Abstract
Contagious bovine pleuropneumonia (CBPP), a severe respiratory disease, is characterized by massive inflammation of the lung especially during the acute clinical stage of infection. Tissue samples from cattle, experimentally infected with Mycoplasma mycoides subsp. mycoides Afadé, were subjected to histopathological and immunohistochemical examination in order to provide insight into innate immune pathways that shape inflammatory host responses. Lung lesions were characterized by vasculitis, necrosis, and increased presence of macrophages and neutrophils, relative to uninfected animals. The presence of three cytokines associated with innate inflammatory immune responses, namely, IL-1β, IL-17A, and TNF-α, were qualitatively investigated in situ. Higher cytokine levels were detected in lung tissue samples from CBPP-affected cattle compared to samples derived from an uninfected control group. We therefore conclude that the cytokines TNF-α and IL-1β, which are prevalent in the acute phase of infections, play a role in the inflammatory response seen in the lung tissue in CBPP. IL-17A gets released by activated macrophages and attracts granulocytes that modulate the acute phase of the CBPP lesions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Contagious bovine pleuropneumonia (CBPP) is a severe respiratory disease in cattle that is present in many countries in sub-Saharan Africa. CBPP is caused by Mycoplasma mycoides subsp. mycoides (Mmm), a member of the “Mycoplasma mycoides cluster” (Fischer et al. 2012) which in addition to Mmm comprises four related mycoplasma lineages, i.e., M. mycoides subsp. capri, M. capricolum subsp. capricolum, M. capricolum subsp. capripneumoniae, and M. leachii. Besides having a negative impact on the productivity of pastoral cattle farming systems, CBPP is a disease of both regional and international trade, limiting the marketability of cattle in affected regions or countries, consequently resulting in substantial financial losses that negatively impact the livelihood of many people in sub-Saharan Africa (Jores et al. 2013; Thiaucourt et al. 2004b). Cattle infected with Mmm can develop acute, subacute, or chronic disease. Acute CBPP is characterized by pyrexia, anorexia, and respiratory signs including rapid and painful breathing and occasionally coughing. Furthermore, large quantities of pleural fluid containing high numbers of mycoplasma are often found during necropsy (Weldearegay et al. 2015). Cattle that exhibit acute disease can either clear the infection, become chronically infected, or die. The current live vaccine against CBPP T1/44 occasionally causes severe side effects at the site of inoculation and, most importantly, lacks efficacy and confers immunity only for up to 1 year, which makes repeated vaccinations essential (Thiaucourt et al. 2004a). The development and subsequent implementation of an improved vaccine, which confers immunity for more than 1.5 years, would greatly benefit a progressive control of CBPP (Ssematimba et al. 2015). However, an increased understanding of the host-pathogen interactions including protective host immune responses is a prerequisite for the rational design of novel vaccines (Jores et al. 2013). Several previous studies have been performed in order to identify how host mechanisms confer immunity to CBPP. The importance of both humoral and T cell-mediated immune responses in mediating protection has been described. Interferon gamma-secreting CD4+ T cells have been associated with protection against CBPP during primary infections (Dedieu et al. 2005). These results could however not be confirmed by Jores et al. (2008) and Sacchini et al. (2011), though it is likely that specific CD4+ T cell subsets are involved in immunity (Totte et al. 2010, 2008).
Typical CBPP lesions show a fibrinous pleuropneumonia and represent a lobar and lobular pneumonia which usually undergoes acute progression. Classically, the CBPP has four stages. (1) Congestion occurs in the first 24 h postinfection. This stage is characterized histologically by vascular engorgement, intraalveolar fluid, small numbers of neutrophils, and infectious agents. Grossly, the lung is severely hyperemic. (2) Red hepatization or consolidation includes vascular congestion with extravasation of red cells into alveolar spaces, along with increased numbers of neutrophils and fibrin. The filling of alveoli by the exudate leads to a gross appearance of solidification, or consolidation, of the alveolar parenchyma. (3) The stage of grey hepatization is characterized by disintegration of red blood cells, with persistence of the neutrophils and fibrin. The alveoli still appear consolidated, but grossly the color is paler and the cut surface is drier. (4) In the stage of resolution, the pulmonary tissue shows complete recovery. Stages 1 and 2 (congestion, red hepatization) represent more acute pathological findings, whereas stage 3 (grey hepatization) describes chronical pathological findings. Congestion and red and grey hepatization can occur within one individual in parallel at the same time.
Sequestra in the pulmonary parenchyma are a characteristic of chronically diseased animals. They consist of a layer of fibrous tissue enclosing necrotic cells, which is composed of a purulent exudate and live Mycoplasma (Caswell and Williams 2007; Schieck et al. 2014). An increased presence of myeloid cells in affected lung tissue was observed in a previous study, though numbers were low (Jores et al. 2008).
Despite a large number of experimental infections performed in the past, documentation of pathological lesions with respect to the in situ presence of distinct host immune cells as well as the presence of cytokines is still lacking. Here, we investigated lungs, mediastinal lymph nodes, and kidneys from ten cattle experimentally infected with CBPP (Sacchini et al. 2011) using standard histologic procedures and immunocytochemistry. The presence of Mmm was confirmed using rabbit polyclonal antibodies. Furthermore, we investigated the in situ presence of the inflammatory cytokines tumor necrosis factor alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-17A (IL-17A) in experimentally infected and non-infected animals. The data revealed an increase in inflammatory cytokine expression in lung tissues in infected animals compared to non-infected animals.
Materials and methods
Ethical considerations
The protocols used in this study were designed and performed in strict accordance with the Kenyan legislation for Animal Experimentation and were approved by the Institutional Animal Care and Use Committee (IACUC reference number 2008.08 [bovine experimental infection] and 2008.14 [rabbit polyclonal antibodies]). Since 1993, the International Livestock Research Institute (ILRI) has complied voluntarily with the United Kingdom’s Animals (Scientific Procedures) Act 1986 that contains guidelines and codes of practice for the housing and care of animals used in scientific protocols.
Sample collection
The cattle population and the experimental procedure used in the present study have been described in detail elsewhere (Sacchini et al. 2011). In brief, Kenyan Boran bullocks (Bos indicus), 14 to 16 months of age at sourcing, were kept at ILRI campus for 2.5 months prior to the transfer to the Animal Biosafety Level 2 facility. Upon arrival to ILRI, the animals were dewormed and treated prophylactically against babesiosis and anaplasmosis. The cattle were revaccinated against lumpy skin disease, anthrax, blackleg, and foot and mouth disease. All animals were tested negative for presence of antibodies against CBPP, East Coast fever, and trypanosomiasis. The cattle were infected by intubation using M. mycoides subsp. mycoides strain Afadé which has been described elsewhere (Fischer et al. 2015). The animals were monitored daily for up to 30 days post infection (dpi) when the cattle were euthanized and subjected to postmortem analysis.
Necropsy
The necropsy was done according to a standard procedure (Strafuss 1988). Specimens were taken from lung, mediastinal lymph nodes, and kidneys and were subsequently transferred to tubes containing 5 % buffered formalin solution. Tubes were stored at room temperature until further processing.
Microbiological and serological analysis
Lung samples and carpal joint fluid and pleural fluid specimens were used for culture isolation of Mmm while seroconversion was confirmed using complement fixation test (CFT) (CIRAD, France).
Polyclonal rabbit antibody production
For antigen preparation, Mmm strain Afadé was grown in 100 ml pleuropneumonia-like organism (PPLO) broth (BD Difco™, Germany) supplemented with 20 % horse serum (Sigma, Germany). Cells were pelleted using centrifugation at 5000g for 20 min and resuspended in 1 ml of PBS. The cell suspension was ruptured using zirconium beads (0.1 mm diameter, Carl Roth, Germany) in a FastPrep® Instrument (Qbiogene, Germany) three times for 40 s at intensity setting 5.0, followed by heat inactivation at 60 °C for 10 min. A rabbit was immunized with Mmm antigen mixed with the same volume of complete Freund’s adjuvant in the first round and the same volume of incomplete Freund’s adjuvant for the booster injection. Heat-inactivated antigen with adjuvant was injected intradermally and subcutaneously (two depots each). Immunizations were given 4 weeks apart. Preimmunization sera were collected 2 weeks before initial immunization and postimmunization sera were collected 4 weeks after the second immunization.
Standard histological examination and immunocytochemistry
Formalin-buffered tissue samples were embedded in paraffin wax and subsequently cut into 2-μm slices, using a rotary microtome, and stained with hematoxylin and eosin (HE staining) according to standard protocols.
Immunocytochemistry employing three cytokine markers has been carried out on paraffin-embedded lung tissues. Bovine IL-1β, IL-17A, and TNF-α were detected by using biotinylated rabbit anti-bovine IL-1β polyclonal antibody (article no. ab23778, Abcam, UK), biotinylated rabbit anti-bovine IL-17A polyclonal antibody (article no. PBB0277B-050, Kingfisher Biotech, Inc., USA), and biotinylated rabbit anti-bovine TNF-α polyclonal antibodies (article no. AHP852B, AbD Serotec, Germany), respectively. The SuperVision 2 HRP Single Species (DCS, Germany) was used for detection of biotinylated rabbit and mouse antibodies. Tissues were stained with an irrelevant antibody from the same species to check non-specific binding, and no non-specific binding was evident. Immunoreactive signals were reddish-brown.
Infected animals, showing fibrinous (BD97 and BD107) and non-fibrinous bronchopneumonia (BD92, BD95, BD102, BD105, BD106, BD111, BD115, and BD116), and non-infected animals (control) were compared. Cytokine-positive cells were evaluated within ten non-overlapping, high-power fields (×40 final magnification) on a single microscope by one experienced pathologist. Sections were assessed with the operator blind to clinical details. Macrophages and granulocytes were easily recognizable by staining, size, and morphologic characteristics. Fields containing parts of lymphoid aggregates, massive necrosis, or fibrosis were avoided.
Results
Clinical symptoms of CBPP such as fever and coughing peaked between 12 and 15 dpi. One animal (BD97) showed severe clinical symptoms and had to be euthanized before the end of the experiment for animal welfare reasons. Infection was confirmed by seroconversion via CFT and isolation of Mmm from cultured lung tissues or pleural fluids (Sacchini et al. 2011). At necropsy, typical CBPP lesions were observed in nine out of ten animals. Most pathomorphological lesions were unilaterally present in the left lung with a comparable frequency in the apical and in the diaphragmatic lobe (Sacchini et al. 2011). Only two animals had lesions in the right lung or on both sides. Acute fibrinous bronchopneumonia was only visible in animals BD97 and BD107. In addition, animal BD97 contained a large amount of pleural fluid infected with Mycoplasma. The other animals showed mainly chronic lesions with fibrosis and sequestrum formation. No significant differences in the presence of γδ+ T cells, monocytes, CD8+ T cells, neutrophils, B cells, or granulocytes (Additional file 1) between CBPP animals showing fibrinous or non-fibrinous bronchopneumonia were detected.
Macroscopically visible renal infarcts were present in three animals (BD97, BD107, and BD116) (Table 1).
The histological findings in the lungs were characterized by a juxtaposition of more acute lesions in parallel to more chronic morphological lesions. In the lung, bronchi and bronchioli frequently contained cellular necrotic debris with transmural full-thickness necrosis of bronchial and bronchiolar walls (Fig. 1a). Besides fibrinous bronchopneumonia, alveolitis (Fig. 1b), and fibrinous pleuritis, dilated and oedematous, interlobular septa occurred in these CBPP pulmonary lesions. Severe leukocytoclastic vasculitis with full-thickness necrosis and lysis of the vascular wall was observed within the pulmonary parenchyma (Fig. 1c).
a Lung—a shows severe, necrotizing bronchiolitis, with severe dense cellular, necrotic debris obstructing the bronchiolar lumen. The remaining bronchiolar wall shows loss of epithelial cells and severe infiltration of pulmonary and interstitial mixed inflammatory infiltrate with loss of pulmonary structure. Arrows indicate delineation of bronchiolar lining. b Lung—b shows intraalveolar proteinaceous, fibrinous precipitates (arrows) with loss of pulmonary architecture as well as thickened alveolar septa with mixed inflammatory, interstitial infiltrates within alveolar septa and interstitial space. c Lung—c shows a severe, necrotizing vasculitis of a pulmonary arteriole with loss of vascular wall (arrows indicate remnants of necrotic vascular wall) and severe, necrotic, cellular debris obliterating the vascular lumen. The perivascular interstitial space shows perivascular, mixed inflammatory cells and protein precipitates. d Lung—d shows perivascular pulmonary fibrosis extending into the interstitial space, with immature extracellular matrix and mixed inflammatory cells. Arrow indicates arterial media hypertrophy. e Lung—e shows an intrapulmonary arteriole with an adhesive, lumen-obstructing thrombus and media hypertrophy. Arrow indicates wall of arteriole. f Lung—f shows severe chronic, diffuse pleuritis with extensive subpleural fibrosis and neovascularization and loss of pulmonary architecture. Arrow indicates pleural surface. g Lung—g shows an interstitial inflammatory infiltrate composed of neutrophilic and eosinophilic granulocytes (arrows) within the interstitial space of the lung. h Pulmonary lymphnode—h shows a pulmonary lymph node with follicular hyperplasia and activated germinal centers (arrows). Size standards are displayed in the lower right corner of each picture: black represents 100 μm; grey represents 50 μm, and yellow represents 20 μm
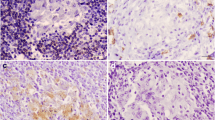
Blood vessels in the interstitial septa often showed thrombosis (Fig. 1e) and vasculitis (Fig. 1c). These fibrinous thrombi often contained leukocytes and macrophages. Occasionally progressive organization of connective tissue and neoangiogenesis within immature granulation tissue were evident in the lung (Fig. 1d, f). The stroma contained variable amounts of neutrophils and lymphocytes and occasionally eosinophilic granulocytes (Fig. 1g). Pulmonary lymph nodes of all animals developed a moderate to severe follicular hyperplasia with activated germinal centers (Fig. 1h). Lung tissues were evaluated using three different cytokine-specific markers, and expression of the these three cytokines (TNF-α, IL-1β, and IL-17A) were investigated. At least three different sections of the lung showing pathomorphological changes were evaluated semiquantitatively (Table 2). CBPP lesions were characterized by increased levels of TNF-α, IL-1β, and IL-17A compared to the group of five non-infected animals (Fig. 2, Table 2).
Representative qualitative results of the detection of the cytokines IL-1β, IL-17A, and TNF-α. The following polyclonal antibodies were used. a Anti- IL-1β: Lung: control animal; b Anti- IL-1β: Lung: acute CBPP lesion; c Anti- IL-17A: Lung: control animal; d Anti- IL-17A: Lung: acute CBPP lesion; e Anti- TNF-α: Lung: control animal; f Anti- TNF-α: Lung: acute CBPP lesion; g Anti- Mycoplasma: Lung: control animal; h Anti- Mycoplasma: Lung: acute CBPP lesion. Black bars in the lower right corner of images in a–f is the size standard representing 100 μm, black bars in images of g, h represent 20 μm. Arrows indicate positive immunoreactive signals (reddish-brown)
Discussion
This study aimed to characterize morphological lesions in animals experimentally infected with Mycoplasma mycoides subsp. mycoides in greater detail, by employing HE staining and immunocytochemistry.
The tissue samples investigated were derived from cattle that had been successfully infected as shown by seroconversion and the isolation of the causative agent from all animals (Sacchini et al. 2011). The gross pathological lung lesions reflected the typical pathological patterns described for CBPP (Caswell and Williams 2007). Necrotic areas of the pulmonary lymph nodes have also been previously reported from animals showing acute clinical symptoms of CBPP (Scanziani et al. 1997). Four out of ten animals in the present study showed mild focal interstitial renal inflammation and necrosis (Table 1) as reported elsewhere (Grieco et al. 2001). Seven out of ten infected animals showed mild inflammation of the liver parenchyma and interstitium. Other investigated organs did not show any noteworthy findings. The lesions characterized by HE staining confirmed previous findings related to CBPP. Altogether, the histological patterns observed in the animals used in this study manifested similar lesions to those observed in natural outbreaks.
The presence of Mycoplasma antigen in CBPP lesions was also investigated. Rabbit polyclonal sera were used to detect the presence of antigen. As expected, the presence of Mycoplasma antigen in macrophages and in lung tissue was confirmed (Fig. 2h).
The results obtained in this study varied within the group of tested animals and could not be correlated with specific clinical and pathomorphological data of individual animals. Thus, use of increased animal numbers in future studies may result in correlations not identified here.
Since it has been indicated that antibody levels can correlate with disease severity (Mulongo et al. 2015; Nicholas et al. 2009; Schieck et al. 2014), immune-mediated vasculitis might be involved in the pathogenesis of CBPP, which will require confirmation in future studies. It has been stated that the presence of vasculitis in CBPP might be caused by antigen-antibody complexes deposited in arterial walls (Thiaucourt et al. 2004b). TNF-α plasma levels are increased in acute diseased animals compared to animals with mild disease symptoms (Sacchini et al. 2012). Therefore, the presence of TNF-α levels in the target tissue was evaluated. As expected, elevated levels of TNF-α in the lung tissues confirmed an involvement of the proinflammatory cytokine in pathogenesis of CBPP as previously proposed (Sacchini et al. 2012). Alveolar macrophages, residing in the lungs, are likely to initiate a response after interaction with the pathogen and be a source of TNF-α and proinflammatory cytokines as indicated in previous in vitro experiments (Jungi et al. 1996). However, it is possible that a component of the CD8+ T cell response also contributed to increased TNF-α levels (Hamada et al. 2013). TNF-α is crucial in protection against other respiratory infections such as tuberculosis. It has been shown that TNF-α released from both macrophages and T cells contributes in distinct ways to the sustained control of Mycobacterium tuberculosis (Allie et al. 2013). A role for this cytokine in the control of CBPP should be tested in future studies.
The presence of macrophages and granulocytes in CBPP lesions prompted us to investigate the presence of the cytokines IL-1β and IL-17A, which are markers for innate immune responses and granulocyte-attracting molecules, respectively (McAleer and Kolls 2014). Both cytokines were evidently present in affected lung tissue and are likely to mediate the immune responses observed in CBPP. An increase in IL-1β is likely to be induced by alveolar macrophages or dendritic cells after contact with mycoplasma molecules and will stimulate the acute phase response and secretion of IL-17 by γδ+ T cells, natural killer cells, or CD4+ T cells in case of a memory immune response. One possible hypothesis is that IL-17 has major protective roles in adaptive and innate responses (McAleer and Kolls 2014). In respiratory infections, IL-17 recruits neutrophils to the site of inflammation and affects lung epithelial cells by stimulating antimicrobial peptide secretion and mucus production (Newcomb et al. 2013). In addition, memory Th17 cells are important in adaptive immunity. However, in some cases IL-17 can drive acute lung injury, as in experimental infection with the influenza strain H1N1 (Li et al. 2012). If HIN1 is a representative model, it is therefore possible that in CBPP, IL-17 could contribute to both protective and deleterious effects.
To conclude, innate inflammatory responses have been observed in affected lungs. The main drivers of the innate response are likely to be macrophages, granulocytes, and to a lesser extent epithelial cells. We confirmed elevated in situ levels of the three proinflammatory cytokines IL-1β, IL-17A, and TNF-α in lung tissues from cattle experimentally infected with Mmm.
Abbreviations
- BALT:
-
Bronchoalveolar lymphoid tissue
- CBPP:
-
Contagious bovine pleuropneumonia
- dpi:
-
Days post infection
- IACUC:
-
Institutional Animal Care and Use Committee
- IL-1β:
-
Interleukin-1 beta
- IL-17A:
-
Interleukin-17A
- ILRI:
-
International Livestock Research Institute
- Mmm :
-
Mycoplasma mycoides subsp. mycoides
- TNF-α:
-
Tumor necrosis factor alpha
References
Allie, N., Grivennikov, S.I., Keeton, R., Hsu, N.J., Bourigault, M.L., Court, N., Fremond, C., Yeremeev, V., Shebzukhov, Y., Ryffel, B., Nedospasov, S.A., Quesniaux, V.F., Jacobs, M., 2013. Prominent role for T cell-derived tumour necrosis factor for sustained control of Mycobacterium tuberculosis infection, Scientific Reports, 3, 1809.
Caswell, J.L., Williams, K.J., 2007. Respiratory System. In: M.G. Maxie (ed), Jubb, Kennedy, and Palmer’s Pathology of domestic animals, 2007. (Elsevier Saunders, Edinburgh; New York), 610–612.
Dedieu, L., Balcer-Rodrigues, V., Yaya, A., Hamadou, B., Cisse, O., Diallo, M., Niang, M., 2005. Gamma interferon-producing CD4 T-cells correlate with resistance to Mycoplasma mycoides subsp. mycoides S.C. infection in cattle, Veterinary Immunology and Immunopathology, 107, 217–233.
Fischer, A., Santana-Cruz, I., Hegerman, J., Gourle, H., Schieck, E., Lambert, M., Nadendla, S., Wesonga, H., Miller, R.A., Vashee, S., Weber, J., Meens, J., Frey, J., Jores, J., 2015. High quality draft genomes of the Mycoplasma mycoides subsp. mycoides challenge strains Afadé and B237, Standarts in Genomic Sciences, 10, 89.
Fischer, A., Shapiro, B., Muriuki, C., Heller, M., Schnee, C., Bongcam-Rudloff, E., Vilei, E.M., Frey, J., Jores, J., 2012. The Origin of the ‘Mycoplasma mycoides Cluster’ Coincides with Domestication of Ruminants, PloS ONE, 7, e36150.
Grieco, V., Boldini, M., Luini, M., Finazzi, M., Mandelli, G., Scanziani, E., 2001. Pathological, immunohistochemical and bacteriological findings in kidneys of cattle with contagious bovine pleuropneumonia (CBPP), Journal of Comparative Pathology, 124, 95–101.
Hamada, H., Bassity, E., Flies, A., Strutt, T.M., Garcia-Hernandez Mde, L., McKinstry, K.K., Zou, T., Swain, S.L., Dutton, R.W., 2013. Multiple redundant effector mechanisms of CD8+ T cells protect against influenza infection, Journal of Immunology, 190, 296–306.
Jores, J., Mariner, J.C., Naessens, J., 2013. Development of an improved vaccine for contagious bovine pleuropneumonia: an African perspective on challenges and proposed actions, Veterinary Research, 44, 122
Jores, J., Nkando, I., Sterner-Kock, A., Haider, W., Poole, J., Unger, H., Muriuki, C., Wesonga, H., Taracha, E.L., 2008. Assessment of in vitro interferon-gamma responses from peripheral blood mononuclear cells of cattle infected with Mycoplasma mycoides ssp. mycoides small colony type, Veterinary Immunology and Immunopathology, 124, 192–197.
Jungi, T.W., Krampe, M., Sileghem, M., Griot, C., Nicolet, J., 1996. Differential and strain-specific triggering of bovine alveolar macrophage effector functions by mycoplasmas. Microbial Pathogenesis, 21, 487–498.
Li, C., Yang, P., Sun, Y., Li, T., Wang, C., Wang, Z., Zou, Z., Yan, Y., Wang, W., Wang, C., Chen, Z., Xing, L., Tang, C., Ju, X., Guo, F., Deng, J., Zhao, Y., Yang, P., Tang, J., Wang, H., Zhao, Z., Yin, Z., Cao, B., Wang, X., Jiang, C., 2012. IL-17 response mediates acute lung injury induced by the 2009 pandemic influenza A (H1N1) virus, Cell Research, 22, 528–538.
McAleer, J.P., Kolls, J.K., 2014. Directing traffic: IL-17 and IL-22 coordinate pulmonary immune defense, Immunological Reviews, 260, 129–144.
Mulongo, M., Frey, J., Smith, K., Schnier, C., Wesonga, H., Naessens, J., McKeever, D., 2015. Vaccination of cattle with the N terminus of LppQ of Mycoplasma mycoides subsp. mycoides results in type III immune complex disease upon experimental infection, Infection and Immunity, 83, 1992–2000.
Newcomb, D.C., Boswell, M.G., Sherrill, T.P., Polosukhin, V.V., Boyd, K.L., Goleniewska, K., Brody, S.L., Kolls, J.K., Adler, K.B., Peebles, R.S., Jr., 2013. IL-17A induces signal transducers and activators of transcription-6-independent airway mucous cell metaplasia, American Journal of Respiratory Cell and Molecular Biology, 48, 711–716.
Nicholas, R.A., Ayling, R.D., McAuliffe, L., 2009. Vaccines for Mycoplasma diseases in animals and man, Journal of Comparative Pathology, 140, 85–96.
Sacchini, F., Luciani, M., Salini, R., Scacchia, M., Pini, A., Lelli, R., Naessens, J., Poole, J., Jores, J., 2012. Plasma levels of TNF-alpha, IFN-gamma, IL-4 and IL-10 during a course of experimental contagious bovine pleuropneumonia, BMC Veterinary Research, 8, 44.
Sacchini, F., Naessens, J., Awino, E., Heller, M., Hlinak, A., Haider, W., Sterner-Kock, A., Jores, J., 2011. A minor role of CD4+ T lymphocytes in the control of a primary infection of cattle with Mycoplasma mycoides subsp. mycoides, Veterinary Research, 42, 77.
Scanziani, E., Paltrinieri, S., Boldini, M., Grieco, V., Monaci, C., Giusti, A.M., Mandelli, G., 1997. Histological and immunohistochemical findings in thoracic lymph nodes of cattle with contagious bovine pleuropneumonia, Journal of Comparative Pathology, 117, 127–136.
Schieck, E., Liljander, A., Hamsten, C., Gicheru, N., Scacchia, M., Sacchini, F., Heller, M., Schnee, C., Sterner-Kock, A., Hlinak, A., Naessens, J., Poole, J., Persson, A., Jores, J., 2014. High antibody titres against predicted Mycoplasma surface proteins do not prevent sequestration in infected lung tissue in the course of experimental contagious bovine pleuropneumonia, Veterinary Microbiology, 172, 285–293.
Ssematimba, A., Jores, J., Mariner, J.C., 2015. Mathematical modelling of the transmission dynamics of contagious bovine pleuropneumonia reveals minimal target profiles for improved vaccines and diagnostic assays, PloS ONE, 10, e0116730.
Strafuss, A.C., 1988. Necropsy: procedures and basic diagnostic methods for practicing veterinarians, (C.C. Thomas, Springfield, Ill., USA).
Thiaucourt, F., Aboubakar, Y., Wesonga, H., Manso-Silvan, L., Blanchard, A., 2004a. Contagious bovine pleuropneumonia vaccines and control strategies: recent data, Developmental Biology (Basel), 119, 99–111.
Thiaucourt, F., Van der Lugt, J.J., Provost, A., 2004b. Contagious bovine pleuropneumonia. In: J.A.W. Coetzer and R.C. Tustin (eds), Infectious diseases of livestock, 2004b, (Oxford University Press, Oxford), 2045–2059.
Totte, P., Duperray, C., Dedieu, L., 2010. CD62L defines a subset of pathogen-specific bovine CD4 with central memory cell characteristics, Developmental and Comparative Immunology, 34, 177–182.
Totte, P., Rodrigues, V., Yaya, A., Hamadou, B., Cisse, O., Diallo, M., Niang, M., Thiaucourt, F., Dedieu, L., 2008. Analysis of cellular responses to Mycoplasma mycoides subsp. mycoides small colony biotype associated with control of contagious bovine pleuropneumonia, Veterinary Research, 39, 8.
Weldearegay, Y.B., Pich, A., Schieck, E., Liljander, A., Gicheru, N., Wesonga, H., Thiaucourt, F., Kiirika, L.M., Valentin-Weigand, P., Jores, J., Meens, J., 2016. Proteomic characterization of pleural effusion, a specific host niche of Mycoplasma mycoides subsp. mycoides from cattle with contagious bovine pleuropneumonia (CBPP), Journal of Proteomics, 131, 93–103.
Acknowledgments
This work was funded by the German Federal Ministry for Economic Cooperation and Development (Project No: 05.7860.9-001.00, contract No: 81099876 and project No 09.7860.1-001.00, contract No: 81121408). Anne Liljander was supported by Centre for International Migration and Development (CIM). Additional financial support was received from the CGIAR research program Livestock and Fish. We thank Angie Colston, Phil Toye and Richard Bishop for their valuable comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of animal rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. This article does not contain any studies with human participants.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary File 1
Comparison of fibrinous and non-fibrinous CBPP lesions with respect to the presence of γδ T-cells, monocytes, granulocytes, CD8+ T-cells, neutrophils and B-cells (XLSX 21 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Sterner-Kock, A., Haider, W., Sacchini, F. et al. Morphological characterization and immunohistochemical detection of the proinflammatory cytokines IL-1β, IL-17A, and TNF-α in lung lesions associated with contagious bovine pleuropneumonia. Trop Anim Health Prod 48, 569–576 (2016). https://doi.org/10.1007/s11250-016-0994-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-016-0994-9