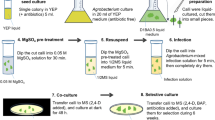
Abstract
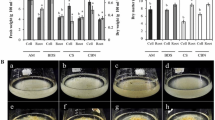
The economic important medicinal plant, Panax ginseng, encompasses a diverse array of pharmacologically beneficial ginsenosides governed by complex metabolic pathways. The cultivation of P. ginseng over an extended period of time poses many challenges to fulfill market requirements. The utilization of in vitro tissue culture presents a viable alternative approach for the generation of P. ginseng root biomass and metabolites. Harnessing the root inducing plasmid (Ri plasmid) of Agrobacterium rhizogenes in the transformation process to create hairy roots in P. ginseng could be a useful metabolic engineering technique. In this investigation, the transformation efficiency and biomass production of five distinct ginseng genotypes were evaluated. Of them, the ‘Yunpoong’ cultivar, and a local landrace ‘Ganghwa’ had the highest transformation efficiencies of 66.11% and 65.00%, respectively. The biomass production of transgenic hairy roots was 1.5–2.1 times faster than that of non-transgenic adventitious roots without hormone supplementation. Various ginsenosides such as Rg1, Rf, Rh1, Rb1, Rb2, Rd, F2, and Rg3, were found to be similar or greater in the hairy roots when compared to the concentrations observed in adventitious roots. Furthermore, the ginsenoside contents of cultivated roots are similar to those cultured in a bioreactor. The findings provide fundamental insights into the metabolic engineering of ginseng, facilitating the in vitro production of ginsenosides.
Key message
In vitro root culture is an ideal method for the large-scale production of biomass for the slowly growing ginseng plant. We report the improved transgenic hairy root culture system for production of biomass and ginsenosides via Ri plasmid transformation using five different ginseng genotypes. This transformation system provides a basic tool for the future metabolic engineering of ginseng.





Similar content being viewed by others
Data availability
The authors confirm that data supporting the findings of this study are available within the article or its supplementary material.
References
Aarrouf J, Castro-Quezada P, Mallard S, Caromel B, Lizzi Y, Lefebvre V (2012) Agrobacterium rhizogenes-dependent production of transformed roots from foliar explants of pepper (Capsicum annuum): a new and efficient tool for functional analysis of genes. Plant Cell Rep 31(2):391–401
Al Abdallat A, Sawwan J, Al Zoubi B (2011) Agrobacterium tumefaciens-mediated transformation of callus cells of Crataegusaronia. Plant Cell Tissue Organ Cult (PCTOC) 104(1):31–39
Ali MB, Yu KW, Hahn EJ, Paek KY (2006) Methyl jasmonate and salicylic acid elicitation induces ginsenosides accumulation, enzymatic and non-enzymatic antioxidant in suspension culture Panax ginseng roots in bioreactors. Plant Cell Rep 25(6):613–620
Bae H, Kim YB, Park NI, Kim HH, Kim YS, Lee MY, Park SU (2012) Agrobacterium rhizogenes-mediated genetic transformation of radish (Raphanus sativus L. cv. Valentine) for accumulation of anthocyanin. Plant Omics 5(4):381
Baque MA, Moh SH, Lee EJ, Zhong JJ, Paek KY (2012) Production of biomass and useful compounds from adventitious roots of high-value added medicinal plants using bioreactor. Biotechnol Adv 30(6):1255–1267
Chen SL, Yu H, Luo HM, Wu Q, Li CF, Steinmetz A (2016) Conservation and sustainable use of medicinal plants: problems, progress, and prospects. Chin Med 11(1):37
Choi KT (2008) Botanical characteristics, pharmacological effects and medicinal components of Korean Panax ginseng CA Meyer. Acta Pharmacol Sin 29(9):1109
Christensen LP (2008) Ginsenosides: chemistry, biosynthesis, analysis, and potential health effects. Adv Food Nutr Res 55:1–99
NR Council (1987) Agricultural biotechnology: strategies for national competitiveness. Natl Acad Press, Washington, DC
DiCosmo F, Misawa M (1995) Plant cell and tissue culture: alternatives for metabolite production. Biotechnol Adv 13(3):425–453
Furuya T, Yoshikawa T, Orihara Y, Oda H (1984) Studies of the culture conditions for Panax ginseng cells in jar fermentors. J Nat Prod 47(1):70–75
Ghiasi SM, Salmanian AH, Sharafi A, Kazemi R, Jafari M, Chinikar S, Zakeri S (2012) Molecular farming, an effective system for the production of immunogenic Crimean-Congo hemorrhagic fever virus glycoprotein. Prog Biol Sci 2(1):12–29
Giri A, Narasu ML (2000) Transgenic hairy roots: recent trends and applications. Biotechnol Adv 18(1):1–22
Gorpenchenko T, Kiselev K, Bulgakov V, Tchernoded G, Bragina E, Khodakovskaya M, Koren O, Batygina T, Zhuravlev YN (2006) The Agrobacterium rhizogenes rolc-gene-induced somatic embryogenesis and shoot organogenesis in Panax ginseng transformed calluses. Planta 223(3):457–467
Han JY, Choi YE (2009) Rapid induction of Agrobacterium tumefaciens-mediated transgenic roots directly from adventitious roots in Panax ginseng. Plant Cell Tissue Organ Cult (PCTOC) 96(2):143
Han H, Jang KM, Chung JS (2017) Selecting suitable sites for mountain ginseng (Panax ginseng) cultivation by using geographically weighted logistic regression. J Mt Sci 14(3):492–500
Hishe M, Asfaw Z, Giday M (2016) Review on value chain analysis of medicinal plants and the associated challenges. J Med Plants Stud 4(3):45–55
Hodal L, Bochardt A, Nielsen JE, Mattsson O, Okkels FT (1992) Detection, expression and specific elimination of endogenous β-glucuronidase activity in transgenic and non-transgenic plants. Plant Sci 87(1):115–122
Hussain MS, Fareed S, Saba Ansari M, Rahman A, Ahmad IZ, Saeed M (2012) Current approaches toward production of secondary plant metabolites. J Pharm Bioallied Sci 4(1):10
Ismael KA, Antar EBN (2014) Establishment of high-efficiency Agrobacterium-mediated transformation conditions of soybean callus. Indian J Biotechnol 13:459–463
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5(4):387–405
Kim YS, Yeung EC, Hahn EJ, Paek KY (2007) Combined effects of phytohormone, indole-3-butyric acid, and methyl jasmonate on root growth and ginsenoside production in adventitious root cultures of Panax ginseng C.A. Meyer. Biotechnol Lett 29:1789–1792. https://doi.org/10.1007/s10529-007-9442-2
Kim YJ, Zhang D, Yang DC (2015) Biosynthesis and biotechnological production of ginsenosides. Biotechnol Adv 33:717–735
Kim YJ, Silva J, Zhang D, Shi J, Joo SC, Jang MG, Kwon WS, Yang DC (2016) Development of interspecies hybrids to increase ginseng biomass and ginsenoside yield. Plant Cell Rep 35(4):779–790
Kim NH, Jayakodi M, Lee SC, Choi BS, Jang WJ, Lee J, Kim HH, Waminal NE, Lakshmanan M, Nguyen VB, Lee YS, Park HS, Koo HJ, Park JY, Perumal S, Joh HJ, Lee H, Kim JK, Kim IS, Kim KH, Koduru L, Kang KB, Sung SH, Yu Y, Park DS, Choi D, Seo E, Kim S, Kim YC, Hyun DY, Park YI, Kim C, Lee TH, Kim HU, Soh MS, Lee Y, In JG, Kim HS, Kim YM, Yang DC, Wing RA, Lee DY, Paterson A, Yang TJ (2017) Genome and evolution of the shade-requiring medicinal herb Panax ginseng. Plant Biotechnol J. https://doi.org/10.1111/pbi.12926
Komari T (1989) Transformation of callus cultures of nine plant species mediated by Agrobacterium. Plant Sci 60(2):223–229
Kumar V, Jones B, Davey M (1991) Transformation by Agrobacterium rhizogenes and regeneration of transgenic shoots of the wild soybean Glycine argyrea. Plant Cell Rep 10(3):135–138
Langhansova L, Marsik P, Vanek T (2005) Production of saponins from Panax ginseng suspension and adventitious root cultures. Biol Plant 49(3):463–465
Lee YS, Park HS, Lee DK, Jayakodi M, Kim NH, Koo HJ, Lee SC, Kim YJ, Kwon SW, Yang TJ (2017) Integrated transcriptomic and metabolomic analysis of five Panax ginseng cultivars reveals the dynamics of ginsenoside biosynthesis. Front Plant Sci 8:1048
Liu S, Su L, Liu S, Zeng X, Zheng D, Hong L, Li L (2016) Agrobacterium rhizogenes-mediated transformation of Arachis hypogaea: an efficient tool for functional study of genes. Biotechnol Biotechnol Equip 30:869–878
Murthy HN, Lee EJ, Paek KY (2014) Production of secondary metabolites from cell and organ cultures: strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tissue Organ Cult (PCTOC) 118(1):1–16
Nguyen TT, Paek KY (2010) Cell suspension culture Panax ginseng CA Meyer: role of plant growth regulators and medium composition on biomass and ginsenoside production. VNU J Sci Nat Sci Technol 26:191–196
Ono NN, Tian L (2011) The multiplicity of hairy root cultures: prolific possibilities. Plant Sci 180(3):439–446
Paek K, Chakrabarty D, Hahn E (2005) Application of bioreactor systems for large scale production of horticultural and medicinal plants. In: Hvoslef-Eide AK, Preil W (eds) Liquid culture systems for in vitro plant propagation. Springer, Berlin
Pratap Chandran R, Potty V (2011) Initiation of hairy roots from Canavalia sp. using Agrobacterium rhizogenes 15834 for the co-cultivation of Arbuscular mycorrhizal fungi, Glomus Microcarpum. J Agric Technol 7(2):235–245
Pritts KD (2010) Ginseng: how to find, grow, and use America’s forest gold, 2nd edn. Stackpole Books, Mechanicsburg
Risseeuw E, Dijk ME, Hooykaas PJ (1997) Gene targeting and instability of Agrobacterium T-DNA loci in the plant genome. Plant J 11(4):717–728
Romano E, Soares A, Proite K, Neiva S, Grossi M, Faria JC, Rech EL, Aragão FJ (2005) Transgene elimination in genetically modified dry bean and soybean lines. Genet Mol Res 4(2):177–184
Sharafi A, Sohi HH, Azadi P, Sharafi AA (2014) Hairy root induction and plant regeneration of medicinal plant Dracocephalum kotschyi. Physiol Mol Biol Plants 20(2):257–262
Tao J, Li L (2006) Genetic transformation of Torenia fournieri L. mediated by Agrobacterium rhizogenes. S Afr J Bot 72(2):211–216
Vaněk T, Langhansová L, MaršÍk P (2005) Cultivation of root cultures of Panaxginseng in different bioreactors and in temporary immersion—comparison of growth and saponin production. Liq Culture Syst In Vitro Plant Propag. https://doi.org/10.1007/1-4020-3200-5_40
Verma S, Singh S (2008) Current and future status of herbal medicines. Vet World 1(11):347–350
Warnock JN, Al-Rubeai M (2006) Bioreactor systems for the production of biopharmaceuticals from animal cells. Biotechnol Appl Biochem 45(1):1–12
Woo SS, Song JS, Lee JY, In DS, Chung HJ, Liu JR, Choi DW (2004) Selection of high ginsenoside producing ginseng hairy root lines using targeted metabolic analysis. Phytochem 65(20):2751–2761
Yu Y, Ohh S (1994) Problems and present status of research on ginseng diseases in Korea. Proc Int Ginseng Conf Vancouver, Canada, 120–130
Yu KW, Gao W, Hahn EJ, Paek KY (2002) Jasmonic acid improves ginsenoside accumulation in adventitious root culture of Panax ginseng CA Meyer. Biochem Eng J 11(2):211–215
Yu KW, Hahn EJ, Paek KY (2003) Ginsenoside production by hairy root cultures of Panax ginseng C.A. Meyer in bioreactors. Acta Hortic 597:237–243
Funding
This work was supported by the Cooperative Research Program for Agriculture Science & Technology Development, Rural Development Administration (Grant number PJ01311901), Republic of Korea.
Author information
Authors and Affiliations
Contributions
VBN, MJK, and TJY conceived and designed the experiments. VBN, MJK, VNLG, YSL, BR, EJP, and TKP prepared the samples and performed the experiments. VBN, MJK, KBK, HSP, and BR analyzed the data. VBN, MJ, PM and TJY wrote and revised the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Communicated by Yi Li.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Van Binh, N., Kim, M.J., Giang, V.N.L. et al. Improved biomass and metabolite production in hairy root culture in various genotypes of Panax ginseng through genetic transformation. Plant Cell Tiss Organ Cult 156, 43 (2024). https://doi.org/10.1007/s11240-023-02644-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-023-02644-x