Abstract
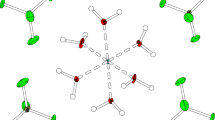
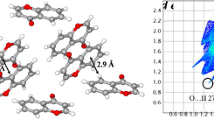
The crystal and molecular structure of tetramethylammonium fluorochromate(VI), (CH3)4N[CrO3F] has been determined at 130(2) K by X-ray diffraction. X-ray data clearly demonstrate inequality between the Cr–O and the Cr–F bonds that is responsible for the higher reactivity of this compound over similar oxidizing agents in terms of the amount of oxidant and solvent required, short reaction times and high yields. The reason for this inequality is due to the CH· · ·F hydrogen bond that forms between the methyl hydrogen of the cation and the fluoride atom of the anion. The IR spectrum and hydrogen bonding of this compound is similar to the tetramethylammonium perchlorate salt and shows the existence of hydrogen bonding.
Similar content being viewed by others
References
A. Bhandari P.K. Sharma K.K. Banerji (2001) Indian J. Chem. 40A 470 Occurrence Handle1:CAS:528:DC%2BD3MXjvFamurk%3D
S. Meenahshisundaram R. Soctaungam (2001) Collect. Czech. Chem. Commun. 66 77
E.J. Corey and D.L. Boger, Tetrahedron Lett., 2461 (1978).
G. Maier H.P. Reisenauer M.D. Marco (1999) Angew. Chem. Int. Ed. Engl. 38 108 Occurrence Handle10.1002/(SICI)1521-3773(19990115)38:1/2<108::AID-ANIE108>3.0.CO;2-U Occurrence Handle1:CAS:528:DyaK1MXpsVaktg%3D%3D
E.J. Corey G. Schmidt (1979) Tetrahedron Lett. 20 399 Occurrence Handle10.1016/S0040-4039(01)93515-4
M.N. Bhattacharjee, M.K. Chaudhuri, H.S. Dasgupta and N. Roy, Synthesis, 588 (1982).
F.A. Luzzio R.A. Bobb (1997) Tetrahedron Lett. 38 1733 Occurrence Handle10.1016/S0040-4039(97)00203-7 Occurrence Handle1:CAS:528:DyaK2sXhvVOhu7g%3D
C.A. Deakyne M. Meot-Ner (1985) J. Am. Chem. Soc. 107 474 Occurrence Handle10.1021/ja00288a034
J. March (1985) Advanced Organic Chemistry John Wiley & Sons Co New York 320
A.R. Mahjoub S. Ghammami M.Z. Kassaee (2003) Tetrahedron Lett. 44 4555 Occurrence Handle10.1016/S0040-4039(03)00989-4 Occurrence Handle1:CAS:528:DC%2BD3sXjvVCnsrY%3D
D.T. Hurle, Handbook of Crystal Growth, Part B, Elsevier Science, 1994, p. 1219.
K.O. Christe W.W. Wilson R.D. Wilson R. Bau J.J. Feng (1990) J.Am. Chem. Soc. 112 7619 Occurrence Handle10.1021/ja00177a025 Occurrence Handle1:CAS:528:DyaK3cXlslWnt7c%3D
G.M. Sheldrick (1998) SHELXTL V. 5.10, Structure Determination Software Suite Bruker AXS Madison, Wisconsin, USA
M. Kaupp (2001) Angew. Chem. Int. Ed. Engl. 40 3534 Occurrence Handle10.1002/1521-3773(20011001)40:19<3534::AID-ANIE3534>3.0.CO;2-# Occurrence Handle1:CAS:528:DC%2BD3MXns1Whu7k%3D
M. Hargittai (2000) Chem. Rev. 100 2233 Occurrence Handle10.1021/cr970115u Occurrence Handle1:CAS:528:DC%2BD3cXjvV2htbk%3D
G. Frenking N. Frohlich (2000) Chem. Rev. 100 717 Occurrence Handle10.1021/cr980401l Occurrence Handle1:CAS:528:DC%2BD3cXlslOguw%3D%3D
M. Kaupp (1999) Chem. Eur. J. 5 3632 Occurrence Handle10.1002/(SICI)1521-3765(19991203)5:12<3631::AID-CHEM3631>3.0.CO;2-1
W. Granier S. Vilminot J.D. Vidal L. Cot (1981) J. Fluor. Chem. 19 123 Occurrence Handle10.1016/S0022-1139(00)81329-9 Occurrence Handle1:CAS:528:DyaL38XksFaruw%3D%3D
K.M. Harmon I. Gennick S.L. Madeira (1974) J. Phys. Chem. 78 2585 Occurrence Handle10.1021/j100618a012 Occurrence Handle1:CAS:528:DyaE2MXitl2ksA%3D%3D
R. Srinivasan P. Stanly K. Balasubramanian (1997) Synth. Commun. 27 2057 Occurrence Handle1:CAS:528:DyaK2sXjtF2ju74%3D
K.K. Banerji, J. Chem. Soc., Perkin. Trans. II, 547 (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghammamy, S., Dastpeyman, S. & Sadjadi, S.A.S. X-ray Structural Analysis of Tetramethylammonium Fluorochromate(VI). A Link between Reactivity and the Extent of Symmetry. Transition Met Chem 31, 482–486 (2006). https://doi.org/10.1007/s11243-006-0017-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11243-006-0017-1