Abstract
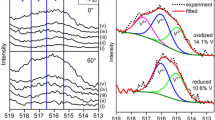
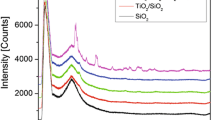
Vanadium oxide (VOx) molecular species supported on high surface area oxide supports are active catalysts for oxidative transformations of organic molecules. Since the reactivity of VOx species depends on their molecular structure, the understanding and control of factors that determine their structure would be useful in surface molecular catalyst design. Reactive adsorption of vanadyl triisopropoxide (VOTP) to form monomeric VOx species on amorphous Al2O3 and SrTiO3 (001) surfaces has been studied by X-ray photoelectron spectroscopy (XPS). Quantitative comparison of C(1s) and V(2p3/2) peak areas has been used to determine the number of isopropoxide ligands that are replaced by V–O surface bonds. On average, three V–O surface bonds are formed during adsorption on an amorphous Al2O3 surface, as expected in the formation of a tridentate, VO4 structure, typically assigned to monomeric, surface VOx species. On the SrTiO3 (001) surface, the number of V–O surface bonds depends on the oxygen density prior to reaction with VOTP. For adsorption on the SrTiO3 surface cleaned and oxygen-annealed in ultrahigh vacuum, the number of V–O surface bonds is ca. 2. When the SrTiO3 surface has been Ar-ion sputtered prior to VOTP adsorption, the number of V–O bonds is ca. 1. This study demonstrates that the atomic structure of the support can strongly influence the molecular nature of surface VOx species.









Similar content being viewed by others
References
Weckhuysen BM, Keller DE (2003) Catal Today 78:25
Ballarini N, Cavani F, Cericola A, Cortelli C, Ferrari M, Trifiro F, Capannelli G, Comite A, Catani R, Cornaro U (2004) Catal Today 91–2:99
Haber J, Kozlowska A, Kozlowski R (1986) J Catal 102:52
Khodakov A, Olthof B, Bell AT, Iglesia E (1999) J Catal 181:205
Wu Z, Kim H-S, Stair PC, Rugumini S, Jackson SD (2005) J Phys Chem 109:2793
Wu Z, Stair PC, Rugmini S, Jackson SD (2007) J Phys Chem C 111:16460
Kim H, Ferguson GA, Cheng L, Zygmunt SA, Stair PC, Curtiss LA (2012) J Phys Chem C 116:2927
Kim HS, Zygmunt SA, Stair PC, Zapol P, Curtiss LA (2009) J Phys Chem C 113:8836
Rice GL, Scott SL (1997) Langmuir 13:1545
Feng H, Elam JW, Libera JA, Pellin MJ, Stair PC (2010) J Catal 269:421
George SM (2010) Chem Rev 110:111
Stair PC (2012) Top Catal 55:93
Keranen J, Guimon C, Iiskola E, Auroux A, Niinisto L (2003) J Phys Chem B 107:10773
Badot JC, Ribes S, Yousfi EB, Vivier V, Pereira-Ramos JP, Baffier N, Lincot D (2000) Electrochem Solid State Lett 3:485
Badot JC, Mantoux A, Baffier N, Dubrunfaut O, Lincot D (2004) J Mater Chem 14:3411
Musschoot J, Deduytsche D, Poelman H, Haemers J, Van Meirhaeghe RL, Van den Berghe S, Detavernier C (2009) J Electrochem Soc 156:P122
Parker BR, Jenkins JF, Stair PC (1997) Surf Sci 372:185
Kanai H, Yoshikawa T, Sone T, Nishimura Y (2002) React Kinet Catal Lett 75:213
Wagner W, Pruss A (2002) J Phys Chem Ref Data 31:387
Schulz KH, Cox DF (1993) J Phys Chem 97:647
Pilleux ME, Grahmann CR, Fuenzalida VM (1994) J Am Ceram Soc 77:1601
Moulder JF, Stickel WF, Sobol PE, Bomben KD (1992) Handbook of X-ray photoelectron spectroscopy. Perkin-Elmer, Eden Praire
Schmidt D, Strehle S, Albert M, Hentsch W, Bartha JW (2008) Microelectron Eng 85:527
Lee SY, Jeon C, Kim SH, Kim Y, Jung W, An KS, Park CY (2012) Jpn J Appl Phys 51:100201
Ma Q, Guo HS, Gordon RG, Zaera F (2010) Chem Mat 22:352
Matero R, Rahtu A, Ritala M, Leskela M, Sajavaara T (2000) Thin Solid Films 368:1
Briggs DS, Seah MP (1990) Practical surface analysis: Auger and X-ray photoelectron spectroscopy, vol 1, 2nd edn. Wiley, Chichester
Hess C, Tzolova-Mueller G, Herbert R (2007) J Phys Chem C 111:9471
Hess C, Schloegl R (2006) Chem Phys Lett 432:139
Sawatzky GA, Post D (1979) Phys Rev B Condens Matter 20:1546
Liu L, Quezada BR, Stair PC (2010) J Phys Chem C 114:17105
Powell CJ, Jablonski A (2002) J Surf Anal 9:322
George SM (2010) Chem Rev 110:111
Badot JC, Ribes S, Yousfi EB, Vivier V, Pereira-Ramos JP, Baffier N, Lincot D (2000) Electrochem Solid-State Lett 3:485
Becerra-Toledo AE, Enterkin JA, Kienzle DM, Marks LD (2012) Surf Sci 606:791
Becerra-Toledo AE, Castell MR, Marks LD (2012) Surf Sci 606:762
Lu J, Stair PC (2010) Langmuir 26:16486
Adiga SP, Zapol P, Curtiss LA (2007) J Phys Chem C 111:7422
Erdman N, Poeppelmeier KR, Asta M, Warschkow O, Ellis DE, Marks LD (2002) Nature 419:55
Erdman N, Warschkow O, Asta M, Poeppelmeier KR, Ellis DE, Marks LD (2003) J Am Chem Soc 125:10050
Warschkow O, Asta M, Erdman N, Poeppelmeier KR, Ellis DE, Marks LD (2004) Surf Sci 573:446
Lanier CH, van de Walle A, Erdman N, Landree E, Warschkow O, Kazimirov A, Poeppelmeier KR, Zegenhagen J, Asta M, Marks LD (2007) Phys Rev B 76:04542
Kienzle DM, Becerra-Toledo AE, Marks LD (2011) Phys Rev Lett 106:176102
Lin YY, Becerra-Toledo AE, Silly F, Poeppelmeier KR, Castell MR, Marks LD (2011) Surf Sci 605:L51
Becerra-Toledo AE, Marshall MSJ, Castell MR, Marks LD (2012) J Chem Phys 136:214701
Kienzle DM, Marks LD (2012) CrystEngComm 14:7833
Enterkin JA, Subramanian AK, Russell BC, Castell MR, Poeppelmeier KR, Marks LD (2010) Nat Mater 9:245
Marshall MSJ, Becerra-Toledo AE, Payne DJ, Egdell RG, Marks LD, Castell MR (2012) Phys Rev B 86:125416
Mayer JT, Diebold U, Madey TE, Garfunkel E, Electron Spectrosc J (1995) Relat Phenom 73:1
Wachs IE, Weckhuysen BM (1997) Appl Catal A 157:67
Du G, Lim S, Pinault M, Wang C, Fang F, Pfefferle L, Haller GL (2008) J Catal 253:74
Chlosta R, Tzolova-Muller G, Schlogl R, Hess C (2011) Catal Sci Technol 1:1175
Todorova TK, Ganduglia-Pirovano MV, Sauer J (2007) J Phys Chem C 111:5141
Wang QG, Madix RJ (2001) Surf Sci 474:L213
Ferreira ML, Volpe M (2000) J Mol Catal A-Chem 164:281
Wachs IE, Chen Y, Jehng JM, Briand LE, Tanaka T (2003) Catal Today 78:13
Baron M, Abbott H, Bondarchuk O, Stacchiola D, Uhl A, Shaikhutdinov S, Freund HJ, Popa C, Ganduglia-Pirovano MV (2009) J Sauer. Angew Chem-Int Ed 48:8006
Penschke C, Paier J, Sauer J (2013) J Phys Chem C 117:5274
Wu Z, Dai S, Overbury SH (2010) J Phys Chem C 114:412
Wegener SL, Kim H, Marks TJ, Stair PC (2011) J Phys Chem Lett 2:170
Caughlan CN, Smith HM, Watenpau K (1966) Inorg Chem 5:2131
Inumaru K, Okuhara T, Misono M (1991) J Phys Chem 95:4826
Tian HJ, Ross EI, Wachs IE (2006) J Phys Chem B 110:9593
Rozanska X, Fortrie R, Sauer J (2007) J Phys Chem C 111:6041
Fu G, Xu X, Lu X, Wan H (2005) J Phys Chem B 109:6416
Acknowledgments
This material is based upon work supported by the National Science Foundation under Grant No. CHE-1058835. Brian Quezada acknowledges support from Chemical Sciences, Geosciences and Biosciences Division, Office of Basic Energy Sciences, Office of Science, US Department of Energy (Award DE-FG02-03-ER15457).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Johnson, A.M., Quezada, B.R., Marks, L.D. et al. Influence of the Metal Oxide Substrate Structure on Vanadium Oxide Monomer Formation. Top Catal 57, 177–187 (2014). https://doi.org/10.1007/s11244-013-0174-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-013-0174-3