Abstract
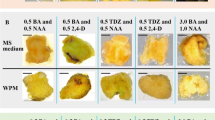
Browning, as the most important problem to be solved in callus differentiation, occupies an important position in tissue culture. In this study, plant regeneration was induced using leaves from germinated Isatis indigotica Fortune seedlings and mature plants as explants. Activated carbon (AC) and a solidification agent (Phytagel) were added simultaneously to inhibit browning. We measured the total phenol content and the PPO and POD activities in callus-induced seedling leaves and mature plant leaves of different ploidy at various stages. The results demonstrated that single plant growth regulators could induce calli with higher rooting and browning rates. Despite the addition of AC and phytagel to the medium, half of the mature plant calli turned yellow and eventually died. The total phenol content and POD activity in calli from mature leaves showed significant differences within 0–4 weeks (P < 0.05) but were still higher than the biochemical parameters in calli induced from seedlings, while the PPO activity did not differ significantly (P > 0.05). Mature materials of different ploidy yielded similar results. The addition of 2 mg·L−1 BAP, 0.1 mg·L−1 NAA, and 1.5 mg·L−1 AC and solidification with 0.2% phytagel in MS medium resulted in the highest callus regeneration rate and the lowest browning rate. During dedifferentiation, the browning of the two may be associated with PPO activity and total phenol content.
Key message
The regeneration system of Isatis indigotica Fortune was optimized and the browning problem was solved. The browning of callus is related to PPO activity and the total phenol content.

Similar content being viewed by others
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- Isatis indigotica Fortune:
-
I. indigotica
- BAP:
-
6-benzylaminopurine
- NAA:
-
1-naphthylacetic acid
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- KT:
-
Kinetin
- MS:
-
Murashige and skoog
- CIM:
-
Callus induction medium
- PPO:
-
Polyphenol oxidase
- POD:
-
Peroxidase
- AC:
-
Activated carbon
- ASA:
-
Ascorbic acid
- D:
-
Day
- H:
-
Hour
- Min:
-
Minute
References
Abdallat AM, Suwwan J, Zoubi B (2011) Agrobacteriumtumefaciens-mediated transformation of callus cells of Crataegusaronia. Plant Cell Tiss Org 104(1):31–39. https://doi.org/10.1007/s11240-010-9798-1
Aghayeh R, Bahram A, Ahmad B, Leila S, Ali T (2021) The first successful report: control of Browning Problem inin vitroCulture of iranian Seedless Barberry, aMedicinally important species. Erwerbs-Obstbau 63(3):319–329. https://doi.org/10.1007/S10341-021-00574-6
Chen R, Li Q, Tan H, Chen J, Xiao Y, Ma R, Gao S, Zerbe P, Chen W, Zhang L (2015) Gene-to-metabolite network for biosynthesis of lignans in MeJA-elicited Isatis indigotica hairy root cultures. Front Plant Sci 6:952. https://doi.org/10.3389/fpls.2015.00952
Chen S, Zhu Z, Ma H, Yang J, Guo Q (2014) DNA barcodes for discriminating the medicinal plant Isatis indigotica Fort. (Cruciferae) and its adulterants. Biochem Syst Ecol 57:287–292. https://doi.org/10.1016/j.bse.2014.08.007
Duan Y, Su Y, Chao E, Zhang GR, Zhao FL, Xue T, Sheng W, Teng JT, Xue JP (2019) Callus-mediated plant regeneration in Isodon amethystoides using young seedling leaves as starting materials. Plant Cell Tiss Org 136(2):247–253. https://doi.org/10.1007/s11240-018-1510-x
Garcia A, Wichers H, Wichers HJ (2007) Decrease of the IgE-binding by Mal d 1, the major apple allergen, by means of polyphenol oxidase and peroxidase treatments. Food Chem 103(1):94–100. https://doi.org/10.1016/j.foodchem.2006.07.029
Gerszberg A (2018) Tissue culture and genetic transformation of cabbage (Brassica oleracea var. capitata): an overview. Planta 248:1037–1048. https://doi.org/10.1007/s00425-018-2961-3
Gong ZQ, Li DJ, Liu CQ, Cheng AW, Wang WL (2015) Partial purification and characterization of polyphenol oxidase and peroxidase from chestnut kernel. Lwt-Food Sci Technol 60(2):1095–1099. https://doi.org/10.1016/j.lwt.2014.10.012
He Y, Liang S, Zheng H, Zhang F, Sun B (2019) Callus Induction and Plant Regeneration in Chinese Kale callus induction and plant regeneration in chinese kale. AIP Conference Proceedings 2058(1):1–4. https://doi.org/10.3724/sp.j.1259.2011.00569
Kang M, Wu H, Yang Q, Huang H, Liu J (2020) A chromosome-scale genome assembly of Isatis indigotica, an important medicinal plant used in traditional chinese medicine. Hortic Res-England 7(1):10. https://doi.org/10.1038/s41438-020-0240-5
Ke SY, Zhang SZ, Liu YJ (2009) Changes of antioxidant enzyme activity during vitrification of Isatis indica in vitro. Dissertation North China Agricultural Journal 24(3):5. https://doi.org/10.7668/hbnxb.2009.03.033
Koohi S, Nasernejad B, Zare MH, Elahifard M, Shirazian S, Ghadiri M (2020) Extraction of oxidative enzymes from green tea leaves and optimization of extraction conditions. Chem Eng Technol 43(12):2548–2556. https://doi.org/10.1002/ceat.202000344
Leng P, Su S, Wei F, Yu F, Duan Y (2009) Correlation between browning, total phenolic content, polyphenol oxidase and several antioxidation enzymes during pistachio tissue culture. Acta Hort 829:127–132. https://doi.org/10.17660/ActaHortic.2009.829.17
Lim WY, Cheun CF, Wong CW (2019) Inhibition of enzymatic browning in sweet potato (Ipomoea batatas (L.)) with chemical and natural anti-browning agents. J Food Process Pres 43:11. https://doi.org/10.1111/jfpp.14195
Li Y, Zhao J, Chen H, Yu X, Li H, Zhang Y, Feng L, Wu Z, Xie W, Hou D, Yu M (2023) Plant regeneration via callus-mediated organogenesis in commercial variety of Chuanbeichai No. 1 in Bupleurum chinense DC. Plant Biotechnol Rep 17(2):159–169. https://doi.org/10.1007/s11816-022-00772-y
Meng Q, Liu ZY, Zhang Y, Liu C, Ren F, Feng H (2014) Effects of antibiotics on in vitro-cultured cotyledons. In Vitro Cell Dev-Pl 50(4):436–441. https://doi.org/10.1007/s11627-014-9595-3
Ndakidemi CF, Mneney E, Ndakidemi PA (2014) Effects of ascorbic acid in controlling lethal browning in culture of using nodal segments. Am J Plant Sci 05(1):187–191. https://doi.org/10.4236/ajps.2014.51024
Santos GS, Sinoti SBP, De Almeida FTC, Silveira D, Simeoni LA, Gomes-Copeland KKP (2020) Use of galantamine in the treatment of Alzheimer’s disease and strategies to optimize its biosynthesis using the in vitro culture technique. Plant Cell Tiss Org: An International Journal on in Vitro Culture of Higher Plants 143(1):13–29. https://doi.org/10.1007/s11240-020-01911-5
Schuch MW, Cellini A, Masia A, Marino G (2010) Aluminium-induced effects on growth, morphogenesis and oxidative stress reactions in in vitro cultures of quince. Sci Hortic-Amsterdam 125(2):151–158. https://doi.org/10.1016/j.scienta.2010.03.007
Sellés M, Viladomat F, Bastida J, Codina C (1999) Callus induction, somatic embryogenesis and organogenesis in Narcissus confusus: correlation between the state of differentiation and the content of galanthamine and related alkaloids. Plant Cell Rep 18(7–8):646–651. https://doi.org/10.1007/s002990050636
Sharma H, Chawla N, Dhatt AS (2019) Nutraceutical content and free radical scavenging capacity of brinjal (Solanum melongena L.) genotypes. Sci Hortic-Amsterdam 244:294–303. https://doi.org/10.1016/j.scienta.2018.09.060
Soltani M, Samari E, Vazirifar S, Ahmadian CN, Sharifi M, Fotovat R (2023) Putrescine induces lignans biosynthesis through changing the oxidative status and reprogramming amino acids and carbohydrates levels in Linum album hairy roots. Plant Cell Tiss Org 153(2):387–402. https://doi.org/10.1007/s11240-023-02479-6
Sundararajan S, Rajendran V, Sivakumar HP, Kumariah M, Ramalingam S (2022) Growth modulation by nitric oxide donor sodium nitroprusside in in vitro plant tissue cultures–A review. Biologia 77(7):1699–1711. https://doi.org/10.1007/s11756-022-01027-5
Tang W, Newton, RJ (2004) Increase of polyphenol oxidase and decrease of polyamines correlate with tissue browning in Virginia pine (Pinus virginiana Mill.). Plant Sci 167(3):621–628. https://doi.org/10.1016/j.plantsci.2004.05.024
Wang L, Xue YN, Li ZW, Zhang W, Ji XP, Fan Z, Li YJ (2018) Efficacy and Safety of Indigo Naturalis in Combination with Narrow-Band Ultraviolet B for Treatment of Pityriasis Rosea: A Meta-Analysis. Evid Based Complement Alternat Med 2018:6816981. https://doi.org/10.1155/2018/6816981
Wu G, Wei X, Wang X, Wei Y (2021) Changes in biochemistry and histochemical characteristics during somatic embryogenesis in Ormosia henryi Prain. Plant Cell Tiss Org 144:505–517. https://doi.org/10.1007/s11240-020-01973-5
Xiao M, Tian J, Zhou Y, Xu X, Tong X (2020) Efficacy of Huoxiang Zhengqi dropping pills and Lianhua Qingwen granules in treatment of COVID-19: a randomized controlled trial. Pharmacol Res 161:105–126. https://doi.org/10.1016/j.phrs.2020.105126
Xu CJ, Ru ZW, Li L, Zeng BY, Huang JM, Huang W, Hu O (2015) The Effects of Polyphenol Oxidase and Cycloheximide on the Early Stage of Browning in Phalaenopsis Explants. Hortic Plant J 1(3):172–180. https://doi.org/10.16420/j.issn.2095-9885.2015-0030
Xu K, Zhang X, Fan C, Chen F, Zhu J, Zhang S, Chen Q, Fu Y (2017) A callus transformation system for gene functional studies in soybean. J Integr Agr 16(9):1913–1922. https://doi.org/CNKI:SUN:ZGNX.0.2017-09-004
Yang H, Guo X, Lu R, Niu B, Pasapula V, Pei H, Cai F, Ying X, Chen F (2009) Changes in morphology and biochemical indices in browning callus derived from Jatropha curcas hypocotyls. Plant Cell Tiss Org 98(1):11–17. https://doi.org/10.1007/s11240-009-9533-y
Yao SC, Ling ZZ, Cui LI, Hui-Zhen LU, Zhang ZJ (2011) Research on differentiation and plant regeneration from in vitro leaf tissues of chirita medica d.fang ex w.t.wang. Seed 30(12):30–29. https://doi.org/10.1016/j.jnca.2010.09.003
Funding
This work was supported by the Jiangsu Modern Agricultural Industrial Technology System Construction Project (JATS|[2022]461, JATS[2022]291).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: YS, MW, QG. Performed the experiments: YS, MW, QG. Analysed the data: YS, MW, QG, JH, KZ, JH. Wrote the paper: YS, MW, QG, JH, KZ, JH.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
We thank all authors for their contributions to this work.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Christell van der Vyver.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yong Su and Min Wei share co-first authorship.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Su, Y., Wei, M., Guo, Q. et al. Investigating the relationships between callus browning in Isatis indigotica Fortune, total phenol content, and PPO and POD activities. Plant Cell Tiss Organ Cult 155, 175–182 (2023). https://doi.org/10.1007/s11240-023-02567-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02567-7