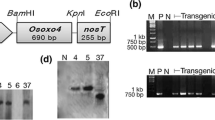
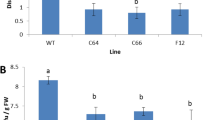
Abstract
Sclerotinia Stem Rot (SSR) caused by the oxalic acid (OA)-secreting necrotrophic fungal pathogen Sclerotinia sclerotiorum, causes significant yields losses in the crop Brassica sps. Oxalate oxidase (OxO) can metabolize OA to CO2 and H2O2. Degradation of OA during the early phase of fungal–host interaction can interfere with the fungal infection and establishment processes. The present study demonstrates the potential of barley oxalate oxidase (BOxO) gene in conferring stable resistance against stem rot in a productive and highly susceptible Brassica juncea cv Varuna under field conditions. Four stable, independent, single-copy transgenic lines (B16, B17, B18, and B53) exhibited a significant reduction in the rate of lesion expansion i.e. 11–26%, 39–47%, and 24–35% reproducibly over the three-generation i.e. T2, T3, and T4 respectively. The enhanced resistance in the transgenic lines correlated with high OxO activity, accumulation of higher levels of H2O2, and robust activation of defense responsive genes upon infection by S. sclerotiorum.





Similar content being viewed by others
Abbreviations
- bp:
-
Base pairs
- bar:
-
Bialaphos resistance
- CaMV:
-
Cauliflower Mosaic Virus
- cDNA:
-
Complementary DNA
- SCF:
-
Sclerotinia culture filtrates
- cm:
-
Centimeter
- cv:
-
Cultivar
- DAB:
-
3,3′-Diaminobenzidine
- dpi:
-
Days post infection
- hpi:
-
Hours post infection
- L:
-
Litre
- mg:
-
Milligram
- O/N:
-
Overnight
- ROS:
-
Reactive oxygen species
- RT-PCR:
-
Reverse transcription PCR
- T1/T2/T3/T4 :
-
Transgenic generation 1,2,3,4
- T-DNA:
-
Transfer DNA
- WT:
-
Wild type
- V/v:
-
Volume/volume
References
Bhullar S, Chakravarthy S, Advani S, Datta S, Pental D, Burma PK (2003) Strategies for development of functionally equivalent promoters with minimum sequence homology for transgene expression in plants: cis-elements in a novel DNA context versus domain swapping. Plant Physiol 132(2):988–998
Bolton MD, Thomma BPHJ, Nelson BD (2006) Sclerotinia sclerotiorum (Lib.) de Bary: biology and molecular traits of a cosmopolitan pathogen. Mol Plant Pathol 7(1):1–16. https://doi.org/10.1111/j.1364-3703.2005.00316.x
Cessna SG, Sears VE, Dickman MB, Low PS (2000) Oxalic acid, a pathogenicity factor for Sclerotinia sclerotiorum, suppresses the oxidative burst of the host plant. Plant Cell 12(11):2191–2199
Chen X, Chen F, Chen L, Zheng L, Lu G, Wang Z (2008) Isolation and analysis of oxalic acid-insensitive mutant of Arabidopsis thaliana. Chin J Biotechnol 24(2):203–208. https://doi.org/10.1016/S1872-2075(08)60011-4
Davidson RM, Reeves PA, Manosalva PM, Leach JE (2009) Germins: a diverse protein family important for crop improvement. Plant Sci 177(6):499–510
Dickman MB, Mitra A (1992) Arabidopsis thaliana as a model for studying Sclerotinia sclerotiorum pathogenesis. Physiol Mol Plant Pathol 41(4):255–263. https://doi.org/10.1016/0885-5765(92)90025-Q
Dong X, Ji R, Guo X, Foster SJ, Chen H, Dong C, Liu Y, Hu Q, Liu S (2008) Expressing a gene encoding wheat oxalate oxidase enhances resistance to Sclerotinia sclerotiorum in oilseed rape (Brassica napus). Planta 228(2):331
Dumas B, Freyssinet G, Pallett KE (1995) Tissue-Specific expression of germin-like oxalate oxidase during development and fungal infection of barley seedlings. Plant Physiol 107(4):1091. https://doi.org/10.1104/pp.107.4.1091
Dunwell JM, Gibbings JG, Mahmood T, Saqlan Naqvi S (2008) Germin and germin-like proteins:evolution, structure, and function. Crit Rev Plant Sci 27:342–375
Guimarães RL, Stotz HU (2004) Oxalate production by Sclerotinia sclerotiorum deregulates guard cells during infection. Plant Physiol 136(3):3703–3711. https://doi.org/10.1104/pp.104.049650
Hu X, Bidney DL, Yalpani N, Duvick JP, Crasta O, Folkerts O, Lu G (2003) Overexpression of a gene encoding hydrogen peroxide-generating oxalate oxidase evokes defense responses in sunflower. Plant Physiol 133(1):170–181
Ilyas M, Rasheed A, Mahmood T (2016) Functional characterization of germin and germin-like protein genes in various plant species using transgenic approaches. Biotechnol Lett 38(9):1405–1421
Kabbage M, Yarden O, Dickman MB (2015) Pathogenic attributes of Sclerotinia sclerotiorum: switching from a biotrophic to necrotrophic lifestyle. Plant Sci 233:53–60
Kanoria S, Burma PK (2012) A 28 nt long synthetic 5′UTR (synJ) as an enhancer of transgene expression in dicotyledonous plants. BMC Biotechnol 12(1):85
Kheiri H-R, Motallebi M, Zamani MR, Deljo A (2014) Beta glucanase (Bgn13. 1) expressed in transgenic Brassica napus confers antifungal activity against Sclerotinia sclerotiorum. J Crop Prot 3(1):31–42
Kim KS, Min JY, Dickman MB (2008) Oxalic acid is an elicitor of plant programmed cell death during Sclerotinia sclerotiorum disease development. Mol Plant Microbe Interact 21(5):605–612. https://doi.org/10.1094/mpmi-21-5-0605
Lamb C, Dixon RA (1997) The oxidative burst in plant disease resistance. Annu Rev Plant Biol 48(1):251–275
Lane BG (2002) Oxalate, germins, and higher-plant pathogens. Rev Art IUBMB Life 53:67–75
Liu F, Wang M, Wen J, Yi B, Shen J, Ma C, Tu J, Fu T (2015) Overexpression of barley oxalate oxidase gene induces partial leaf resistance to Sclerotinia sclerotiorum in transgenic oilseed rape. Plant Pathol 64(6):1407–1416
Mehra S, Pareek A, Bandyopadhyay P, Sharma P, Burma PK, Pental D (2000) Development of transgenics in Indian oilseed mustard (Brassica juncea) resistant to herbicide phosphinothricin. Curr Sci Bangalore 78(11):1358–1364
Molla KA, Karmakar S, Chanda PK, Ghosh S, Sarkar SN, Datta SK, Datta K (2013) Rice oxalate oxidase gene driven by green tissue-specific promoter increases tolerance to sheath blight pathogen (Rhizoctonia solani) in transgenic rice. Mol Plant Pathol 14(9):910–922
Nagarajkumar M, Jayaraj J, Muthukrishnan S, Bhaskaran R, Velazhahan R (2005) Detoxification of oxalic acid by Pseudomonas fluorescens strain PfMDU2: implications for the biological control of rice sheath blight caused by Rhizoctonia solani. Microbiol Res 160(3):291–298. https://doi.org/10.1016/j.micres.2005.02.002
Pei Y, Li X, Zhu Y, Ge X, Sun Y, Liu N, Jia Y, Li F, Hou Y (2019) GhABP19, a novel germin-like protein from Gossypium hirsutum, plays an important role in the regulation of resistance to verticillium and fusarium wilt pathogens. Front Plant Sci 10:583
Rana K, Atri C, Akhatar J, Kaur R, Goyal A, Singh MP, Kumar N, Sharma A, Sandhu PS, Kaur G, Barbetti MJ, Banga SS (2019) Detection of first marker trait associations for resistance against Sclerotinia sclerotiorum in Brassica juncea–Erucastrum cardaminoides introgression lines. Front Plant Sci. https://doi.org/10.3389/fpls.2019.01015
Schoonbeek H-j, Jacquat-Bovet A-C, Mascher F, Métraux J-P (2007) Oxalate-degrading bacteria can protect Arabidopsis thaliana and crop plants against Botrytis cinerea. Mol Plant Microbe Interact 20(12):1535–1544
Solgi T, Moradyar M, Zamani MR, Motallebi M (2015) Transformation of canola by chit33 gene towards improving resistance to Sclerotinia sclerotiorum. Plant Prot Sci 51(1):6–12
Telenko D, Hu J, Livingstone D, Shew B, Phipps P, Grabau E (2011) Sclerotinia blight resistance in virginia-type peanut transformed with a barley oxalate oxidase gene. Phytopathology 101:786–793. https://doi.org/10.1094/PHYTO-10-10-0266
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley—powdery mildew interaction. Plant J 11(6):1187–1194
Walz A, Zingen-Sell I, Loeffler M, Sauer M (2008) Expression of an oxalate oxidase gene in tomato and severity of disease caused by Botrytis cinerea and Sclerotinia sclerotiorum. Plant Pathol 57:453–458. https://doi.org/10.1111/j.1365-3059.2007.01815
Wang Z, Mao H, Dong C, Ji R, Cai L, Fu H, Liu S (2009) Overexpression of Brassica napus MPK4 enhances resistance to Sclerotinia sclerotiorum in oilseed rape. Mol Plant Microbe Interact 22(3):235–244
Wang Z, Bao L-L, Zhao F-Y, Tang M-Q, Chen T, Li Y, Wang B-X, Fu B, Fang H, Li G-Y (2019) BnaMPK3 is a key regulator of defense responses to the devastating plant pathogen Sclerotinia sclerotiorum in oilseed rape. Front Plant Sci 10:91
Wei D, Mei J, Fu Y, Disi JO, Li J, Qian W (2014) Quantitative trait loci analyses for resistance to Sclerotinia sclerotiorum and flowering time in Brassica napus. Mol Breed 34(4):1797–1804
Williams B, Kabbage M, Kim H-J, Britt R, Dickman MB (2011) Tipping the balance: Sclerotinia sclerotiorum secreted oxalic acid suppresses host defenses by manipulating the host redox environment. PLoS Pathog 7(6):e1002107
Woo E-J, Dunwell JM, Goodenough PW, Marvier AC, Pickersgill RW (2000) Germin is a manganese containing homohexamer with oxalate oxidase and superoxide dismutase activities. Nat Struct Biol 7(11):1036–1040
Yang X, Yang J, Wang Y, He H, Niu L, Guo D, Xing G, Zhao Q, Zhong X, Sui L (2019) Enhanced resistance to Sclerotinia stem rot in transgenic soybean that overexpresses a wheat oxalate oxidase. Transgenic Res 28(1):103–114
Yin X, Yi B, Chen W, Zhang W, Tu J, Fernando WD, Fu T (2010) Mapping of QTLs detected in a Brassica napus DH population for resistance to Sclerotinia sclerotiorum in multiple environments. Euphytica 173(1):25–35
Zhang Y, Wang X, Chang X, Sun M, Zhang Y, Li W, Li Y (2018) Overexpression of germin-like protein GmGLP10 enhances resistance to Sclerotinia sclerotiorum in transgenic tobacco. Biochem Biophys Res Commun. https://doi.org/10.1016/j.bbrc.2018.02.046
Zhao J, Meng J (2003) Genetic analysis of loci associated with partial resistance to Sclerotinia sclerotiorum in rapeseed (Brassica napus L.). Theor Appl Genet 106(4):759–764
Zhou F, Zhang Z, Gregersen PL, Mikkelsen JD, de Neergaard E, Collinge DB, Thordal-Christensen H (1998) Molecular characterization of the oxalate oxidase involved in the response of barley to the powdery mildew fungus. Plant Physiol 117(1):33. https://doi.org/10.1104/pp.117.1.33
Acknowledgements
The Project was funded by the grant from the Department of Biotechnology (COE Project)-BT/01/COE/08/06-II and fellowship to RV from the UGC Government of India. We are thankful to Prof. Deepak Pental for his interest in work and discussion throughout. We extend our thanks to Dr. Sivasubramanian for his help with statistical analysis. BS Yadav is acknowledged for his help in the tissue culture experiments. The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the Supplementary Information.
Rights and permissions
About this article
Cite this article
Verma, R., Kaur, J. Expression of barley oxalate oxidase confers resistance against Sclerotinia sclerotiorum in transgenic Brassica juncea cv Varuna. Transgenic Res 30, 143–154 (2021). https://doi.org/10.1007/s11248-021-00234-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-021-00234-1