Abstract
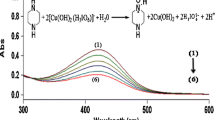
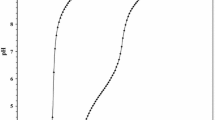
The kinetics of hydrolysis and reduction of the diperiodatoargentate(III) ion (DPA) has been studied in aqueous acidic medium spectrophotometrically. Upon dilution the silver (III) complex was found to be unstable in the presence of H2O. Addition of [H+], largely increased the hydrolysis rate, whereas [OH−] does not have any effect. Under pseudo-first-order conditions ([paracetamol] > [DPA]), the reduction rate was very fast. Second-order conditions were used to determine the reaction rate. The reaction was acid-catalyzed and the rate decreased by the addition of periodate. The Arrhenius equation was valid for the reaction. The changes observed in the direction of the rate constant-[H+] profile correspond to aquation of the diperiodatoargentatate(III) complex. The proposed mechanism and the derived rate law are consistent with the observed kinetics.
Similar content being viewed by others
References
G.L. Cohen G. Atkinson (1964) Inorg. Chem. 3 1741 Occurrence Handle10.1021/ic50022a018 Occurrence Handle1:CAS:528:DyaF2MXnt1Kl
L.J. Kirschenbaum J.H. Ambrus G. Atkinson (1973) Inorg. Chem. 12 2832 Occurrence Handle10.1021/ic50130a018 Occurrence Handle1:CAS:528:DyaE3sXlsV2ru7k%3D
W. Levason M.D. Spicer (1987) Coord. Chem. Rev. 76 45 Occurrence Handle10.1016/0010-8545(87)85002-6 Occurrence Handle1:CAS:528:DyaL2sXhs1OlsLw%3D
R. Banerjee, R. Das and S. Mukhopadhyay, J. Chem. Soc. Dalton Trans., 1317 (1992) and the ref. cited therein.
L.J. Krischenbaum L. Mrozowski (1978) Inorg. Chem. 17 3718 Occurrence Handle10.1021/ic50190a085
A. Kumar A. Panwar (1994) Bull. Chem. Soc. Jpn. 67 1207 Occurrence Handle1:CAS:528:DyaK2cXktlCls74%3D
P.J.P. Rao B. Sethuram T.N. Rao (1981) Indian J. Chem. 20A 733 Occurrence Handle1:CAS:528:DyaL3MXltlWqu7s%3D
A. Kumar P. Kumar P. Ramamurthy (1999) Polyhedron 8 773 Occurrence Handle10.1016/S0277-5387(98)00352-0
M.S. Manhas and Z. Khan, Colloid and Surf. A: Physico. Chem. Eng. Aspects, 277, 207 (2006).
V. Rodenas M.S. Garcia C. Sanchez-Pedrena M.I. Albera (2000) Talanta 52 517 Occurrence Handle10.1016/S0039-9140(00)00397-0 Occurrence Handle1:CAS:528:DC%2BD3cXksV2rsbw%3D
F.A. Mohamed M.A. AbdAllah S.M. Shammat (1997) Talanta 44 61 Occurrence Handle10.1016/S0039-9140(96)02013-9 Occurrence Handle1:CAS:528:DyaK2sXjslGi
J.F. Staden ParticleVan M. Tsanwani (2002) Talanta 58 1095 Occurrence Handle10.1016/S0039-9140(02)00406-X
C.A. Georgiou M.A. Koupparis T.P. Hadjiioannou (1991) Talanta 38 689 Occurrence Handle10.1016/0039-9140(91)80187-5 Occurrence Handle1:CAS:528:DyaK3MXls1KktLk%3D
Z. Khan Kabir-ud-Din (1999) Int. J. Chem. Kinet. 31 409 Occurrence Handle10.1002/(SICI)1097-4601(1999)31:6<409::AID-KIN2>3.0.CO;2-8 Occurrence Handle1:CAS:528:DyaK1MXjsl2rtbg%3D
Z. Khan Raju Kabir-ud-Din (2003) Colloid and Surf. A: Physico. Chem. Eng. Aspects 225 75 Occurrence Handle10.1016/S0927-7757(03)00259-0 Occurrence Handle1:CAS:528:DC%2BD3sXntVGrsL8%3D
Raju Z. Khan (2005) Bull. Chem. Soc. Jpn. 78 1218 Occurrence Handle10.1246/bcsj.78.1218 Occurrence Handle1:CAS:528:DC%2BD2MXmslGjtLY%3D
H.H. Willard L.L. Merritt SuffixJr. J.A. Dean (1965) Instrumental Methods of Analysis EditionNumber4 Affiliated East-West Press New Delhi
G.D. Christian (1986) Analytical Chemistry EditionNumber4 John Wiley Singapore
A.K. Panda S.N. Mahapatro G.P. Panigarhi (1981) J. Org. Chem. 46 400 Occurrence Handle10.1021/jo00333a013
R.S. Verma, M.J. Reddy and V.R. Shastry, J. Chem. Soc., Perkin II, 469 (1976).
T. Raviprasad B. Sethuram T.N. Rao (1979) Indian J.Chem. 18A 40 Occurrence Handle1:CAS:528:DyaE1MXlt1eitLw%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ilyas, M., Khan, Z. Kinetics of Hydrolysis of Diperiodatoargentate(III) Ion and its Reduction by Paracetamol. Transition Met Chem 31, 516–521 (2006). https://doi.org/10.1007/s11243-006-0022-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11243-006-0022-4