Abstract
Roost locations can be an important contributor to vulture conflicts with humans, but factors influencing roost-site selection at a landscape level remain largely unexplored. Further, there has been little research comparing how these factors vary between nocturnal and diurnal roosting sites. We used remote cameras to document daily variation in vulture use of 21 roosts (20 communication/water towers and 1 natural roost) near Beaufort, South Carolina, USA from October 2019–August 2020. Numbers of vultures on roosts increased with decreasing urban cover and with greater distance to water, but were not influenced by habitat fragmentation or elevation. Roosts surrounded by greater proportions of urban cover were used more often during the day, whereas roosts adjacent to less urban cover were more commonly used at night. We suggest that this relationship results from a greater daytime association with human development, areas that likely provide food and favorable soaring conditions for vultures. Vultures tended to depart nocturnal roosts before sunrise and return within two hours of sunset, indicating that aircraft collision risk resulting from movement around nocturnal roosts would be elevated during these times. Several communication towers routinely had > 100 vultures roosting on them at once, likely contributing to conflict with humans. Our findings reiterate the generalism of these species and their capacity to exploit novel structures for roosting, which has likely contributed to range expansions and resultant increases in human-vulture conflicts over the past several decades.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthropogenic development has produced novel ecosystems, the components of which are markedly different from ecosystems of the past (Hobbs et al. 2009). Although such changes have engendered the decline of numerous avian species, some birds thrive in novel ecosystems due to the presence of resources including food and habitat (Bonier et al. 2007). Novel ecosystems are not uniform, however, and birds may interact with them in complex and nuanced ways. Rainbow lorikeets (Trichoglossus hameatodus) in urban areas, for example, select groups of trees with certain configurations for roosting while avoiding roosting in solitary trees (Jaggard et al. 2015). In agricultural landscapes, grey partridges (Perdix perdix) shift habitat selection throughout the course of the day to avoid encountering people (Harmange et al. 2021). Understanding how animals interact with human development is important for predicting and mitigating human-wildlife conflicts, which are common in areas where wildlife and people coexist (Nyhus 2016).
Black vultures (Coragyps atratus) and turkey vultures (Cathartes aura) are often associated with human development; both species have increased in abundance while expanding their geographic ranges across North America in recent decades (Kiff 2000; Sauer et al. 2017). In places where human development exists, black and turkey vultures benefit from increased food availability, as well as favorable flight and roosting conditions (Novaes and Cintra 2013; Novoselova et al. 2020; Hill et al. 2021, 2022). However, the success of these species in areas where humans are present has led to increases in human-vulture conflicts such as property damage and human health concerns (Lowney 1999; Kluever et al. 2020). At night when they are inactive, vultures frequently form communal roosts that may exceed several hundred individuals (Sweeney and Fraser 1986; Buckley et al. 2022). During the day, vultures perch at diurnal resting sites or loafing areas (hereafter diurnal roosts) when they are not foraging (Tillman et al. 2002; Holland et al. 2019). A substantial portion of property damage by vultures occurs in the vicinity of roosts (Tillman et al. 2002; Kluever et al. 2020). The convergence of many animals in a single location also leads to the accumulation of pellets and feces, along with associated odors, which is another source of conflict with humans (Lowney 1999; Tillman et al. 2002).
In addition to these conflicts, black and turkey vultures are also commonly involved in aircraft collisions, with both species considered among the avian species most likely to cause substantial damages to aircraft (DeVault et al. 2018; Pfeiffer et al. 2018). Between 2010 and 2019, damages to civilian and military aircraft resulting from collisions with black vultures exceeded $120 million USD (Kluever et al. 2020). Collisions with both species are also likely to increase in frequency considering their expanding ranges (Kirk and Mossman 2020; Kluever et al. 2020). Most strategies for mitigating aircraft collisions with birds have focused on management of the airport environment based on the assumption that collisions are most likely to occur < 152 m above ground level and current tools have limited reach (Dolbeer 2011; Pfeiffer et al. 2021). For vultures, however, many strikes occur at high altitudes (Arrondo et al. 2021), diminishing the effectiveness of on-airport management. Therefore, an important component of vulture strike management is to determine where large aggregations of vultures, such as roosts, occur and disperse these when necessary to reduce the risk of vulture collisions with aircraft (Walter et al. 2012).
Roost-site selection of black and turkey vultures is influenced by air currents that aid in flight, as well as food availability (Novaes and Cintra 2015; KC et al. 2019). Landscape attributes influence the occurrence of these resources and in turn impact roost-site selection by vultures (Hill et al. 2021). Vultures may roost in urban areas where they forage on trash and roadkill and use thermal currents from impervious surface cover for flight (Novaes and Cintra 2013; Campbell 2014). The contrasting surface temperatures of fragmented landscapes produce strong thermal currents that can serve as an attractant for roosting (Mallon et al. 2016; Novoselova et al. 2020). Roosts at greater elevations may be selected because they create greater uplift, which aids in flight (Thompson et al. 1990). Additionally, roosts often occur near water sources because they offer prospects for drinking and bathing (Thompson et al. 1990; Evans 2013).
Despite the growing prevalence of black and turkey vultures in human-modified areas, information regarding the influence of landscape attributes on roosting sites is limited. Most studies have focused on roosting in areas with either very high (e.g., Novaes and Cintra 2015) or very low (e.g., Holland et al. 2019) amounts of human development, whereas comparatively little work has been done in areas that contain a mix of developed and natural areas. Additionally, most research has focused on factors contributing to nighttime roost site selection, with relatively little work investigating daytime roost selection. As such, it remains unclear whether landscape attributes have the same influence on vulture selection of these two types of sites. This information is especially important for management because differential use of roost sites throughout the day could create a temporally dynamic pattern of aircraft collision risk (Walter et al. 2012).
We placed remote cameras on vulture roosts in and around Beaufort, South Carolina, USA to compare the role of landscape attributes on nocturnal and diurnal roosting site selection. Specifically, our aims were to (1) assess how natural and anthropogenic landscape features affect the spatial distribution and abundance of vultures at roosts, and (2) determine whether daily roost use by vultures varies temporally and is affected by urbanization.
Methods
Field methods
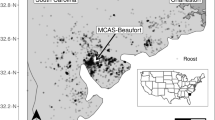
We conducted this study near Marine Corps Air Station Beaufort (MCAS), located in Beaufort County in the low country salt marsh region of South Carolina, USA (32.418°N, 80.640°W, Fig. 1). The population of Beaufort County is approximately 186,000, but tourism is a major part of the local economy, with the annual number of visitors exceeding 3 million (Carey and Salazar 2017; US Census Bureau 2021). The coastline of this area is characterized by a chain of land masses known as Sea Islands, separated from the mainland by marshes and tidal creeks (Zeigler 1959). Mean annual temperature is 19.6 °C and mean annual precipitation is 121.51 cm (NOAA 2021). Predominate land cover types in the area include open water (33%), wetlands (33%), forest (21%), developed (9%), and agriculture (3%). The area has a year-round population of turkey vultures and black vultures (Walter et al. 2012).
We located roosts by driving roads within the study area at dusk and looking for vultures returning to roosts (e.g., groups of low flying vultures flying back and forth to roost). We aimed to carry out the study over a complete year (September 2019-August 2020). However, we faced delays receiving permission from landowners or organizations that owned the land where the roost existed to place cameras. These delays resulted in a staggered entry of roosts into our study which included 1 roost in October, 2 in November, 15 in December, and 21 during January–August 2020.
At each roost, we mounted two wildlife cameras (Bushnell Trophy Cam HD Aggressor, Reconyx model RC60, or Reconyx model PC900) from opposite vantage points to maximize vulture detection. Cameras were mounted to trees, fenceposts, or utility poles depending on availability of structures providing adequate views of the roost. Cameras were positioned so that as much of the roost as possible was within the field of view with the sky in the background to facilitate counts. We programmed cameras to take images daily at half hour intervals beginning at 0500 h and ending at 1930 h (30 images per day); the two cameras at each roost took images simultaneously. We counted the number of vultures perched on roosts in all images. From each pair of images (i.e., from the two cameras at a roost capturing images simultaneously), we used the higher vulture count for analysis. Because it was not possible to distinguish between vulture species from the images, we also periodically performed physical counts at all roosts to better understand species composition. We did not observe vultures flushing from roosts as a result of our presence during physical counts.
Analysis
Spatial variation in vulture counts. Our first objective was to assess how natural and anthropogenic landscape features affect the spatial distribution and abundance of vultures. To address this, we modeled number of vultures at a roost as a function of landscape covariates. We calculated the daily maximum total vultures observed from images at each monitored roost location as our response variable. We considered four landscape-level covariates for each roost location: distance to water, elevation, percent urban land use in the area, and the number of unique habitat types around the roost (i.e., habitat richness, a proxy for landscape fragmentation) (Hill et al. 2021). We obtained distance to nearest water (lake or river) from TIGER/Line spatial data (US Census Bureau 2019) and elevation from a digital elevation model downloaded from Amazon Terrain Tiles (Amazon 2017). We obtained land cover data at 30 m resolution from the 2016 National Land Cover Database (NLCD; Yang et al. 2018) and calculated percent urban cover (NLCD classes 23–24) and number of unique land cover types in a 500 m buffer around the roost. We chose a 500 m buffer and landscape-level covariates based on previous studies of vulture roosting habitat (Elías 1987; Holland et al. 2019; Hill et al. 2021). All covariates had pairwise correlations < 0.5. We also included a covariate for season (winter: January–March, spring: April–June, summer: July–September, fall: October–December) (Avery et al. 2011; Hill et al. 2021).
We performed model selection using a two-step process. In the first step, we fit a pair of univariate negative binomial regression models of vulture count using each continuous covariate, one with a linear relationship and one with a quadratic relationship. We ranked the two models using Akaike information criterion for small sample sizes (AICc) and used the relationship with the lower AICc score in the next step. In the second modeling step, we fit negative binomial regression models with all subsets of the five covariates. All models included random site intercepts. We ranked the candidate models with AICc and selected the top-ranked model for further inference. We fit negative binomial regression models instead of Poisson regression models in both steps due to overdispersion in the count data. We fit models using package glmmTMB (Brooks et al. 2017) in R version 4.1 (R Core Team 2020). For the top-ranked models, we calculated marginal R2 (variance explained by only the fixed effects in the model) and conditional R2 (the variance explained by the entire model) (Nakagawa and Schielzeth 2013).
Daily temporal variation in vulture counts. Our second objective was to determine whether daily roost use by vultures varies temporally, and if this pattern was affected by urbanization. To address this, we fit circular kernel distributions to the semi-hourly vulture roost counts to determine how roost use changed throughout the day (Rowcliffe et al. 2014). We expected that sunrise and sunset times would be important drivers of variability in vulture count, and these times vary across seasons. Thus, we first transformed vulture count timestamps to solar time, which is a transformation anchored by sunrise and sunset times that allows for direct comparison throughout the year as sunrise and sunset times change (Rowcliffe et al. 2014). Using the timestamped vulture counts, we fit three circular kernel distribution analyses. Each distribution was bounded at one hour before sunrise (5 AM solar time) and one hour after sunset (7 PM solar time), because accurate counts from camera images were not possible in the dark and we did not expect changes in vulture attendance overnight.
For the first analysis, we fit a model to the entire dataset, pooling across all sites and dates. For the second, we fit separate distributions for each season as defined in the previous section. For the final analysis, we pooled sites by degree of urbanization: one category had all roost sites with proportion of urban cover < 0.5 in a 500 m buffer (n = 15 sites) and the other category had all sites with proportion of urban cover ≥ 0.5 (n = 6 sites) (Fig. 1). Proportion urban cover was calculated using NLCD data within a 500-m buffer (Yang et al. 2018). All distributions were fit in R using function fitact in package activity (Rowcliffe 2016). Circular distributions in the final two analyses were compared using randomization tests with function compareCkern from package activity (Rowcliffe 2016).
Results
We monitored 21 roosts (20 anthropogenic [17 communication and 3 water towers] and 1 natural [tree]). We aimed for an even number of anthropogenic and natural roosts but found it difficult to follow vultures closely enough to locate their natural roosts. By contrast, the anthropogenic roosts were well above the tree canopy, which facilitated visual detection. Additionally, birds were easily seen perched on anthropogenic roosts without the need to observe them returning.
We obtained 131,228 roost images overall with an average count of 2.01 vultures per image (maximum = 184). There was an average of 6,249 (standard deviation = 365) images per roost location. Of these images, 49,061 were taken during winter, 53,508 in spring, 26,460 in summer, and 2,199 in fall. We performed 245 visual counts at the 21 roosts (8–19 counts per roost) and observed 1232 vultures (mean = 5.03, standard deviation = 17.40). Of these, 1168 (95%) were black vultures, and 64 (5%) were turkey vultures.
Spatial variation in vulture counts
The top-ranked model of vulture roost count included season, linear effects of distance to water and proportion urban land use, and a quadratic effect of habitat type richness (Table 1). A model that also included a linear effect of elevation had similar support based on AICc (Table 1), but we chose the simpler model for further inference. Marginal R2 for the top model was 0.42 and conditional R2 was 0.69. The proportion of urban habitat within 500 m of the roost had a negative effect on vulture count, with a one standard deviation increase in proportion urban (about 0.22) corresponding to a 68% decrease in max vulture count (Table 2; Figs. 2 and 3). Distance to water was positively related to vulture count, with a one standard deviation increase in distance to water (about 314 m) corresponding to a 93% increase in maximum daily vulture count at a roost (Table 2; Fig. 3). The model predicted peak vulture counts at intermediate levels of habitat richness (Fig. 3), but the effect of richness was not significant at the p = 0.05 level (Table 2). Predicted vulture counts peaked during fall and were similar in winter, spring and summer (Fig. 3).
Vulture roost locations (points) near Marine Corps Air Station Beaufort, South Carolina, USA, October 2019 – August 2020. Point color corresponds to the proportion of urban land cover in a 500-m radius around the roost, and size corresponds to the average number of vultures counted from camera images at the site per hour
Plotted marginal effects of each covariate in the top-ranked model of daily maximum vulture count at roost sites near Marine Corps Air Station Beaufort, South Carolina, USA, October 2019 – August 2020. In each panel, all other covariates besides the focal covariate were held constant at the mean value (continuous covariates) or baseline level (categorical covariates). Error envelopes and bars represent 95% confidence intervals. We defined fall as Oct-Dec, winter as Jan-Mar, spring as April-June, and summer as July-Sept.
Daily temporal variation in vulture counts
Vulture roost counts peaked at and just after sunset, with 53% of all vultures on roosts counted from 5 to 7 pm solar time (Fig. 4a). Counts were lowest in the middle of the day (9 am to 3 pm solar time, Fig. 4a). There were seasonal differences in the daily distribution of counts (Fig. 4b). All seasons came from different circular distributions based on randomization tests (all p < 0.01), but winter, spring, and summer had visually similar hourly distributions whereas for fall, counts were higher, particularly early in the day (6 am – 9 pm solar time) and lower at the end of the day (5 pm – 6 pm solar time) relative to the other seasons (Fig. 4b).
Circular distributions for vulture roost counts at sites near Marine Corps Air Station Beaufort, South Carolina, USA, October 2019 – August 2020. Counts are shown by hour in solar time for (a) all seasons and (b) by season. Shaded areas represent 95% confidence intervals. The histogram in panel (a) represents raw vulture count data. We defined fall as Oct-Dec, winter as Jan-Mar, spring as April-June, and summer as July-Sept.
Vultures roosted at sunrise and sunset to a much greater degree at roost locations with low urbanization relative to locations with high urbanization (Fig. 5a). The two distributions differed based on a randomization test (p < 0.05). At roosts with lower urbanization, 53% of vultures were counted from 5 to 7 pm solar time, whereas only 17% of vultures were counted during this period at more urban roost locations. The contrast between more and less urbanized roost locations persisted across seasons (Fig. 5b).
Circular distributions for vulture roost counts at sites near Marine Corps Air Station Beaufort, South Carolina, USA, October 2019– August 2020. Counts are shown by hour in solar time for (a) sites in two categories of urbanization in all seasons and (b) sites in two categories of urbanization by season. Shaded areas represent 95% confidence intervals. We defined fall as Oct-Dec, winter as Jan-Mar, spring as April-June, and summer as July-Sept.
Discussion
The complexities of wildlife interaction with anthropogenic development have important consequences for prediction and mitigation of human-wildlife conflict. We documented a shift in diel pattern of black and turkey vulture roosting across a landscape with moderate levels of development and human influence. Roosts in more developed areas were used more frequently during the day, whereas roosts occurring in less developed areas were more commonly used at night. Thus, vultures appeared to use this landscape in a temporally dynamic fashion wherein they occupy more anthropogenically developed areas during daylight hours before migrating to outlying rural cover for nocturnal roosting.
Resource availability likely influenced observed patterns of vulture use of roosts. During the day when vultures are active, they use thermal currents emanating from paved surfaces for flight which are more prevalent in developed areas (Novoselova et al. 2020). Food availability is a primary driver of avian home range ecology (Rolando 2002) and urban cover can be an important source of food to vultures (Novaes and Cintra 2015). Indeed, vultures in this area consumed substantial amounts of garbage, indicating extensive foraging in areas with human presence (Hill et al. 2022). The greater road density in urban areas may also provision food via trash and roadkill (Hill et al. 2022). As vultures are almost exclusively diurnal, our results suggest that during the day, they use more developed areas due to increased availability of food and favorable soaring conditions, occupying roosts in these areas to reduce energetic costs. At the conclusion of daily activity, they move to less human influenced areas for nighttime roosting, possibly where the decreased human influence results in less disturbance. Alternatively, less human areas, such as stands of trees, may provide more favorable refugia from nighttime inclement weather (e.g., high winds and rain).
We were unable to detect vultures at night with our methodology. However, the much higher number of vultures counted at roosts during evening compared to morning indicates that vultures departed nocturnal roosts when it was dark but returned before or just after sundown when daylight was still present. This pattern likely results from their foraging ecology; feeding opportunities for vultures depend on arriving at carcasses before they are consumed, creating a strong ecological motivation to begin daily activity as soon as environmental conditions permit (Kendall 2014; Alarcón et al. 2017). Furthermore, white-tailed deer (Odocoileus virginianus) are an important component of vulture diets in this area (Hill et al. 2022), and there is likely a high availability of carcasses in the morning due to increased mortality from predation and vehicle collisions at night (Smith et al. 2019; Mayer et al. 2021). Consequently, both species of vultures concentrate foraging early in the morning (Houston 1988), making early roost departure pivotal to foraging success. Additionally, nocturnal roosts may serve as information centers where naïve birds follow knowledgeable birds to carrion, which promotes relatively synchronous early morning roost departure (Rabenold 1987). Vultures often move from nocturnal roosts to staging areas in early morning where they preen, stretch, and spread their wings to dry their feathers or assist in thermoregulation (Mossman 1976; Clark and Ohmart 1985). Therefore, vultures may not have been foraging the first few hours of the day, but rather departing from nocturnal roosts to initiate daily non-foraging activity.
Our detected patterns of landscape attributes associated with vulture roosting apply more to nocturnal than diurnal roosts because vulture roosting peaked in the evening, and we used maximum number of daily vultures counted as a response variable. This is reflected in the increase in roosting in areas with less developed land cover, which was the pattern for our nocturnal roosts (i.e., those with greater concentrations of counts at sunrise and sunset). A similar relationship between urban land cover and nocturnal roosting based on telemetry data was also documented for black and turkey vultures in this area (Hill et al. 2021). The inclusion of land cover richness in the top model suggests that there was some influence of landscape fragmentation on roosting, though not a significant effect. This diverges from a prior study in the same area where habitat fragmentation was consistently associated with greater roosting probability (Hill et al. 2021). However, that study was based on individual roosting behavior which was not limited to roosting on towers. Thus, the factors contributing to an individual’s selection of roosting habitat may be different than those contributing to differential roosting probability among anthropogenic structures.
The reasons for an increase in roosting with greater distance to water are not altogether clear as our results contrast with previous findings (Thompson et al. 1990; Evans 2013). Considering the large home ranges of these species in South Carolina (annual mean 30.3–340.5 km2) (DeVault et al. 2004; Holland et al. 2017), water sources are likely readily accessible even at roosts furthest from water, about 1500 m in this study. Therefore, although distance to water was included in the top model, actual distances may not be biologically meaningful considering the extensive movement of these animals. Vultures could have also obtained water from sources other than the permanent water bodies included in our analysis such as drainage pipes, water sprinklers, or pools formed by rainwater (Gbogbo and Awotwe-Pratt 2008; Evans 2013). Alternatively, distance to water may be correlated with other landscape attributes unaccounted for in our analysis.
The disproportionate number of black vultures in our physical counts (~ 95% of vultures counted overall) indicates that our conclusions may not be particularly applicable to turkey vultures. Since we mostly monitored communication towers and turkey vultures are present in the area (Avery et al. 2011; Walter et al. 2012), the lack of turkey vultures in our counts suggests interspecific segregation of roosting. Black vultures appear to be the predominate species using towers, whereas turkey vultures are likely more heavily using natural roosts which we largely did not monitor. Similarly, other studies in the Southeastern US have documented higher use of transmission towers by black vultures and a separation among species between anthropogenic and natural roosts (Avery et al. 2002; Partridge 2021).
Apparent segregation at roosts may result from competition, as higher perches are generally preferred, and black vultures tend to be dominant over turkey vultures (Buckley 1998; Evans and Sordahl 2009). At an electricity pylon in Texas, turkey vultures used the highest points of the structure, but were forced into lower, less preferred perches when black vultures were present (Buckley 1998). Turkey vultures in Ohio roost on the highest perches on pylons when there are few black vultures (M. Pfeiffer, personal observation). Variation in roost locations may also relate to differences in flight and foraging behavior. Turkey vultures tend to fly closer to the ground because they locate carrion by olfaction, whereas black vultures fly at higher altitudes, detecting carrion visually by observing the activity of turkey vultures (Stolen 2000; Avery et al. 2011). Differences in roosting may thus mirror divergent vertical space use between the species. Additionally, black vultures have a higher wing loading, which makes them more reliant on uplift to initiate flight (Graves 2017; Byrne et al. 2019), and roosting at greater heights may facilitate departure from roosts. Lastly, differences in foraging strategies and social behavior make communal roosting more important for black vultures compared to turkey vultures (Buckley 1996, 1997) and towers may permit greater aggregations of birds than do natural roosts. In addition to the partitioning of carrion resources (Holland et al. 2017; Byrne et al. 2019), our results indicate that segregation of roosting habitat may also be a component of coexistence among the two species.
In concordance with previous research in this area, the relationships between landscape factors and vulture roosting were similar across seasons (Hill et al. 2021). Our data further suggests that temporal patterns in roost use remain constant seasonally, despite seasonal variation in home range sizes (Avery et al. 2011). During fall, there was an estimated increase in vulture counts and diel patterns appeared to diverge from other seasons. However, the much smaller fall sample sizes make it difficult to discern whether this was an actual seasonal effect (e.g., addition of fledglings to the roosts) on roosting or an artifact of disparate sample sizes. Overall, the risk of conflict with vultures resulting from roosting behavior appears to be relatively static throughout the year in our study area.
Our physical counts indicate that turkey vultures remain largely unaccounted for in our analysis. As a result, it remains unclear whether the same patterns of roost use and landscape attributes we documented apply to turkey vultures. Additionally, the structures we examined likely vary regarding the amount of space suitable for vulture roosting, but we were unable to quantify these differences. If larger structures are placed in areas of less human influence, this may impact our documented relationships between vulture abundance and landscape attributes. Lastly, our focus on anthropogenic structures makes it uncertain whether the relationships we found also apply to natural roosts. However, natural roosts can also be a source of conflict with humans (Tillman et al. 2002), so the factors influencing vulture use of natural roosts should be a topic of future research.
Our results have implications for understanding human-vulture conflict in this area. Diel patterns of roost use suggest that when roosts are near aircraft activity, there would be an elevated collision risk before sunrise because vultures would be departing the roosts during this time. In the evening, collision risk would be greater in the 2–3 h before sunset because this is the period when vultures are moving back to the roost. Risk would be comparatively low after sunset because vultures are already on the roost where they will be inactive until the next morning. Vulture-aircraft collisions can occur during midday as this is when vultures generally spend the greatest amount of time in flight (Avery et al. 2011), but in terms of movements around roosts, the risk of vulture collisions with aircraft would be greater in the few hours before sunrise and sunset. It is important to note in areas where turkey vultures are believed to be abundant, it may be necessary to conduct aerial flights or radio-tag a subset of birds in order detect a high proportion of their roosts, given the low counts of turkey vultures we observed with our methodology.
Communication towers appear to be an important source of roosting habitat for vultures in this area and may contribute to local abundances. Three of the towers hosted particularly large committees of vultures, with numbers of birds in a single camera image routinely exceeding 100 and on occasion greater than 180. As roosting aggregations are associated with increased conflict with humans (Kluever et al. 2020), these towers have the potential to be a root cause of human-vulture conflict in this area. Consequently, dispersal efforts such as effigies and harassment may be necessary to eliminate these large roosts (Lowney 1999; Avery et al. 2002; Pfeiffer et al. 2021). The identification and dispersal of major vulture roosts will be an important component of wildlife damage management as vulture populations continue to grow across North America.
Data availability
Dataset is available in the Supporting Information.
Literature cited
Alarcón PA, Morales JM, Donázar JA, Sánchez-Zapata JA, Hiraldo F, Lambertucci SA (2017) Sexual-size dimorphism modulates the trade-off between exploiting food and wind resources in a large avian scavenger. Sci Rep 7:1–9
Amazon (2017) Terrain Tiles on AWS
Arrondo E, García-Alfonso M, Blas J, Cortes‐Avizanda A, De la Riva M, Devault TL, Fiedler W, Flack A, Jimenez J, Lambertucci SA (2021) Use of avian GPS tracking to mitigate human fatalities from bird strikes caused by large soaring birds. J Appl Ecol 58:1411–1420
Avery ML, Humphrey JS, Daughtery TS, Fischer JW, Milleson MP, Tillman EA, Bruce WE, Walter WD (2011) Vulture flight behavior and implications for aircraft safety. J Wildl Manage 75:1581–1587
Avery ML, Humphrey JS, Tillman EA, Phares KO, Hatcher JE (2002) Dispersing vulture roosts on communication towers. J Rapt Res 36:45–50
Bonier F, Martin PR, Wingfield JC (2007) Urban birds have broader environmental tolerance. Biol Lett 3:670–673
Brooks ME, Kristensen K, Van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400
Buckley NJ (1996) Food finding and the influence of information, local enhancement, and communal roosting on foraging success of north american vultures. Auk 113:473–488
Buckley NJ (1997) Experimental tests of the information-center hypothesis with black vultures (Coragypsatratus) and turkey vultures (Cathartesaura). Behav Ecol Sociobiol 41:267–279
Buckley NJ (1998) Interspecific competition between vultures for preferred roost positions. Wilson Bull: 122–125
Buckley NJ, Kluever BM, Driver R, Rush SA (2022) Black vulture (Coragyps atratus). Rodewald P editor. Birds of the World. Cornell Lab of Ornithology, Ithaca, NY
Byrne ME, Holland AE, Turner KL, Bryan AL, Beasley JC (2019) Using multiple data sources to investigate foraging niche partitioning in sympatric obligate avian scavengers. Ecosphere 10:e02548
Campbell MON (2014) The impact of urbanization and agricultural development on vultures in El Salvador. Vulture News 66:16–28
Carey RT, Salazar JS (2017) Estimated total impact of tourism in Beaufort County, SC, 2016. Pendleton, South Carolina, USA: Regional Transactions Concepts, LLC
Clark RG, Ohmart RD (1985) Spread-winged posture of Turkey vultures: single or multiple function? Condor 87:350–355
DeVault TL, Blackwell BF, Seamans TW, Begier MJ, Kougher JD, Washburn JE, Miller PR, Dolbeer RA (2018) Estimating interspecific economic risk of bird strikes with aircraft. Wildl Soc Bull 42:94–101
DeVault TL, Reinhart BD, Brisbin IL, Rhodes OE (2004) Home ranges of sympatric black and turkey vultures in South Carolina. Condor 106:706–711
Dolbeer RA (2011) Increasing trend of damaging bird strikes with aircraft outside the airport boundary: implications for mitigation measures. Human-Wildlife Interact 5:235–248
Elías EEI (1987) Feeding habits and ingestion of synthetic products in a black vulture population from Chiapas, Mexico. Acta Zool Mex 19:1–15
Evans BA (2013) Dynamics of a problematic vulture roost in southwest Florida and responses of vultures to roost-dispersal management efforts. Florida Gulf Coast University
Evans BA, Sordahl TA (2009) Factors influencing perch selection by communally roosting Turkey vultures. J Field Ornithol 80:364–372
Gbogbo F, Awotwe-Pratt V (2008) Waste management and hooded vultures on the Legon campus of the University of Ghana in Accra, Ghana, West Africa. Vulture News 58:16–22
Graves GR (2017) Sexual monomorphism in wing loading and wing aspect ratio in black vulture (Coragyps atratus) and Turkey vulture (Cathartes aura). Proc Biol Soc Wash 130:240–249
Harmange C, Bretagnolle V, Chabaud N, Sarasa M, Pays O (2021) Diel cycle in a farmland bird is shaped by contrasting predation and human pressures. Biol J Linn Soc 134:68–84
Hill JE, Holland AE, Brohl LK, Kluever BM, Pfeiffer MB, DeVault TL, Belant JL (2022) Diets of black vultures and Turkey vultures in Coastal South Carolina, USA with a review of species’ dietary information. Southeast Nat 21:11–27
Hill JE, Kellner KF, Kluever BM, Avery ML, Humphrey JS, Tillman EA, DeVault TL, Belant JL (2021) Landscape transformations produce favorable roosting conditions for turkey vultures and black vultures. Sci Rep 11:1–10
Hobbs RJ, Higgs E, Harris JA (2009) Novel ecosystems: implications for conservation and restoration. Trends Ecol Evol 24:599–605
Holland AE, Byrne ME, Bryan AL, DeVault TL, Rhodes OE, Beasley JC (2017) Fine-scale assessment of home ranges and activity patterns for resident black vultures (Coragyps atratus) and turkey vultures (Cathartes aura). PLoS ONE 12:e0179819
Holland AE, Byrne ME, Hepinstall-Cymerman J, Bryan AL, DeVault TL, Rhodes OE, Beasley JC (2019) Evidence of niche differentiation for two sympatric vulture species in the Southeastern United States. Mov Ecol 7:31
Houston D (1988) Competition for food between neotropical vultures in forest. Ibis 130:402–417
Jaggard AK, Smith N, Torpy FR, Munro U (2015) Rules of the roost: characteristics of nocturnal communal roosts of rainbow lorikeets (Trichoglossus haematodus, Psittacidae) in an urban environment. Urban Ecosyst 18:489–502
KC KB, Koju NP, Bhusal KP, Low M, Ghimire SK, Ranabhat R, Panthi S (2019) Factors influencing the presence of the endangered egyptian vulture Neophron percnopterus in Rukum, Nepal. Glob Ecol Conserv 20:e00727
Kendall CJ (2014) The early bird gets the carcass: temporal segregation and its effects on foraging success in avian scavengers. The Auk: Ornithological Advances 131:12–19
Kiff LF (2000) The current status of north american vultures. In: Chancellor R, Meyburg B-U (eds) Raptors at risk. WWGBP/Hancock House, Berlin and Surrey, Canada, pp 175–189
Kirk D, Mossman J (2020) Turkey vulture (Cathartes aura). In: Poole A, Gill F (eds) Birds of the World. Cornell Lab of Ornithology, Ithaca, NY
Kluever BM, Pfeiffer MB, Barras SC, Dunlap BG, Humberg LA (2020) Black vulture conflict and management in the United States: damage trends, management overview, and research needs. Human–Wildlife Interact 14:376–389
Lowney MS (1999) Damage by black and turkey vultures in Virginia, 1990–1996. Wildl Soc Bull 27:715–719
Mallon JM, Bildstein KL, Katzner TE (2016) In-flight turbulence benefits soaring birds. The Auk: Ornithological Advances 133:79–85
Mayer M, Nielsen JC, Elmeros M, Sunde P (2021) Understanding spatio-temporal patterns of deer-vehicle collisions to improve roadkill mitigation. J Environ Manage 295:113148
Mossman M (1976) Turkey vultures in the Baraboo Hills, Sauk County, Wisconsin. Passeng Pigeon 38:93–99
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142
NOAA (2021) Climate Data Online. National Oceanic and Atmospheric Administration
Novaes WG, Cintra R (2013) Factors influencing the selection of communal roost sites by the black vulture Coragyps atratus (Aves: Cathartidae) in an urban area in Central Amazon. Zoologia (Curitiba) 30:607–614
Novaes WG, Cintra R (2015) Anthropogenic features influencing occurrence of black vultures (Coragyps atratus) and Turkey vultures (Cathartes aura) in an urban area in central amazonian Brazil. The Condor: Ornithological Applications 117:650–659
Novoselova NS, Novoselov AA, Macarrão A, Gallo-Ortiz G, Silva WR (2020) Remote sensing applications for abating aircraft–bird strike risks in southeast Brazil. Human–Wildlife Interact 14:8
Nyhus PJ (2016) Human–wildlife conflict and coexistence. Annu Rev Environ Resour 41:143–171
Partridge HC (2021) The local and Landscape features Associated with Roost Attendance and Nesting Success in Urban Black vulture (Coragyps atratus) and Turkey vulture (Cathartes aura) populations. The University of North Carolina at Charlotte
Pfeiffer MB, Blackwell BF, DeVault TL (2018) Quantification of avian hazards to military aircraft and implications for wildlife management. PLoS ONE 13:e0206599
Pfeiffer MB, Blackwell BF, Seamans TW, Buckingham BN, Hoblet JL, Baumhardt PE, DeVault TL, Fernández-Juricic E (2021) Responses of turkey vultures to unmanned aircraft systems vary by platform. Sci Rep 11:1–13
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria
Rabenold PP (1987) Recruitment to food in black vultures: evidence for following from communal roosts. Anim Behav 35:1775–1785
Rolando A (2002) On the ecology of home range in birds. Revue d’ecologie 57:53–73
Rowcliffe M (2016) Package ‘activity’
Rowcliffe M, Kays R, Kranstauber B, Carbone C, Jansen PA (2014) Quantifying levels of animal activity using camera trap data. Methods Ecol Evol 5:1170–1179
Sauer J, Niven D, Hines J, Ziolkowski D Jr, Pardieck K, Fallon J, Link W (2017) The North American Breeding Bird Survey, results and analysis 2015–2017.Laurel, Maryland USA: US Geological Survey Patuxent Wildlife Research Center
Smith JA, Donadio E, Pauli JN, Sheriff MJ, Middleton AD (2019) Integrating temporal refugia into landscapes of fear: prey exploit predator downtimes to forage in risky places. Oecologia 189:883–890
Stolen ED (2000) Foraging behavior of vultures in central Florida. Fla Field Naturalist 28:173–181
Sweeney TM, Fraser JD (1986) Vulture roost dynamics and monitoring techniques in southwest Virginia. Wildlife Society Bulletin (1973–2006) 14: 49–54
Thompson WL, Yahner RH, Storm GL (1990) Winter use and habitat characteristics of vulture communal roosts. J Wildl Manage 54:77–83
Tillman EA, Humphrey JS, Avery ML (2002) Use of vulture carcasses and effigies to reduce vulture damage to property and agriculture. Timm R, Schmidt R editors. Proceedings of the Vertebrate Pest Conference. Reno, NV: University of California, Davis, p123-128
US Census Bureau (2021) 2020 Census
Walter WD, Fischer JW, Humphrey JS, Daughtery TS, Milleson MP, Tillman EA, Avery ML (2012) Using three-dimensional flight patterns at airfields to identify hotspots for avian–aircraft collisions. Appl Geogr 35:53–59
Yang L, Jin S, Danielson P, Homer C, Gass L, Bender SM, Case A, Costello C, Dewitz J, Fry J (2018) A new generation of the United States National Land Cover Database: requirements, research priorities, design, and implementation strategies. ISPRS J Photogrammetry Remote Sens 146:108–123
Zeigler JM (1959) Origin of the Sea Islands of the southeastern United States. Geogr Rev 49:222–237
Acknowledgements
J. Humphrey assisted with initial field reconnaissance. This research was supported in part by the U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Wildlife Services, and the U.S. Air Force. The findings and conclusions in this article are those of the authors and do not necessarily represent the views of the US Fish and Wildlife Service or the US Department of Agriculture.
Funding
This research was supported in part by the U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Wildlife Services, and the U.S. Air Force.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study design and conception. Data collection was performed by Amanda Holland. Analysis was performed by Kenneth Keller and Amanda Holland. Jacob Hill and Kenneth Kellner wrote the first draft of the manuscript and all authors commented on previous versions. The final manuscript was read and approved by all authors.
Corresponding author
Ethics declarations
Ethics approval
Institutional animal care and use approval was not required for this study because there was no animal capture or handling.
Competing interests
The authors have no relevant or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hill, J.E., Kellner, K.F., Holland, A.E. et al. Urbanization influences spatiotemporal patterns of roost site selection by black vultures and turkey vultures. Urban Ecosyst 26, 967–979 (2023). https://doi.org/10.1007/s11252-023-01328-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-023-01328-w