Abstract
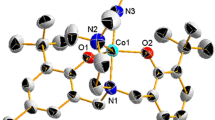
N-phenylpyridin-2-ylmethanimine, HL reacts with CoBr2 to afford a water-soluble cobalt(II) complex [CoII(HL)2Br2] 1, whose crystal structure has been determined by X-ray diffraction. Electrochemical studies show that this complex can electrocatalyze hydrogen generation from a neutral buffer with a turnover frequency (TOF) of 875.17 mol of hydrogen per mole of catalyst per hour (mol H2/mol catalyst/h) at an overpotential (OP) of 837.6 mV. Compared with the cobalt complex 1, the previously described nickel(II) complex [Ni(HL)2Cl2] (970.45 mol H2/mol catalyst/h at an OP of 837.6 mV) exhibits more efficient activity for hydrogen evolution.








Similar content being viewed by others
References
Gan L, Groy TL, Tarakeshwar P, Mazinani SKS, Shearer J, Mujica V, Jones AK (2015) J Am Chem Soc 137:1109–1115
Chen J, Wang X, Shao Y, Zhu J, Zhu Y, Li Y, Xu Q, Guo Z (2007) Inorg Chem 46:3306–3312
Mandal D, Chauhan M, Arjmand F, Arom G, Ray D (2009) Dalton Trans 38:9183–9191
Du Bois J, Mizoguchi TJ, Lippard SJ (2000) Coord Chem Rev 200–202:443–485
Que L Jr, Tolman W (2002) Angew Chem Int Ed 41:1114–1137
Fontecilla-Camps JC, Volbeda A, Cavazza C, Nicolet Y (2007) Chem Rev 107:4273–4303
Vignais PM, Billoud B (2007) Chem Rev 107:4206–4272
Sampson MD, Kubiak CP (2015) Inorg Chem 54:6674–6676
Hou J, Fang M, Cardenas AJP, Shaw WJ, Helm ML, Bullock RM, Roberts JAS, O’Hagan M (2014) Energy Environ Sci 7:4013–4017
Cao JP, Fang T, Fu LZ, Zhou LL, Zhan SZ (2014) Int J Hydrogen Energy 39:10980–10986
Das A, Han Z, Brennessel WW, Holland PL, Eisenberg R (2015) ACS Catal 5:1397–1406
Gan L, Groy TL, Tarakeshwar P, Mazinani SKS, Shearer J, Mujica V, Jones AK (2015) J Am Chem Soc 137:1109–1115
Fang T, Fu LZ, Zhou LL, Zhan SZ, Chen S (2015) Electrochim Acta 178:368–373
Rodenberg A, Orazietti M, Probst B, Bachmann C, Alberto R, Baldridge KK, Hamm P (2015) Inorg Chem 54:646–657
Kandemir B, Kubie L, Guo Y, Sheldon B, Bren KL (2016) Inorg Chem 55:1355–1357
Cao JP, Fang T, Fu LZ, Zhou LL, Zhan SZ (2014) Int J Hydrogen Energy 39(2014):13972–13978
Zhou LL, Fang T, Cao JP, Zhu Z, Su X, Zhan SZ (2015) J Power Sources 273:298–304
Fang T, Fu LZ, Zhou LL, Zhan SZ (2015) Electrochim Acta 161:388–394
Hu X, Brunschwig BS, Peter JC (2007) J Am Chem Soc 129:8988–8998
Fillol JL, Codolà Z, Garcia-Bosch I, Gómez L, Pla JJ, Costas M (2011) Nat Chem 3:807–813
Vennampalli M, Liang G, Katta L, Webster CE, Zhao X (2014) Inorg Chem 53:10094–10100
Xue D, Peng QX, Zhan SZ (2017) Inorg Chem Commun 82:11–15
Sheldrick GM (1996) SADABS, program for empirical absorption correction of area detector data. University of Götingen, Götingen
Sheldrick GM (2015) Acta Crystallogr C71:3–8
Bozic-Weber B, Constable EC, Housecroft CE, Neuburger M, Price JR (2010) Dalton Trans 39:3585–3594
Fu LZ, Tang LZ, Zhang YX, Liang QN, Fang C, Zhan SZ (2016) Int J Hydrogen Energy 41:249–254
Karunadasa HI, Chang CJ, Long JR (2010) Nature 464:1329–1333
Zhang YX, Lin CN, Liu J, Zhan SZ, Xie XH (2016) Transit Met Chem 41:623–627
Tang LZ, Lin CN, Zhan SZ (2016) Transit Met Chem 41:819–825
Tong L, Zong R, Thummel RP (2014) J Am Chem Soc 136:4881–4884
Felton GAN, Glass RS, Lichtenberger DL, Evans DH (2006) Inorg Chem 45:9181–9184
Nichloson RS, Shain I (1964) Anal Chem 36:706–723
Sun Y, Bigi JP, Piro NA, Tang ML, Long JR, Chang CJ (2011) J Am Chem Soc 133(2011):9212–9215
Singh WM, Baine T, Kudo S, Tian S, Ma XAN, Zhou H, DeYonker NJ, Pham TC, Bollinger JC, Baker GL, Yan B, Webster CE, Zhao X (2012) Angew Chem Int Ed 51:5941–5944
Wang ZQ, Tang LZ, Zhang YX, Zhan SZ, Ye JS (2015) J Power Sources 287:50–57
Fu LZ, Tang LZ, Zhang YX, Liang QN, Fang C, Zhan SZ (2016) Int J Hydrogen Energy 41:249–254
Acknowledgements
This work was supported by the National Science Foundation of China (Nos. 20971045 and 21271073).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xue, D., Luo, SP., Yang, LF. et al. Synthesis, characterization and electrocatalytic performance of a cobalt(II) complex of N-phenylpyridin-2-ylmethanimine. Transit Met Chem 42, 615–621 (2017). https://doi.org/10.1007/s11243-017-0166-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-017-0166-4