Abstract
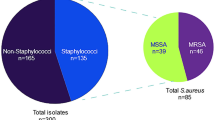
Biofilm formation in Staphylococcus aureus is considered an important virulence factor in bovine mastitis. Intercellular adhesion gene A (icaA) is a significant genetic determinant that contributes in biofilm formation. The aim of the present study was to determine the presence of the icaA gene in S. aureus isolated from bovine mastitis from seven states of India. A total of 88 out of 150 Staphylococcus aureus strains were found to be positive for biofilm marker icaA gene by PCR. The icaA gene was confirmed by dot blot hybridization in 41 of 150 S. aureus strains tested. Results obtained with dot blot hybridization were comparable to those obtained with PCR. Partial sequences of the icaA gene of the two S. aureus isolates showed deletion of some bases in different positions that might reduce/stop transcription leading to no biofilm formation. PCR was found to be a rapid test but dot blot hybridization was more accurate than PCR for detection of icaA genes. This study showed that detection of biofilm marker the icaA gene in S. aureus would allow the detection of virulence factors present in mastitis and early application of corrective measures.



Similar content being viewed by others
References
Anand Kumar, P., 2009. Evaluation of PCR test for detecting major pathogens of bubaline mastitis directly from mastitic milk samples of buffaloes. Tropical Animal Health and Production, 41, 1643–1651
Arciola, C.R., Baldassarri, L., Montanaro, L., 2001. Presence of ica A and ica D genes and slime production in a collection of staphylococcal strains from catheter-associated infections. Journal of Clinical Microbiology, 39, 2151–2156
Coelho, S.M., Pereira, I.A., Soares, L.C., Pribul, B.R., Souza, M.M., 2011. Short communication: profile of virulence factors of Staphylococcus aureus isolated from subclinical bovine mastitis in the state of Rio de Janeiro, Brazil. Journal of Dairy Science, 94, 3305–3310
Costerton, J.W., Stewart, P.S., Greenberg, E.P., 1999. Bacterial biofilms: a common cause of persistent infections. Science, 284, 1318–1322
Cowan, S.T., Steel, K.J., 1970. Manual for the Identification of Medical Bacteria. The Syndics of the Cambridge University Press, London
Cramton, S.E., Gerke, C., Schnell, N.F., Nichols, W.W., Gotz, F., 1999. The intercellular adhesion (ica) locus is present in Staphylococcus aureus and is required for biofilm formation. Infection and Immunity, 67, 5427–5433
Cruickshank, R., Duguid, J.P., Marmoin, B.P., Swain, R.H.A., 1975. Medical Microbiology vol. 2, 12th edn. Churchill Livingstone, Edinburgh
Cucarella, C., Solano, C., Valle, J., Amorena, B., Lasa, I., Penades, J. R., 2001. Bap, a Staphylococcus aureus surface protein involved in biofilm formation. Journal of Bacteriology, 183, 2888–2896
Dhanawade, N.B., Kalorey, D.R., Srinivasan, R., Barbuddhe, S.B., Kurkure, N.V., 2010. Detection of intercellular adhesion genes and biofilm production in Staphylococcus aureus isolated from bovine subclinical mastitis. Veterinary Research Communications, 34, 81–89.
Fijalkowski, K., Czernomysy-Furowicz, D., Irwin, J.A., Nawrotek, P., Pobucewicz, A., 2011. Secretory virulence factors produced by Staphylococcus aureus isolates obtained from mastitic bovine milk—effect on bovine polymorphonuclear neutrophils. Research in Veterinary Science, doi:10.1016/j.rvsc.2011.08.017
Fox, L.K., Zadoks, R.N., Gaskins, C.T., 2005. Biofilm production by Staphylococcus aureus associated with intramammary infection. Veterinary Microbiology, 107, 295–299
Hamann, J., 2005. Diagnosis of mastitis and indicators of milk quality. In: Hogeveen, H. (eds) Mastitis in dairy production: current knowledge and future solutions. Wageningen Academic, Wageningen, 82–90
Hogeveen, H., Østerås, O., 2005. Mastitis management in an economic framework. In: Hogeveen, H. (ed), Proceedings of the 4th International Mastitis Conference, Maastrich, The Netherlands, 41–52
Kalorey, D.R., Yuvaraj, S., Kurkure, N.V., Chousalkar, K.K., Barbuddhe, S.B., 2007. PCR-based detection of genes encoding virulence determinants in Staphylococcus aureus from bovine subclinical mastitis. Journal of Veterinary Science, 8, 151–154
Kuang, Y., Tani, K., Synnott, A. J., Ohshima, K., Higuchi, H., Nagahata, H., Tanji, Y., 2009. Characterization of bacterial population of raw milk from bovine mastitis by culture-independent PCR–DGGE method. Biochemical Engineering Journal, 45, 76–81.
Little, C. L., Rhoades, J. R., Sagoo, S. K., Harris, J., Greenwood, M., Mithani, V., Grant, K., McLauchlin, J., 2008. Microbiological quality of retail cheeses made from raw, thermized or pasteurized milk in the UK. Food Microbiology, 25, 304–312
Mehrzad, J., Duchateau, L., Burvenich, C., 2009. Phagocytic and bactericidal activity of blood and milk-resident neutrophils against Staphylococcus aureus in primiparous and multiparous cows during early lactation. Veterinary Microbiology, 134, 106–112
Melchior, M.B., Vaarkamp, H., Fink-Gremmels, J., 2006. Biofilms: a role in recurrent mastitis infections? The Veterinary Journal, 171, 398–407
Melchior, M.B., Fink-Gremmels, J., Gaastra, W., 2007. Extended antimicrobial susceptibility assay for Staphylococcus aureus isolates from bovine mastitis growing in biofilms. Veterinary Microbiology, 125, 141–149
Moretro, T., Hermansen, L., Holck, A.L., Sidhu, M.S., Rudi, K., Langsrud, S., 2003. Biofilm formation and the presence of the intercellular adhesion locus ica among staphylococci from food and food processing environments. Applied and Environmental Microbiology, 69, 5648–5655
National Mastitis Council, 1987. Laboratory and Field Handbook on Bovine Mastitis. National Mastitis Council Inc., Arlington, VA
Oliveira, M., Bexiga, R., Nunes, S.F., Carneiro, C., Cavaco, L.M., Bernardo, F., Vilela, C.L., 2006. Biofilm-forming ability profiling of Staphylococcus aureus and Staphylococcus epidermidis mastitis isolates. Veterinary Microbiology, 118, 133–40
Oliveira, L., Rodrigues, A.C., Hulland, C., Ruegg, P.L., 2011a. Enterotoxin production, enterotoxin gene distribution, and genetic diversity of Staphylococcus aureus recovered from milk of cows with subclinical mastitis. American Journal of Veterinary Research, 72, 1361–1368
Oliveira, M., Bexiga, R., Nunes, S.F., Vilela, C.L., 2011b. Invasive potential of biofilm-forming staphylococci bovine subclinical mastitis isolates. Journal of Veterinary Science, 12, 95–97
Planchon, S., Gaillard-Martinie, B., Dordet-Frisoni, E., Bellon-Fontaine, M.N., Leroy, S., Labadie, J., Hebraud, M., Talon, R., 2006. Formation of biofilm by Staphylococcus xylosus. International Journal of Food Microbiology, 109, 88–96
Rohde, H., Frankenberger, S., Zähringer, U., Mack, D., 2010. Structure, function and contribution of polysaccharide intercellular adhesin (PIA) to Staphylococcus epidermidis biofilm formation and pathogenesis of biomaterial-associated infections. European Journal of Cell Biology, 89, 103–111
Salasia, S., Khusnan, Z., Lammler, C., Zschock, M., 2004. Comparative studies on pheno- and genotypic properties of Staphylococcus aureus isolated from bovine subclinical mastitis in central Java in Indonesia and Hesse in Germany. Journal of Veterinary Science, 5 103–109
Thurlow, L.R., Hanke, M.L., Fritz, T., Angle, A., Aldrich, A., Williams, S.H., Engebretsen, I.L., Bayles, K.W., Horswill, A.R., Kielian, T., 2011. Staphylococcus aureus biofilms prevent macrophage phagocytosis and attenuate inflammation in vivo. Journal of Immunology, 186, 6585–6596
Vasudevan, P., Nair, M.M., Annamalai, T., Venkitanarayanan, K.S., 2003. Phenotypic and genotypic characterization of bovine mastitis isolates of Staphylococcus aureus for biofilm formation. Veterinary Microbiology, 92, 179–185
Vijayakumar, K., 2003. Test for detection of mastitis. In: Proceedings on Fourth Round Table on Mastitis. Indian Veterinary Research Institute, Izatnagar, India, 124–136
Wilson, K., 1987. Preparation of genomic DNA from bacteria. In: Ausubal, F. M., Brent, R., Kirston, R. L., Moore, D.D., Seidman, J. G., Smith, J. A. and Struhl, K. (eds) Current Protocols in Molecular Biology, vol. 1. Wiley, New York
Wu, X., Wang, Y., Tao, L., 2011. Sulfhydryl compounds reduce Staphylococcus aureus biofilm formation by inhibiting PIA biosynthesis. FEMS Microbiology Letters, 316, 44–50
Yazdani, R., Oshaghi, M., Havayi, A., Pishva, E., Salehi, R., Sadeghizadeh, M., Foroohesh, H., 2006. Detection of icaAD gene and biofilm formation in Staphylococcus aureus isolates from wound infection. Iranian Journal of Public Health, 35, 25–28
Zmantar, T., Chaieb, K., Makni, H., Miladi, H., Abdallah, F.B., Mahdouani, K., Bakhrouf, A., 2008. Detection by PCR of adhesins genes and slime production in clinical Staphylococcus aureus. Journal of Basic Microbiology, 48, 308–314
Acknowledgments
The authors are thankful to the Director, National Environmental Engineering Research Institute, Nagpur, India and Dr. A. G. Bhandarkar, Professor and Head, Department of Veterinary Pathology, Nagpur Veterinary College, Nagpur, India for providing the facilities to conduct this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chavhan, S.K., Kalorey, D.R., Nagdive, A.A. et al. Molecular characterization of intercellular adhesion gene in Staphylococcus aureus isolated from bovine mastitic milk. Trop Anim Health Prod 44, 247–252 (2012). https://doi.org/10.1007/s11250-011-0009-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-011-0009-9