Abstract
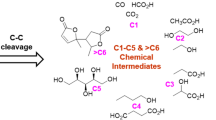
The synthesis of commodity polymers from biomass presents interesting challenges and opportunities for new catalytic chemistries. Starting from glucose, reaction pathways to a variety of monomers for commodity polymers are presented. The first step for each pathway is the isomerization of glucose to fructose. This reaction can now be accomplished in aqueous media with hydrophobic, large pore molecular sieves that contain Lewis acid sites as catalysts. A typical catalyst is a pure-silica molecular sieve having the zeolite beta topology and containing a small amount of framework Sn4+ (Sn-Beta). Dehydration of fructose into 5-hydroxymethylfurfural (HMF) provides a convenient path that can lead to 2,5-furandicarboxylic acid (FDCA) for the synthesis of polyethylene furanoate (PEF) or purified terephthalic acid (PTA) (via Diels–Alder reactions) for the synthesis of polyethylene terephthalate (PET). Recent progress on the dehydration of lactic acid could open new routes to acrylic acid and acrylonitrile that are used to produce polyacrylic acid and polyacrylonitrile, respectively, provided more selective catalysts for the retro-aldol deconstruction of fructose are developed.








Similar content being viewed by others
References
Corma A, Iborra S, Velty A (2007) Chem Rev 107:2411–2502
Dusselier M, Van Wouwe P, Dewaele A, Makshina E, Sels BF (2013) Energy Environ Sci 6:1415–1442
Vennestrom PNR, Osmundsen CM, Christensen CH, Taarning E (2011) Angew Chem Int Ed 50:10502–10509
www.braskem.com.br. Accessed 2014
Moliner M, Roman-Leshkov Y, Davis ME (2010) Proc Natl Acad Sci USA 107:6164–6168
Bermejo-Deval R, Assary RS, Nikolla E, Moliner M, Roman-Leshkov Y, Hwang SJ, Palsdottir A, Silverman D, Lobo RF, Curtiss LA, Davis ME (2012) Proc Natl Acad Sci USA 109:9727–9732
Bermejo-Deval R, Orazov M, Gounder R, Hwang SJ, Davis ME (2014) ACS Catal 4:2288–2297
Raj N, Caratzoulas S, Vlachos DG (2013) ACS Catal 3:2294–2298
Rajabbeigi N, Torres AI, Lew CM, Elyassi B, Ren L, Wang Z, Cho HJ, Fan W, Daoutidis P, Tsapatsis M (2014) Chem Eng Sci 116:235–242
Zhao H, Holladay JE, Brown H, Zhang ZC (2007) Science 316:1597–1600
Takagaki A, Ohara M, Nishimura S, Ebitani K (2009) Chem Commun 194:6276–6278
Nakajima K, Noma R, Kitano M, Hara M (2014) J Mol Catal A 388–389:100–105
Nikolla E, Roman-Leshkov Y, Moliner M, Davis ME (2011) ACS Catal 1:408–410
www.ava-biochem.com. Accessed 2014
www.avantium.com. Accessed 2014
Masuno M, Cannon D, Bissell J, Smith RL, Foster M, Wood AB, Smith PB, Hucul DA WO 2013/040514 A1
Chang CC, Green SK, Williams CL, Dauenhauer P, Fan W (2014) Green Chem 16:585–588
Lin Z, Ierapetritou M, Nikolakis V (2013) AIChE J 59:2079–2087
Pacheco JJ, Davis ME (2014) Proc Natl Acad Sci USA 111:8363–8367
Casanova O, Iborra S, Corma A (2009) ChemSusChem 2:1138–1144
Yan B, Tao LZ, Liang Y, Xu BQ (2014) ACS Catal 4:1931–1943
Peng J, Li X, Tang C, Bai W (2014) Green Chem 16:108–111
Holm MS, Saravanamurugan S, Taarning E (2010) Science 328:602–605
Lobo RF (2010) ChemSusChem 3:1237–1240
Bermejo-Deval R PhD Thesis, California Institute of Technology, 2014
Acknowledgments
I thank my coworkers who are listed on the publications referenced. Financial support for our work listed in the references was primarily from two sources: (i) work supported as part of the Catalysis Center for Energy Innovation, an Energy Frontier Research Center funded by the U. S. Department of Energy, Office of Science, Office of Basic Energy Sciences under the Award Number DE-SC1004, and (ii) Toray Industries.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davis, M.E. Heterogeneous Catalysis for the Conversion of Sugars into Polymers. Top Catal 58, 405–409 (2015). https://doi.org/10.1007/s11244-015-0386-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-015-0386-9