s Terrestrial Mammals. Assessment by the Mammal Society under contract to Natural England, Natural Resources Wales and Scottish Natural Heritage
Mayer JJ (2013) Wild pig attacks on humans. Proceedings of the 15th Wildlife Damage Management Conference. (J. B. Armstrong, G. R. Gallagher, Eds)
McLauchlan L (2019) Wild disciplines and multispecies erotics: on the power of wanting like a hedgehog champion. Aust Fem Stud 34(102):509–523
Mori AS, Lertzman KP, Gustafsson L (2017) Biodiversity and ecosystem services in forest ecosystems: A research agenda for applied forest ecology. J Appl Ecol 54(1):12–27
Mortelliti A, Amori G, Capizzi D, Cervone C, Fagiani S, Pollini B, Boitani L (2011) Independent effects of habitat loss, habitat fragmentation and structural connectivity on the distribution of two arboreal rodents. J Appl Ecol 48(1):153–162
Neill JT (1997) Outdoor Education in the Schools: What can it achieve? 10th National Outdoor Education Conference. Australia, Sydney, pp 1–6
Oksanen J, Kindt R, Legendre P, O'Hara B, Simpson GL, Solymos P, Stevens MHH, Wagner H (2008) Vegan: Community ecology package (R package version 1.15–1)
Ordeñana MA, Crooks KR, Boydston EE, Fisher RN, Lyren LM, Siudyla S, Haas CD, Harris S, Hathaway SA, Turschak GM (2010) Effects of urbanization on carnivore species distribution and richness. J Mammal 91(6):1322–1331
Ordnance Survey (2023) Using the National Grid. Available online at: https://www.ordnancesurvey.co.uk/documents/resources/guide-to-nationalgrid.pdf
Palumbi SR, Sandifer PA, Allan JD, Beck MW, Fautin DG, Fogarty MJ, Halpern BS, Incze LS, Leong J-A, Norse E (2009) Managing for ocean biodiversity to sustain marine ecosystem services. Front Ecol Environ 7(4):204–211
Prendergast JR, Quinn RM, Lawton JH, Eversham BC, Gibbons DW (1993) Rare species, the coincidence of diversity hotspots and conservation strategies. Nature 365(6444):335–337
Rega-Brodsky CC, Aronson MFJ, Piana MR, Carpenter E, Hahs AK, Herrera-Montes A, Knapp S, Kotze DJ, Lepczyk CA, Moretti M, Salisbury AB, Williams NSG, Jung K, Katti M, MacGregor-Fors I, La MacIvor JS, Sorte FA, Sheel V, Threfall CG, Nilon CH (2022) Urban biodiversity: State of the science and future directions. Urban Ecosyst 25:1083–1096
Richards DR, Maltby L, Moggridge HL, Warren PH (2014) European water voles in a reconnected lowland river floodplain: Habitat preferences and distribution patterns following the restoration of flooding. Wetlands Ecol Manag 22(5):539–549
Robinson OJ, Ruiz-Gutierrez V, Fink D (2018) Correcting for bias in distribution modelling for rare species using citizen science data. Divers Distrib 24(4):460–472
Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, Cambridge
Rolnik R (2019) Urban warfare: Housing under the empire of finance. Verso Books
Schell CJ, Stanton LA, Young JK, Angeloni LM, Lambert JE, Breck SW, Murray MH (2020) The evolutionary consequences of human–wildlife conflict in cities. Evol Appl 14(1):178–197
Schlinkert H, Ludwig M, Batáry P, Holzschuh A, Kovács-Hostyánszki A, Tscharntke T, Fischer C (2016) Forest specialist and generalist small mammals in forest edges and hedges. Wildl Biol 22(3):86–94
Scott DM, Baker R, Charman N, Karlsson H, Yarnell RW et al (2018) A citizen science based survey method for estimating the density of urban carnivores. PLoS ONE 13(5):e0197445
Seress G, Lipovits A, Bokony V, Czuny L (2014) Quantifying the urban gradient: A practical method for broad measurements. Landsc Urban Plan 131:42–50
Seto KC, Fragkias M, Güneralp B, Reilly MK (2011) A meta-analysis of global urban land expansion. PLoS ONE 6(8):e23777
Smith T, Nelischer M, Perkins N (1997) Quality of an urban community: A framework for understanding the relationship between quality and physical form. Landsc Urban Plan 39(2–3):229–241
Soulsbury CD, White PCL (2015) Human–wildlife interactions in urban areas: A review of conflicts, benefits and opportunities. Wildl Res 42(7):541–553
Stafford R, Chamberlain B, Clavey L, Gillingham PK, McKain S, Morecroft MD, Morrison-Bell C, Watts O (2021) Nature-based solutions for climate change in the UK. Available from: https://www.britishecologicalsociety.org//wp-content/uploads/2022/02/NbS-Report-Final-Updated-Feb-2022.pdf
Stewart RA, Clark TJ, Shelton J, Stringfellow M, Scott C, White SA, McCafferty DJ (2017) Urban grasslands support threatened water voles. J Urban Ecol 3(1):jux007
Strachan R (2004) Conserving water voles: Britain’s fastest declining mammal. Water Environ J 18(1):1–4
Thompson K, Jones A (1999) Human population density and prediction of local plant extinction in Britain. Conserv Biol 13(1):185–189
UK Centre for Ecology and Hydrology (2021) UKCEH Land Cover Maps. Available from: https://www.ceh.ac.uk/data/ukceh-land-cover-maps
United Nations (2018) Department of Economic and Social Affairs, P.D. The World ’s Cities in 2018. The World’s Cities in 2018 - Data Booklet (ST/ESA/ SER.A/417) 34. https://digitallibrary.un.org/record/3799524
Van Strien AJ, Van Swaay CAM, Termaat T (2013) Opportunistic citizen science data of animal species produce reliable estimates of distribution trends if analysed with occupancy models. J Appl Ecol 50(6):1450–1458
Van der Ree R, Heinze D, McCarthy M, Mansergh I (2009) Wildlife tunnel enhances population viability. Ecol Soc 14(2)
Williams B, Mann N, Neumann JL, Yarnell RW, Baker PJ (2018) A prickly problem: Developing a volunteer-friendly tool for monitoring populations of a terrestrial urban mammal, the West European hedgehog (Erinaceus europaeus). Urban Ecosyst 21:1075–1086
Wright PGR, Coomber FG, Bellamy CC, Perkins SE, Mathews F (2020) Predicting hedgehog mortality risks on British roads using habitat suitability modelling. PeerJ 7:e8154
Young RP, Davison J, Trewby ID, Wilson GJ, Delahay RJ, Doncaster CP (2006) Abundance of hedgehogs (Erinaceus europaeus) in relation to the density and distribution of badgers (Meles meles). J Zool 269(3):349–356
Zhong W, Wang G, Zhou Q, Ma L, Wan X, Liu W (2016) Spatial niche partitioning of coexisting small mammals in sand dunes. Ital J Zvool 83(2):248–254
Acknowledgements
The authors are very grateful to DERC for supplying and managing the data analysed in the present study. The authors would also like to thank all the local wildlife recorders and conservation organisations which collected the data. The authors are also very grateful to the editor and reviewers who helps improving the manuscript.
Author information
Authors and Affiliations
Contributions
EAH and MC conceived the study. Data analysis was carried out by ZB, IB, RS, EAH and MC. The manuscript was written by ZB. All authors commented on previous versions of the manuscript, read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Boakes, Z., Stafford, R., Bramer, I. et al. The importance of urban areas in supporting vulnerable and endangered mammals. Urban Ecosyst 27, 883–894 (2024). https://doi.org/10.1007/s11252-023-01492-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-023-01492-z