Abstract
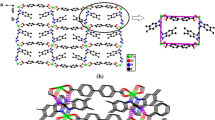
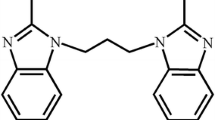
Two metal–organic coordination polymers, {Co(bbbi)0.5(bm)(Hbtc)} n (1) and {Ag2(bbbi)2(ntp)(H2O)·4H2O} n (2), [bbbi = 1,1-(1,4-butanediyl)bis-1H-benzimidazole, bm = benzimidazole, H3btc = 1,2,4-trimellitic acid, and H2ntp = 2-nitroterephthalic acid], have been hydrothermally synthesized and characterized by physico-chemical and spectroscopic methods and single-crystal diffraction. 1 Features a 1D ladder-like chain and is further connected by O–H···O hydrogen bonding interactions to yield a 3D supramolecular architecture. 2 Possesses a 1D infinite zigzag chain connected by bbbi ligands in bis-monodentate mode, which is further extended into a 3D complicated supramolecular network by face-to-face π–π stacking interactions and O–H···O hydrogen bonds. Moreover, both compounds exhibit catalytic properties on degradation of methyl orange in Fenton-like process.







Similar content being viewed by others
References
Etaiw SEH, Badr El-din AS, El-bendary MM (2013) Z Anorg Allg Chem 639:810–816
Maynard BA, Sykora RE, Maguec JT, Gorden AEV (2010) Chem Commun 46:4944–4946
Ma LF, Wang LY, Du M, Batton SR (2010) Inorg Chem 49:365–367
Cui GH, Li JR, Tian JL, Bu XH, Batten SR (2005) Gryst Growth Des 5:1775–1780
Carballo R, Covelo B, Fernández-Hermida N, Lago AB, Vázquez-López EM (2009) CrystEngComm 11:817–826
Batten SR (2005) J Solid State Chem 178:2475–2479
Pollino JM, Weck M (2005) Chem Soc Rev 34:193–207
Beatty AM (2003) Coord Chem Rev 246:131–143
Nishio M (2004) CrystEngComm 6:130–158
Xiao SL, Du X, Qin L, He CH, Cui GH (2012) J Inorg Organomet Polym 22:1384–1390
Liu GC, Huang JJ, Zhang JW, Wang XL, Lin HY (2013) Transition Met Chem 38:359–365
Xiao SL, Zhao YQ, He CH, Cui GH (2013) J Coord Chem 66:89–97
Fan J, Yee GT, Wang GB, Hanson BE (2006) Inorg Chem 45:599–608
Li SL, Lan YQ, Ma JF, Yang J, Wei GH, Zhang LP, Su ZM (2008) Cryst Growth Des 8:675–684
Zhu X, Sun PP, Ding JG, Li BL, Li HY (2012) Cryst Growth Des 12:3992–3997
Effendy A, Marchetti F, Pettinari C, Pettinari R, Ricciutelli M, Skelton BW, White AH (2004) Inorg Chem 43:2157–2165
Jiao CH, He CH, Geng JC, Cui GH (2012) Transition Met Chem 37:17–23
Wang XL, Hou LL, Zhang JW, Zhang JX, Liu GC, Yang S (2012) CrystEngComm 14:3936–3944
Geng JC, Qin L, He CH, Cui GH (2012) Transition Met Chem 37:579–585
Wang XL, Zhang JX, Liu GC, Lin HY, Chen YQ, Kang ZH (2011) Inorg Chim Acta 368:207–215
Xiao B, Yang LJ, Xiao HY, Fang SM (2011) J Coord Chem 64:4408–4420
Yang Q, Zhang XF, Zhao JP, Hu BW, Bu XH (2011) Cryst Growth Des 11:2839–2845
Yan Y, Wu CD, He X, Sun YQ, Lu CZ (2005) Cryst Growth Des 5:821–827
Ma AQ (2008) Zhu LG J Coord Chem 61:3172–3179
Hoskins BF, Robson R, Slizys DA (1997) J Am Chem Soc 119:2952–2953
Qin L, Liu LW, Du X, Cui GH (2013) Transition Met Chem 38:85–91
Technologies Agilent (2012) CrysAlis PRO agilent technologies. Yarnton, England
Sheldrick GM (2008) Acta Crystallog. SectA 64:112–122
Liu QY, Yuan DQ, Xu L (2007) Cryst Growth Des 7:1832–1843
Du M, Zhang ZH, You YP, Zhao XJ (2008) CrystEngComm 10:306–321
Du JL, Hu TL, Zhang SM, Zeng YF, Bu XH (2008) CrystEngComm 10:1866–1874
Wang CC, Wang P, Guo GS (2010) Transition Met Chem 35:721–729
Amo-Ochoa P, Sanz Miguel PJ, Castilloc O, Zamora F (2007) CrystEngComm 9:987–990
Braverman MA, LaDuca RL (2007) Cryst Growth Des 7:2343–2351
Zhai QG, Lu CZ, Wu XY, Batten SR (2007) Cryst Growth Des 7:2332–2342
Jiang H, Ma JF, Zhang WL, Liu YY, Yang J, Ping GJ, Su ZM (2008) Eur J Inorg Chem 5:745–755
Allendorf MD, Bauer CA, Bhakta RK, Houk RJT (2009) Chem Soc Rev 38:1330–1352
Haquea E, Lee JE, Jang IT, Hwang YK, Chang JS, Jegal J, Jhung SH (2010) J Hazard Mater 181:535–542
Geng JC, Qin L, Du X, Xiao SL, Cui GH (2012) Z Anorg Allg Chem 638:1233–1238
Carriazo JG, Guelou E, Barrault J, Tatibouët JM, Moreno S (2003) Appl Clay Sci 22:303–308
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, L., Xiao, S.L., Ma, P.J. et al. Synthesis, crystal structures and catalytic properties of Ag(I) and Co(II) 1D coordination polymers constructed from bis(benzimidazolyl)butane. Transition Met Chem 38, 627–633 (2013). https://doi.org/10.1007/s11243-013-9730-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-013-9730-8