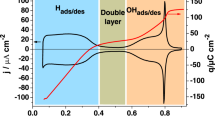
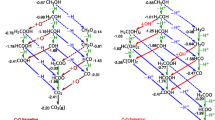
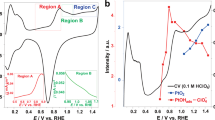
Abstract
Recent electrochemical studies have reported aqueous CO2 reduction to formic acid, formaldehyde and methanol at potentials of ca. −600 mV versus SCE, when using a Pt working electrode in acidic pyridine solutions. In those experiments, pyridinium is thought to function as a one-electron shuttle for the underlying multielectron reduction of CO2. DFT studies proposed that the critical step of the underlying reaction mechanism is the one-electron reduction of pyridinium at the Pt surface through proton coupled electron transfer. Such reaction forms a H adsorbate that is subsequently transferred to CO2 as a hydride, through a proton coupled hydride transfer mechanism where pyridinium functions as a Brønsted acid. Here, we find that imidazolium exhibits an electrochemical behavior analogous to pyridinium, as characterized by the experimental and theoretical analysis of the initial reduction on Pt. A cathodic wave, with a cyclic voltammetric half wave potential of ca. −680 mV versus SCE, is consistent with the theoretical prediction based on the recently proposed reaction mechanism suggesting that positively charged Brønsted acids could serve as electrocatalytic one-electron shuttle species for multielectron CO2 reduction.








Similar content being viewed by others
References
Kumar B, Llorente M, Froehlich J, Dang T, Sathrum A, Kubiak CP (2012) Annu Rev Phys Chem 63:541
Benson EE, Sathrum AJ, Smieja JM, Kubiak CP (2009) Chem Soc Rev 38:89
Costentin C, Robert M, Savéant J-M (2013) Chem Soc Rev 42:2423
Barton CE, Lakkaraju PS, Rampulla DM, Morris AJ, Abelev E, Bocarsly AB (2010) J Am Chem Soc 132:11539
Morris AJ, McGibbon RT, Bocarsly AB (2011) ChemSusChem 4:191
Barton EE, Rampulla DM, Bocarsly AB (2008) J Am Chem Soc 130:6342
Kamrath MZ, Relph RA, Johnson MA (2010) J Am Chem Soc 132:15508
Boston DJ, Xu C, Armstrong DW, MacDonnell FM (2013) J Am Chem Soc 135:16252
Yan Y, Zeitler EL, Gu J, Hu Y, Bocarsly AB (2013) J Am Chem Soc 135:14020
Keith JA, Carter EA (2012) J Am Chem Soc 134:7580
Keith JA, Carter EA (2013) Chem Sci 4:1490
Lim C-H, Holder AM, Musgrave CB (2012) J Am Chem Soc 135:142
Ertem MZ, Konezny SJ, Araujo CM, Batista VS (2013) J Phys Chem Lett 4:745
Barrette WC, Johnson HW, Sawyer DT (1890) Anal Chem 1984:56
Costentin C, Canales JC, Haddou B, Saveant J-M (2013) J Am Chem Soc 135:17671
Canhoto C, Matos M, Rodrigues A, Geraldo MD, Bento MF (2004) J Electroanal Chem 570:63
Peremans A, Tadjeddine A (1995) J Chem Phys 103:7197
Tian Z-Q, Ren B (2004) Annu Rev Phys Chem 55:197
Conway BE, Jerkiewicz G (2002) Solid State Ionics 150:93
Ren B, Xu X, Li XQ, Cai WB, Tian ZQ (1999) Surf Sci 427–428:157
Zolfaghari A, Chayer M, Jerkiewicz G (1997) J Electrochem Soc 144:3034
Clavilier J, Armand D (1986) J Electroanal Chem Interfacial Electrochem 199:187
Barber J, Morin S, Conway BE (1998) J Electroanal Chem 446:125
Conway BE, Barber J, Morin S (1998) Electrochim Acta 44:1109
Skulason E, Karlberg GS, Rossmeisl J, Bligaard T, Greeley J, Jonsson H, Norskov JK (2007) Phys Chem Chem Phys 9:3241
Skulason E, Tripkovic V, Bjorketun ME, Gudmundsdottir S, Karlberg G, Rossmeisl J, Bligaard T, Jonsson H, Norskov JK (2010) J Phys Chem C 114:18182
Nanbu N, Kitamura F, Ohsaka T, Tokuda K (2000) J Electroanal Chem 485:128
Wiberg KB (1955) Chem Rev 55:713
Oh Y, Hu X (2013) Chem Soc Rev 42:2253
Morris AJ, Meyer GJ, Fujita E (1983) Acc Chem Res 2009:42
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865
Giannozzi P, Baroni S, Bonini N, Calandra M, Car R, Cavazzoni C, Ceresoli D, Chiarotti GL, Cococcioni M, Dabo I, Dal Corso A, de Gironcoli S, Fabris S, Fratesi G, Gebauer R, Gerstmann U, Gougoussis C, Kokalj A, Lazzeri M, Martin-Samos L, Marzari N, Mauri F, Mazzarello R, Paolini S, Pasquarello A, Paulatto L, Sbraccia C, Scandolo S, Sclauzero G, Seitsonen AP, Smogunov A, Umari P, Wentzcovitch RM (2009) J Phys-Condens Matter 21:395
Dolg M, Wedig U, Stoll H, Preuss H (1987) J Chem Phys 86:866–872
Hehre WJ, Radom L, PvR Schleyer, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2010) Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford, CT
Cramer CJ (2004) Essentials of computational chemistry: theories and models, 2nd edn. Wiley, Chichester
Marenich AV, Cramer CJ, Truhlar DG (2009) J Phy Chem B 113:6378
Bard AJ, Faulkner LR (2000) Electrochemical methods: fundamentals and applications, 2nd edn. John Wiley and Sons Inc., New York
Acknowledgements
VSB acknowledges support from the AFOSR Grant# FA9550-13-1-0020 and supercomputing time from NERSC and from the high-performance computing facilities at Yale University. The authors thank M. Zahid Ertem for valuable discussions. ABB and KL acknowledges support from the Air Force Office of Scientific Research through the MURI program under AFOSR Award No. FA9550-10-1-0572 and the NSF under Award CHE-1308652.
Author information
Authors and Affiliations
Corresponding authors
Appendix
Appendix
The reduction potential of the Brønsted acid (HA = PyrH+, ImH+) relative to the SHE, is obtained as the cell potential for the equilibrium:
which can be expressed as two half reactions:
and
The Gibbs free energy change ΔG for the overall reaction of Eq. (1) is given by
Which is essentially the Nernst equation upon substitution of ΔGo = −F Eo and ΔG = −F E:
At equilibrium, ΔG = 0 and [A−][H+]/[AH] = Ka. Therefore,
When T = 298.15 K. Considering that E0 = E 0PyrH +/H(Pt)Pyr−E 0H +/H(Pt), we obtain:
Rights and permissions
About this article
Cite this article
Liao, K., Askerka, M., Zeitler, E.L. et al. Electrochemical Reduction of Aqueous Imidazolium on Pt(111) by Proton Coupled Electron Transfer. Top Catal 58, 23–29 (2015). https://doi.org/10.1007/s11244-014-0340-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-014-0340-2