Abstract
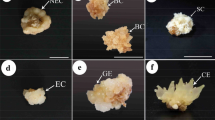
Bambusa nutans Wall. is a clump-forming, evergreen bamboo species that is most often found in Southeast Asian forests. Comparative activities of nitrate reductase (NR), glutamine synthetase (GS) and peroxidase (POX) as well as expression of peroxidase isozymes during somatic embryogenesis (SE) were investigated in the segregated embryogenic callus (EC) and non-embryogenic callus (NEC) from the same genotype (CPC-648) of B. nutans Wall. The EC was compact, with large prominent nuclei and dense cytoplasm, while the NEC was fragile, with rudimentary nuclei and hyaline cytoplasm. SE in EC encompassed induction, maturation and germination stages each of 30 days on MS medium supplemented with ascorbic acid 50 mg L− 1, citric acid 25 mg L− 1, cysteine 25 mg L− 1and glutamine 100 mg L− 1 + 2, 4-D 2 mg L− 1 + coconut water 10%. EC exhibited dedifferentiation and growth leading to SE, while NEC remained amorphous loose lump throughout. EC had lower NR and POX activities and less number of peroxidase isozymes but higher GS activity than NEC during three stages of SE. NR activity in EC followed a trend as induction > germination > maturation; GS activity, maturation > germination > induction and POX activity. EC exhibited less peroxidase isozymes than NEC. SE is an epigenetically regulated process that leads to the expression of enzymes involved in primary metabolism in EC and secondary metabolism in NEC. GS activity corresponding to SE process may be developed as biochemical marker. Peroxidase activity and isozyme expression in NEC express a disorderly oxidative stress scenario.
Key message
1. This investigation provides insight into the physiological and biochemical changes during SE and factors contributing to it.
2. GSA has a strong correlation with SE and can be a potential biochemical marker for the process.
3. Peroxidase activity and expression of its isozymes denote a chaotic situation rather than a marker of SE.





Similar content being viewed by others
References
Bajaj Y, Bopp M, Bajaj S (1973) Patterns of peroxidases and differentiation in Sinapis alba L. Phytomorphology 23:43–52
Bapat SA, Rawal SK, Mascarenhas AF (1992) Isozyme profiles during ontogeny of somatic embryos in wheat (Triticum aestivum L). Plant Sci 82(2):235–242. https://doi.org/10.1016/0168-9452(92)90225-B
Bernard SM, Habash DZ (2009) The importance of cytosolic glutamine synthetase in nitrogen assimilation and recycling. New Phytol 182(3):608–620. https://doi.org/10.1111/j.1469-8137.2009.02823.x
Bian Z, Wang Y, Zhang X, Li T, Grundy S, Yang Q, Cheng R (2020) A review of environment effects on nitrate accumulation in leafy vegetables grown in controlled environments. Foods 9(6):732. https://doi.org/10.3390/foods9060732
Bonfill M, Cusidó RM, Palazón J, Canut E, Piñol MT, Morales C (2003) Relationship between peroxidase activity and organogenesis in Panax ginseng calluses. Planr Cell Tissue Organ Cult 73(1):37–41. https://doi.org/10.1023/A:1022615112745
Carlsson J, Svennerstam H, Moritz T, Egertsdotter U, Ganeteg U (2017) Nitrogen uptake and assimilation in proliferating embryogenic cultures of Norway spruce—investigating the specific role of glutamine. PLoS ONE 12(8):e0181785. https://doi.org/10.1371/journal.pone.0181785
Carlsson J, Egertsdotter U, Ganeteg U, Svennerstam H (2019) Nitrogen utilization during germination of somatic embryos of Norway spruce: revealing the importance of supplied glutamine for nitrogen metabolism. Trees 33(2):383–394. https://doi.org/10.1007/s00468-018-1784-y
Chan A, Stasolla C (2023) Light induction of somatic embryogenesis in Arabidopsis is regulated by PHYTOCHROME E. Plant Physiol Biochem 195:163–169. https://doi.org/10.1016/j.plaphy.2023.01.007
Coppens L, Dewitte D (1990) Esterase and peroxidase zymograms from barley (Hordeum vulgare L.) callus as a biochemical marker system of embryogenesis and organogenesis. Plant Sci 67(1):97–105. https://doi.org/10.1016/0168-9452(90)90055-S
Fan X-x, Bian Z-h, Song B, Xu H (2022) Transcriptome analysis reveals the differential regulatory effects of red and blue light on nitrate metabolism in pakchoi (Brassica campestris L). J Integr Agric 21(4):1015–1027. https://doi.org/10.1016/S2095-3119(21)63784-X
Hirel B, Gadal P (1980) Glutamine synthetase in rice: a comparative study of the enzymes from roots and leaves. Plant Physiol 66(4):619–623. https://doi.org/10.1104/pp.66.4.619
Jaworski EG (1971) Nitrate reductase assay in intact plant tissues. Biochem Biophys Res Commun 43(6):1274–1279. https://doi.org/10.1016/S0006-291X(71)80010-4
Kamada H, Harada H (1984) Changes in nitrate reductase activity during somatic embryogenesis in carrot. Biochemie und Physiologie der Pflanzen 179(5):403–410. https://doi.org/10.1016/S0015-3796(84)80016-5
Kenis JD, Silvente ST, Luna CM, Campbell WH (1992) Induction of nitrate reductase in detached corn leaves: the effect of the age of the leaves. Physiol Plant 85(1):49–56. https://doi.org/10.1111/j.1399-3054.1992.tb05262.x
Lowry O, Rosebrough N, Farr AL, Randall R (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Mehta R, Sharma V, Sood A, Sharma M, Sharma RK (2011) Induction of somatic embryogenesis and analysis of genetic fidelity of in vitro-derived plantlets of Bambusa nutans Wall., using AFLP markers. Eur J For Res 130(5):729–736. https://doi.org/10.1007/s10342-010-0462-4
Méndez-Hernández HA, Ledezma-Rodríguez M, Avilez-Montalvo RN, Juárez-Gómez YL, Skeete A, Avilez-Montalvo J, De-la-Peña C, Loyola-Vargas VM (2019) Signaling overview of plant somatic embryogenesis. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.00077
Neves M, Correia S, Cavaleiro C, Canhoto J (2021) Modulation of organogenesis and somatic embryogenesis by ethylene: an overview. Plants 10(6):1208. https://doi.org/10.3390/plants10061208
Oulbi S, Kohaich K, Baaziz M, Belkoura I, Loutfi K (2021) Peroxidase enzyme fractions as markers of somatic embryogenesis capacities in Olive (Olea europaea L). Plants 10(5):901. https://doi.org/10.3390/plants10050901
Pateman JA (1969) Regulation of synthesis of glutamate dehydrogenase and glutamine synthetase in micro-organisms. Biochem J 115(4):769–775. https://doi.org/10.1042/bj1150769
Rajput DS, Ram B, Rathore TS (2021) In vitro regeneration of Bambusa nutans Wall. via somatic embryogenesis and evaluation of genetic fidelity using ISSR markers. Plant Cell Biotechnology And Molecular Biology 22(15–16):145–155
Rao NR, Naithani SC, Singh YD (1982) Physiological and biochemical changes associated with cotton fibre development. II. Auxin oxidising system. Physiol Plant 55(2):204–208. https://doi.org/10.1111/j.1399-3054.1982.tb02288.x
Rao KV, Suprasanna P, Reddy GM (1990) Biochemical changes in embryogenic and non-embryogenic calli of Zea mays L. Plant Sci 66(1):127–130. https://doi.org/10.1016/0168-9452(90)90177-P
Rodríguez MJP, Suárez MF, Heredia R, Ávila C, Breton D, Trontin J-F, Filonova L, Bozhkov P, Von Arnold S, Harvengt L, Cánovas FM (2006) Expression patterns of two glutamine synthetase genes in zygotic and somatic pine embryos support specific roles in nitrogen metabolism during embryogenesis. New Phytol 169(1):35–44. https://doi.org/10.1111/j.1469-8137.2005.01551.x
Sachdev S, Ansari SA, Ansari MI, Fujita M, Hasanuzzaman M (2021) Abiotic stress and reactive oxygen species: generation, signaling, and defense mechanisms. Antioxidants 10(2):277. https://doi.org/10.3390/antiox10020277
Salaün C, Lepiniec L, Dubreucq B (2021) Genetic and molecular control of somatic embryogenesis. Plants 10(7):1467. https://doi.org/10.3390/plants10071467
Sarkar AK, Rai E, Shah SN, Bouddha S, Bansal YK, Ansari SA (2010) Changes in enzyme activities, soluble sugars and phenols during in vitro adventitious shoot regeneration from leaflet explants of Albizia procera (Roxb.) Benth. New Forest 40(3):323–334. https://doi.org/10.1007/s11056-010-9202-3
Shirin F, Bhadrawale D, Mishra JP, Sonkar MK, Maravi S (2020) Evaluation of biochemical changes during different stages of somatic embryogenesis in a vulnerable timber tree Dalbergia latifolia (Indian rosewood). In Vitro Cellular & Developmental Biology - Plant 56(6):894–902 https://doi.org/10.1007/s11627-020-10099-4
Sivasankar S, Oaks A (1996) Nitrate assimilation in higher plants: the effects of metabolites and light. Plant Physiol Biochem (France) 34(5):609–620
Somleva MN, Schmidt EDL, de Vries SC (2000) Embryogenic cells in Dactylis glomerata L. (Poaceae) explants identified by cell tracking and by SERK expression. Plant Cell Rep 19(7):718–726. https://doi.org/10.1007/s002999900169
Takatsuka H, Umeda M (2015) Epigenetic control of cell division and cell differentiation in the root apex. Front Plant Sci 6. https://doi.org/10.3389/fpls.2015.01178
Thorpe TA, Thanh Van MT, Gaspar T (1978) Isoperoxidases in epidermal layers of tobacco and changes during organ formation in vitro. Physiol Plant 44(4):388–394. https://doi.org/10.1111/j.1399-3054.1978.tb01643.x
Tsala G, Omokolo D, Balange A (1996) In vitro culture of cocoyam shoot tips, variation of phenol and protein contents and peroxidase and IAA-oxidase activities during adventitious bud formation. Plant Peroxidase Newsletter 8:17–24
Wickramasuriya AM, Dunwell JM (2015) Global scale transcriptome analysis of Arabidopsis embryogenesis in vitro. BMC Genomics 16(1):301. https://doi.org/10.1186/s12864-015-1504-6
Xue W, Liu N, Zhang T, Li J, Chen P, Yang Y, Chen S (2022) Substance metabolism, IAA and CTK signaling pathways regulating the origin of embryogenic callus during dedifferentiation and redifferentiation of cucumber cotyledon nodes. Sci Hort 293:110680. https://doi.org/10.1016/j.scienta.2021.110680
Yue J, Dong Y, Du C, Shi Y, Teng Y (2022) Transcriptomic and physiological analyses reveal the acquisition of somatic embryogenesis potential in Agapanthus praecox. Sci Hort 305:111362. https://doi.org/10.1016/j.scienta.2022.111362
Funding
The authors are thankful to the Researchers Supporting Project Number (RSP-2023R86), King Saud University, Riyadh, Saudi Arabia for financial support.
Author information
Authors and Affiliations
Contributions
ALL authors contributed to this study. Material preparation, data collection, and analysis were performed and first draft of the manuscript by DSR and TSR. SAA, MF, AAA, EMA and AS checked the data analysis, provided interpretation and finalized the manuscript to be submitted to the journal.
Corresponding authors
Ethics declarations
Ethical standard
Research work reported in the MS complies all ethical responsibilities.
Conflict of interest
the authors declare that they have no conflict of interest.
Additional information
Communicated by Ranjith Pathirana.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rajput, D.S., Rathore, T.S., Ansari, S.A. et al. Biochemical changes in embryogenic and non-embryogenic callus of Bambusa nutans Wall. during somatic embryogenesis. Plant Cell Tiss Organ Cult 155, 127–135 (2023). https://doi.org/10.1007/s11240-023-02559-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02559-7