Abstract
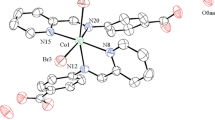
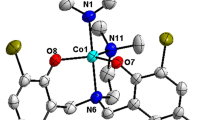
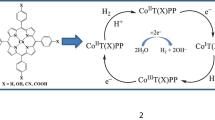
The reaction of N,N-dimethylethylenediamino-N,N-bis(2-tert-butyl-4-ethylphenol) (H2L′) with CoBr2 gives a Co(II) complex [CoL′(NCMe)] 1, whose structure has been determined by X-ray diffraction. Complex 1 can electrocatalyze hydrogen evolution from both acetic acid, with a turnover frequency (TOF) of 43 mol of H2 per mole of catalyst per hour under −1.30 V versus Ag/AgNO3 (in CH3CN), and from buffer (pH 7.0) with a TOF of 851.1 mol of H2 per mole of catalyst per hour under −1.45 V versus Ag/AgCl. The electronic properties of the ligands are found to play a vital role in determining the catalytic activity.






Similar content being viewed by others
References
Lewis NS, Nocera DG (2006) Proc Nat Acad Sci USA 103:15729–15735
Fontecilla-Camps JC, Volbeda A, Cavazza C, Nicolet Y (2007) Chem Rev 107:4273–4303
Vignais PM, Billoud B (2007) Chem Rev 107:4206–4272
Pickett CJ, Tard C (2009) Chem Rev 109:2245–2274
McKone JR, Marinescu SC, Brunschwig BS, Winkler JR, Gray HB (2014) Chem Sci 5:865–878
Vincent KA, Parkin A, Armstrong FA (2007) Chem Rev 107:4366–4413
Darensbourg MY, Wegand W (2011) Eur J Inorg Chem 2011:994–1004
Darensbourg MY, Lyon EJ, Smee JJ (2000) Coord Chem Rev 206:533–561
Marr AC, Spencer DJE, Schroder M (2001) Coord Chem Rev 219:1055–1074
Nguyen AD, Rail MD, Shanmugam M, Fettinger JC, Berben LA (2013) Inorg Chem 52:12847–12854
Quentel F, Passard G, Gloaguen F (2012) Energy Environ Sci 5:7757–7761
Darensbourg MY, Lyon EJ, Zhao X, Georgakaki IP (2003) Proc Natl Acad Sci USA 100:3683–3688
Evans DJ, Pickett CJ (2003) Chem Soc Rev 32:268–275
Canaguier S, Fourmond V, Perotto CU, Fize J, Pecaut J, Fontecave M, Fielde MJ, Artero V (2013) Chem Commun 49:5004–5006
Carroll ME, Barton BE, Rauchfuss TB, Carroll PJ (2012) J Am Chem Soc 134:18843–18852
Wang N, Wang M, Wang Y, Zheng D, Han H, Ahlquist MSG, Sun L (2013) J Am Chem Soc 135:13688–13691
Gross MA, Reynal A, Durrant JR, Reisner E (2014) J Am Chem Soc 136:356–366
Das A, Han Z, Brennessel WW, Holland PL, Eisenberg R (2015) ACS Catal 5:1397–1406
Cao JP, Fang T, Fu LZ, Zhou LL, Zhan SZ (2014) Int J Hydrogen Energy 39:10980–10986
Cao JP, Fang T, Wang ZQ, Ren YW, Zhan SZ (2014) J Mol Catal A Chem 391:191–197
Thoi VS, Karunadasa HI, Surendranath Y, Long JR, Chang CJ (2012) Energy Environ Sci 5:7762–7770
Zhou LL, Fu LZ, Liang Q, Fang C, Zhan SZ (2016) J Coord Chem 69:628–637
Cowper CJ, DeRose AJ (1983) The analysis of gases by chromatography. British Gas Corporation, London Research Station, Section 4.7 Mixed Carrier Gas, pp 56–60
Sheldrick GM (1996) SADABS, program for empirical absorption correction of area detector data. University of Göttingen, Göttingen
Sheldrick GM (2008) Acta Crystallogr Sect A A64:112–122
Karunadasa HI, Chang CJ, Long JR (2010) Nature 464:1329–1333
Codol Z, Garcia-Bosch I, Acuna-Pares F, Prat I, Luis JM, Costas M, Lloret-Fillol J (2013) Chem Eur J 19:8042–8047
Acknowledgments
This work was supported by (1) the National Science Foundation of China (Nos. 20971045, 21271073) and (2) the Student Research Program (SRP) of South China University of Technology (No. B15-B7050170).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, YX., Lin, CN., Liu, J. et al. Synthesis and electrocatalytic properties of a cobalt(II) complex supported by N,N-dimethylethylenediamino-N,N-bis(2-tert-butyl-4-ethylphenol). Transit Met Chem 41, 623–627 (2016). https://doi.org/10.1007/s11243-016-0061-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-016-0061-4