Abstract
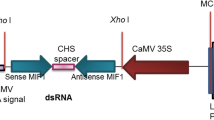
RNA interference (RNAi) describes the ability of double-stranded RNA (dsRNA) to inhibit homologous gene expression at the RNA level. Its specificity is sequence-based and depends on the sequence of one strand of the dsRNA corresponding to part or all of a specific gene transcript. In this study we adopted plant-mediated RNAi technology that targets Sitobion avenae (S. avenae) to enable gene silencing in the aphid and to minimize handling of the insects during experiments. S. avenae was selected for this study because it causes serious economic losses to wheat throughout the world. The carboxylesterase (CbE E4) gene in S. avenae was homologously cloned, which increased synthesis of a protein known to be critical to the resistance (tolerance) this species has developed to a wide range of pesticides. A plant RNAi vector was constructed, and transgenic Triticum aesticum (dsCbE1-5 and dsCbE2-2 lines) expressing CbE E4 dsRNA were developed. S. avenae were fed on dsCbE1-5 and dsCbE2-2 lines stably producing the CbE E4 dsRNA. CbE E4 gene expression in S. avenae was reduced by up to 30–60 %. The number of aphids raised on dsCbE1-5 and dsCbE2-2 was lower than the number raised on non-transgenic plants. A solution of CbE E4 enzyme from S. avenae fed on dsCbE1-5 and dsCbE2-2 plants hydrolyzed only up to 20–30 % Phoxim solution within 40 min whereas a solution of the enzyme from CbE E4 fed on control plants hydrolyzed 60 % of Phoxim solution within 40 min. CbE E4 gene silencing was achieved by our wheat-mediated RNAi approach. This plant-mediated RNAi approach for addressing degradation-based pesticide resistance mechanisms in aphids and may prove useful in pest management for diverse agro-ecosystems.




Similar content being viewed by others
Abbreviations
- CbE E4 gene:
-
Carboxylesterase gene
- RNAi:
-
RNA interference
- PCR:
-
Polymerase chain reaction
- qRT-PCR:
-
Quantitative real-time PCR
- S. avenae :
-
Sitobion avenae (Fabricus)
References
Aravin AA, Naumova NM, Tulin AV, Vagin VV, Rozovsky YM, Gvozdev VA (2001) Double-stranded RNA-mediated silencing of genomic tandem repeats and transposable elements in the D. melanogaster germline. Curr Biol 11(13):1017–1027
Baum JA, Bogaert T, Clinton W, Heck GR, Feldmann P, Ilagan O, Johnson S, Plaetinck G, Munyikwa T, Pleau M, Vaughn T, Roberts J (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25(11):1322–1326
Blain PG (1990) Aspects of pesticide toxicology. Adverse Drug React Acute Poisoning Rev 9(1–2):37–68
Brown P, Charlton A, Cuthbert M, Barnett L, Ross L, Green M, Gillies L, Shaw K, Fletcher M (1996) Identification of pesticide poisoning in wildlife. J Chromatogr A 754(1–2):463–478
Chen J, Zhang D, Yao Q, Zhang J, Dong X, Tian H, Chen J, Zhang W (2010) Feeding-based RNA interference of a trehalose phosphate synthase gene in the brown planthopper, Nilaparvata lugens. Insect Mol Biol 19(6):777–786
Chuang CF, Meyerowitz EM (2000) Specific and heritable genetic interference by double-stranded RNA in Arabidopsis thaliana. Proc Natl Acad Sci 97(9):4985–4990
Devonshire AL (1977) The properties of a carboxylesterase from the peach-potato aphid, Myzus Persicae (Sulze.), and its role in conferring insecticide resistance. Biochem J 167(3):675–683
Devonshire AL, Moores GD (1982) A carboxylesterase with broad substrate specificity causes organophosphorus, carbamate and pyrethroid resistance in peach–potato aphids (Myzus Persicae). Pestic Biochem Physiol 18(2):235–246
Hayatsu M, Nagata T (1993) Purification and characterization of Carbaryl Hydrolase from Blastobacter sp. M501. Appl Environ Microbiol 597:2121–2125
Jauber-Possamai S, Trionnaire GL, Bonhomme J, Christophides GK, Rispe C, Tagu D (2007) Gene knockdown by RNAi in the pea aphid Acyrthosiphon pisum. BMC Biotechnol 63(7):1–8
Lan WS, Cong J, Jiang H, Jiang SR, Qiao CL (2005) Expression and characterization of carboxylesterase E4 gene from peach–potato aphid (Myzus persicae) for degradation of Carbaryl and Malathion. Biotechnol Lett 27(15):1141–1146
Leng XF, Qiao CL (1986) The biodegradation of the Decis, Acetofenate (7504) and DDT by the perfusion isolated rat cell (in Chines). Sci Bull 19:1505–1508
Liu Y, Wang WL, Guo GX, Ji XL (2009) Volatile emission in wheat and parasitism by Aphidius avenae after exogenous application of salivary enzymes of Sitobion avenae. Entomol Exp Appl 130(3):215–221
Mao JJ, Zeng FR (2013) Plant-mediated RNAi of a gap gene-enhanced tobacco tolerance against the Myzus persica. Transgenic Res. doi:10.1007/s11248-01309739-y
Mao YB, Cai WJ, Wang JW, Hong GJ, Tao XY, Wang LJ, Huang YP, Chen XY (2007) Silencing a cotton bollworw P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 11(25):1307–1313
Marco A, Griffiths-Jones S (2012) Detection of microRNAs in color space. Bioinformatics 28(3):318–323
Mlotshwa S, Voinnet O, Mette MF, Matzke M, Matzke M, Vaucheret H, Ding SW, Pruss G, Vance VB (2002) RNA silencing and the mobile silencing signal. Plant Cell 14:5289–5301
Mutti NS, Park Y, Reese JC, Reeck GR (2006) RNAi knockdown of a salivary transcript leading to lethality in the pea aphid, Acyrthosiphon pisum. J Insect Sci 38(6):1–7
Pitino M, Coleman AD, Maffei ME, Ridout CJ, Hogenhout SA (2011) Silencing of aphid genes by dsRNA feeding from plants. PLoS One 6(10):1–8
Pons L (2004) Germplasm from previous study may thwart new aphid biotype. Agric Res 4(52):16–18
Smith NA, Singh SP, Wang M, Stoutjesdijk PA, Green AG, Waterhouse PM (2000) Total silencing by intron-spliced hairpin RNAs. Nature 407:319–320
Sogorb MA, Vilanova E (2002) Enzymes involved in the detoxification of organophosphorus, carbamate and pyrethroid insecticides through hydrolysis. Toxicol Lett 128(1–3):215–228
Tabara H, Sarkissian M, Kelly WG, Fleenor J, Grishok A, Timmons L, Fire A, Mello CC (1999) The rde-1 gene, RNA interference, and transposon silencing in C. elegans. Cell 99(2):123–132
Van Asperen K (1962) A study of housefly asterases by means of a sensitive colorimetric method. J Insect Physiol 8:401–416
Vasil V, Castillo AM, Fromm ME, Vasil IK (1992) Herbicide resistant fertile transgenic wheat plants obtained by microprojectile Bombardment of Regenerable embryogenic callus. Nat Biotechnol 10:667–674
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Regul 19(2):195–216
Wang H, Zhang M, Zhang XH, Xian LQ (2012) Silencing of cytochrome P45o in Sitobion avenae and Myzus persicae through RNA interference. China Agric Sci 17(45):3463–3472
Wesley SV, Helliwell CA, Smith NA, Wang M, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, Robinson SP, Gleave AP, Green AG, Waterhouse PM (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27(6):581–590
Acknowledgments
This research was financially supported by the grants from the Key Project of National Plant Transgenic Genes of China (2008ZX08002-001, 20011ZX08002-001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Lanjie Xu and Xiaoliang Duan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xu, L., Duan, X., Lv, Y. et al. Silencing of an aphid carboxylesterase gene by use of plant-mediated RNAi impairs Sitobion avenae tolerance of Phoxim insecticides. Transgenic Res 23, 389–396 (2014). https://doi.org/10.1007/s11248-013-9765-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-013-9765-9