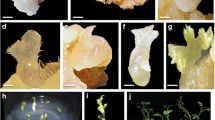
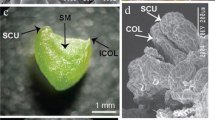
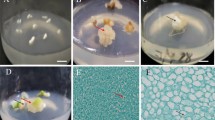
Abstract
A reliable and reproducible embryogenic cell suspension culture system established successfully for regeneration of plants using pro-embryogenic cell mass derived from secondary somatic embryos as explants. A semi-solid medium was employed, which consisted of Murashige and Skoog (MS) basal salts and vitamins supplemented with 4 mg l− 1 2,4-dichlorophenoxyacetic acid (2,4-D), 1 mg l− 1 biotin, 1 mg l− 1 indole acetic acid (IAA), 1 mg l− 1 naphthalene acetic acid (NAA), 25 mg l− 1 ascorbic acid, 3% (w/v) sucrose and 2 g l− 1 phytagel. The genetic integrity of the plants regenerated from the suspension cells was confirmed through various techniques, such as flow cytometry, simple-sequence repeats (SSR) markers and inter simple sequence repeats (ISSR) markers. The field planting displayed no phenotypic deviations and no adverse effects on the vegetative and yield characteristics in banana cv. Sabri, a rapidly declining landrace in Tripura, India due to severe Fusarium wilt incidence. Based on these insights, we propose research priorities and complementary strategies aimed at the efficient conservation and rejuvenation of such valuable landraces.
Key Message
The secondary somatic embryogenesis technique has demonstrated efficiency in establishing an embryogenic cell suspension culture system, offering great promise for producing genetically identical plants to rejuvenate banana cv. Sabri, a declining landrace.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abu Hassan A, Noordin N, Ahmad Z, Akil M, Ahmad F, Ibrahim R (2022) Protocol for mass propagation of plants using a low-cost bioreactor. In: Jankowicz-Cieslak J, Ingelbrecht IL (eds) Efficient screening techniques to identify mutants with TR4 resistance in banana. Springer, Berlin, pp 177–187
Adero M, Tripathi JN, Tripathi L (2023) Advances in somatic embryogenesis of banana. Int J Mol Sci 24:10999. https://doi.org/10.3390/IJMS241310999
Aguilar ME, Wang XY, Escalona M et al (2022) Somatic embryogenesis of Arabica coffee in temporary immersion culture: advances, limitations, and perspectives for mass propagation of selected genotypes. Front Plant Sci 13:994578. https://doi.org/10.3389/FPLS.2022.994578
Backiyarani S, Uma S, Saranya S et al (2021) Multiple shoot induction in zygotic embryos: a strategy for acceleration of banana breeding. Plant Cell Tissue Organ Cult 147:339–350. https://doi.org/10.1007/s11240-021-02127-x
Bogdanović MD, Ćuković KB, Subotić AR et al (2021) Secondary somatic embryogenesis in Centaurium erythraea Rafn. Plants 10:199. https://doi.org/10.3390/plants10020199
Chin WYW, Annuar MSM, Tan BC, Khalid N (2014) Evaluation of a laboratory scale conventional shake flask and a bioreactor on cell growth and regeneration of banana cell suspension cultures. Sci Hortic 172:39–46. https://doi.org/10.1016/j.scienta.2014.03.042
Côte FX, Domergue R, Monmarson S et al (1996) Embryogenic cell suspensions from the male flower of Musa AAA cv. Grand Naine. Physiol Plant 97:285–290. https://doi.org/10.1034/J.1399-3054.1996.970211.X
Côte FX, Folliot M, Domergue R, Dubois C (2000) Field performance of embryogenic cell suspension-derived banana plants (Musa AAA, cv. Grand Naine). Euphytica 112:245–251. https://doi.org/10.1023/A:1003960724547
Dale J, James A, Paul JY et al (2017) Transgenic cavendish bananas with resistance to Fusarium wilt tropical race 4. Nat Commun 2017 8(1):1–8. https://doi.org/10.1038/s41467-017-01670-6
Dhed’A DB, Dumortier F, Panis B, Vuylsteke D (1991) Plant regeneration in cell suspension cultures of the cooking banana cv. Bluggoes (Musa spp. ABB group). Fruits 46:125–135
Dita M, Barquero M, Heck D et al (2018) Fusarium wilt of banana: current knowledge on epidemiology and research needs toward sustainable disease management. Front Plant Sci 871:1–21. https://doi.org/10.3389/fpls.2018.01468
Eun JL, Mobin M, Eun JH, Kee YP (2006) Effects of sucrose, inoculum density, auxins, and aeration volume on cell growth of Gymnema sylvestre. J Plant Biol 49:427–431. https://doi.org/10.1007/BF03031122
FAOSTAT (2021) https://www.fao.org/faostat/en/#data/QCL/visualize. Accessed 21 Mar 2023
Ganapathi TR, Negi S, Tak H, Bapat VA (2021) Transgenic banana: current status, opportunities and challenges. In: Kavi Kishor PB, Rajam MV, Pullaiah T (eds) Genetically modified crops. Springer, Singapore. https://doi.org/10.1007/978-981-15-5932-7_5
Georget F, Domergue R, Ferrière N, Côte FX (2000) Morphohistological study of the different constituents of a banana (Musa AAA, cv. Grande Naine) embryogenic cell suspension. Plant Cell Rep 19:748–754. https://doi.org/10.1007/s002999900188
Ghag SB, Shekhawat UKS, Ganapathi TR (2014) Characterization of Fusarium wilt resistant somaclonal variants of banana cv. Rasthali by cDNA-RAPD. Mol Biol Rep 41:7929–7935. https://doi.org/10.1007/S11033-014-3687-3
Gómez Kosky R, De Feria Silva M, Posada Pérez L et al (2002) Somatic embryogenesis of the banana hybrid cultivar FHIA-18 (AAAB) in liquid medium and scaled-up in a bioreactor. Plant Cell Tissue Organ Cult 68:21–26. https://doi.org/10.1023/A:1012905825307
Hinge VR, Shaikh IM, Chavhan RL et al (2022) Assessment of genetic diversity and volatile content of commercially grown banana (Musa spp.) cultivars. Sci Rep 202212:1–16. https://doi.org/10.1038/s41598-022-11992-1
Ibrahim R (2017) The potential of bioreactor technology for large-scale plant micropropagation. Acta Hortic 1155:573–584. https://doi.org/10.17660/ACTAHORTIC.2017.1155.84
Ismaila AA, Ahmad K, Siddique Y et al (2023) Fusarium wilt of banana: current update and sustainable disease control using classical and essential oils approaches. Hortic Plant J 9:1–28. https://doi.org/10.1016/J.HPJ.2022.02.004
Joanna J, Ivan LI (2022) Efficient screening techniques to identify mutants with TR4 resistance in banana. Springer, Berlin. https://doi.org/10.1007/978-3-662-64915-2
Kannan G, Saraswathi MS, Thangavelu R et al (2022) Development of Fusarium wilt resistant mutants of Musa spp. cv.Rasthali (AAB, Silk subgroup) and comparative proteomic analysis along with its wild type. Planta 255:1–20. https://doi.org/10.1007/S00425-022-03860-Z
Karthic R, Vel K, Uma S, Suthanthiram B (2017) Growth characteristics of embryogenic cell suspension of banana (Musa spp.). In: International symposium on horticulture: priorities & emerging trends (5–8 September, 2017). Society for Promotion of Horticulture, India, Bangaluru, p S2 P41 A685 pp202
Khalil S, Cheah K, Perez E et al (2002) Regeneration of banana (Musa spp. AAB cv. Dwarf brazilian) via secondary somatic embryogenesis. Plant Cell Rep 20:1128–1134. https://doi.org/10.1007/s00299-002-0461-0
Krishna H, Alizadeh M, Singh D et al (2016) Somaclonal variations and their applications in horticultural crops improvement. 3 Biotech 6:1–18. https://doi.org/10.1007/S13205-016-0389-7
Kumaravel M, Uma S, Backiyarani S et al (2020) Antioxidant enzyme activities during somatic embryogenesis in Musa acuminata Colla (AAA group) ‘Grand Naine’ and Musa spp. (AAB group) ‘Rasthali’. Vitro Cell Dev Biol - Plant 56:41–50. https://doi.org/10.1007/s11627-019-10017-3
Le KC, Dedicova B, Johansson S et al (2021) Temporary immersion bioreactor system for propagation by somatic embryogenesis of hybrid larch (Larix × eurolepis Henry). Biotechnol Rep 32:e00684. https://doi.org/10.1016/J.BTRE.2021.E00684
Mohandas S, Sowmya HD, Manjula R et al (2011) Development of highly regenerative embryogenic cell suspensions of “Nanjangud Rasbale” (syn. “Rasthali”, Musa, AAB, silk subgroup) and transformants with amp gene. Acta Hortic 897:245–253. https://doi.org/10.17660/ActaHortic.2011.897.27
Mohandas S, Sowmya HD, Saxena AK et al (2013) Transgenic banana cv. Rasthali (AAB, Silk gp) harboring Ace-AMP1 gene imparts enhanced resistance to Fusarium oxysporum f.sp. Cubense race 1. Sci Hortic 164:392–399. https://doi.org/10.1016/j.scienta.2013.09.018
Molina AB, Roa VN, Van den Bergh I et al (2004) Advancing banana and plantain R&D in Asia and the Pacific - Vol. 12. Proceedings of the 2nd BAPNET Steering Committee meeting held in Jakarta, Indonesia, 6–11 October 2003. International Network for the Improvement of Banana and Plantain - Asia Pacific, Los Baños, Laguna, Philippines
Morais-Lino LS, Santos-Serejo JA, Amorim EP et al (2016) Somatic embryogenesis, cell suspension, and genetic stability of banana cultivars. In Vitro Cell Dev Biol - Plant 52:99–106. https://doi.org/10.1007/s11627-015-9729-2
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/J.1399-3054.1962.TB08052.X
Nandhakumar N, Kumar K, Sudhakar D, Soorianathasundaram K (2018) Plant regeneration, developmental pattern and genetic fidelity of somatic embryogenesis derived Musa spp. J Genet Eng Biotechnol 16:587–598. https://doi.org/10.1016/j.jgeb.2018.10.001
Nyine M, Uwimana B, Blavet N et al (2018) Genomic prediction in a multiploid crop: genotype by environment Interaction and allele dosage effects on predictive ability in banana. Plant Genome 11:170090. https://doi.org/10.3835/PLANTGENOME2017.10.0090
Paul JY, Khanna H, Kleidon J et al (2017) Golden bananas in the field: elevated fruit pro-vitamin A from the expression of a single banana transgene. Plant Biotechnol J 15:520–532. https://doi.org/10.1111/PBI.12650
Pegg KG, Coates LM, O’Neill WT, Turner DW (2019) The epidemiology of Fusarium wilt of banana. Front Plant Sci 10:1–19. https://doi.org/10.3389/fpls.2019.01395
Raemakers CJJM, Jacobsen E, Visser RGF (1995) Secondary somatic embryogenesis and applications in plant breeding. Euphytica 81:93–107. https://doi.org/10.1007/BF00022463
Rai MK, Phulwaria M, Harish et al (2012) Genetic homogeneity of guava plants derived from somatic embryogenesis using SSR and ISSR markers. Plant Cell Tissue Organ Cult 111:259–264. https://doi.org/10.1007/S11240-012-0190-1
Rebouças TA, de Jesus Rocha A, Cerqueira TS et al (2021) Pre-selection of banana somaclones resistant to Fusarium oxysporum f. sp. cubense, subtropical race 4. Crop Prot 147:105692. https://doi.org/10.1016/J.CROPRO.2021.105692
Rehana S, Alam MS, Islam KS, Samad MA (2009) Influence of growth regulators on shoot proliferation and plantlet production from shoot tips of banana. Prog Agric 20:9–16. https://doi.org/10.3329/PA.V20I1-2.16840
Sahijram L, Soneji JR, Bollamma KT (2003) Analyzing somaclonal variation in micropropagated bananas (Musa spp.). In Vitro Cell Dev Biol Plant 39:551–556. https://doi.org/10.1079/IVP2003467
Schavemaker CM, Jacobsen E (1995) Development of a cyclic somatic embryogenesis regeneration system for leek (Allium ampeloprasum L.) using zygotic embryos. Plant Cell Rep 14:227–231. https://doi.org/10.1007/BF00233638
Shao X, Wu S, Dou T et al (2020) Using CRISPR/Cas9 genome editing system to create MaGA20ox2 gene-modified semi-dwarf banana. Plant Biotechnol J 18:17–19. https://doi.org/10.1111/PBI.13216
Strosse H, Domergue R, Panis B et al (2003) Banana and plantain embryogenic cell suspensions. INIBAP Technical Guidelines 8. The International Network for the Improvement of Banana and Plantain, Montpellier, France
Strosse H, Schoofs H, Panis B et al (2006) Development of embryogenic cell suspensions from shoot meristematic tissue in bananas and plantains (Musa spp.). Plant Sci 170:104–112. https://doi.org/10.1016/j.plantsci.2005.08.007
Subrahmanyeswari T, Gantait S (2022) Biotechnology of banana (Musa spp.): multi-dimensional progress and prospect of in vitro–mediated system. Appl Microbiol Biotechnol 106:3923–3947. https://doi.org/10.1007/S00253-022-11973-4
Sukhada M, Prathibha KY, Soumya HD, Ranganath RM (2010) The origin and development of secondary somatic embryos in banana cv. Nanjangud Rasbale (Silk gp. Rasthali, AAB). Acta Hortic 865:349–351. https://doi.org/10.17660/ActaHortic.2010.865.49
Tripathi JN, Oduor RO, Tripathi L (2015) A high-throughput regeneration and transformation platform for production of genetically modified banana. Front Plant Sci 6:1–13. https://doi.org/10.3389/FPLS.2015.01025
Tripathi L, Ntui VO, Tripathi JN (2020) CRISPR/Cas9-based genome editing of banana for disease resistance. Curr Opin Plant Biol 56:118–126. https://doi.org/10.1016/J.PBI.2020.05.003
Uma S, Karthic R, Kalpana S et al (2021a) A novel temporary immersion bioreactor system for large scale multiplication of banana (Rasthali AAB—Silk). Sci Rep 11:20371. https://doi.org/10.1038/s41598-021-99923-4
Uma S, Kumaravel M, Backiyarani S et al (2021b) Somatic embryogenesis as a tool for reproduction of genetically stable plants in banana and confirmatory field trials. Plant Cell Tissue Organ Cult 147:181–188. https://doi.org/10.1007/s11240-021-02108-0
Uma S, Karthic R, Kalpana S, Backiyarani S (2023) Evaluation of temporary immersion bioreactors for in vitro micropropagation of banana (Musa spp.) and genetic fidelity assessment using flow cytometry and simple-sequence repeat markers. Afr J Bot 157:553–565. https://doi.org/10.1016/J.SAJB.2023.04.006
Wang X, Yu R, Li J (2021) Using genetic engineering techniques to develop banana cultivars with Fusarium wilt resistance and ideal plant architecture. Front Plant Sci 11:617528. https://doi.org/10.3389/FPLS.2020.617528
Wang J, Gan S, Zheng Y et al (2022) Banana somatic embryogenesis and biotechnological application. Tropical Plants 1:1–13. https://doi.org/10.48130/TP-2022-0012
Wilken D, Jiménez Gonzalez E, Gerth A et al (2014) Effect of immersion systems, lighting, and TIS designs on biomass increase in micropropagating banana (Musa spp. cv. “Grande Naine” AAA). In Vitro Cell Dev Biol Plant 50:582–589. https://doi.org/10.1007/s11627-014-9605-5
Zorrilla-Fontanesi Y, Pauwels L, Panis B et al (2020) Strategies to revise agrosystems and breeding to control Fusarium wilt of banana. Nat Food 1(10):599–604. https://doi.org/10.1038/s43016-020-00155-y
Funding
The authors acknowledge the Tripura Biotechnology Council, Govt of Tripura, India, for partial financial support. RK acknowledges Young Scientist Award received from SYST-SEED Division (SP/YO/124/2017(G)), DST, Govt of India.
Author information
Authors and Affiliations
Contributions
SU: administration, funding acquisition, supervision, validation. RK: conceptualization, visualization, investigation, methodology, formal analysis, data curation, writing—original draft—review & editing. SK: investigation, data curation, formal analysis, writing—review & editing. SB: supervision, formal analysis, writing—review & editing. MK: investigation, methodology, formal analysis, writing—review & editing. SS: investigation, methodology. MSS: investigation, methodology, supervision. PD: investigation, methodology. All authors read, and approved final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Additional information
Communicated by Wagner Campos Otoni.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uma, S., Karthic, R., Kalpana, S. et al. An efficient embryogenic cell suspension culture system through secondary somatic embryogenesis and regeneration of true-to-type plants in banana cv. Sabri (silk subgroup AAB). Plant Cell Tiss Organ Cult 155, 313–322 (2023). https://doi.org/10.1007/s11240-023-02570-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02570-y