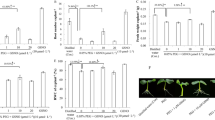
Abstract
Water stress is one constraint of the development potential of many plant species under in vitro tissue culture. The present study aims to assess the responses of anise callus tissues to two concentrations (5 and 7.5%) of PEG-6000 under darkness and light (12 h-photoperiod) for 30 days. The exposure of calli to water stress recorded morphological and physiological variations in fresh weight and water content in the callus decreased with increasing dry weight. Malondialdehyde content increased progressively with concentrations used with higher values under dark than in light. The activity of antioxidant enzymes (SOD, CAT, APX, and GPOX) was affected by PEG-6000 concentrations and culture conditions. APX and CAT increased gradually with 5 and 7.5% of PEG-6000 with higher values under light, but SOD enzyme reacted differently with a slight increase with 5% in light and higher ones under darkness with 7.5%. Additionally, GPOX activity decreased with both concentrations independently of light or darkness. The results revealed that anise calli under drought stress with 7.5% PEG-6000 were more resistant to the lack of water in our experimental conditions.
Key message
The calli increased antioxidant enzyme activities, associated with water loss, lipid peroxidation, and decreased fresh weight in light/dark with PEG treatment. Tissue culture applications might offer solutions to water scarcity.



Similar content being viewed by others
Data availability
The experiment data generated and analysed during the current study are available from the corresponding author on request.
Change history
16 June 2023
The original version of this article has been revised: In the Discussion section, a duplicate heading and paragraph following the subsection ‘In vitro callus induction and growth’ were removed.
Abbreviations
- PEG:
-
Polyethylene glycol
- MDA:
-
Malondialdehyde
- CAT:
-
Catalase
- SOD:
-
Superoxide dismutase
- APX:
-
Ascorbate peroxidase
- GPOX:
-
Guaiacol peroxidase
References
Aazami MA, Torabi M, Jalili E (2010) In vitro response of promising tomato genotypes for tolerance to osmotic stress. Afr J Biotechnol 9(26):4014–4017
Abid G, Ouertani RN, Jebara SH, Boubakri H, Muhovski Y, Ghouili E, Abdelkarim S, Chaieb O, Hidri Y, Kadri S, El Ayed M, Elkahoui S, Barhoumi F, Jebara M (2020) Alleviation of drought stress in faba bean (Vicia faba L.) by exogenous application of β-aminobutyric acid (BABA). Physiol Mol Biol Plant 26(6):1173–1186. https://doi.org/10.1007/s12298-020-00796-0
Aćimović M, Korać J, Jaćimović G, Oljača S, Đukanović L, Vuga-Janjatov V (2014) Influence of ecological conditions on seeds traits and essential oil contents in anise (Pimpinella anisum L). Not Bot Hortic Agrobot Cluj-Napoca 42(1):232–238
Adil M, Ren X, Jeong BR (2019) Light elicited growth, antioxidant enzymes activities and production of medicinal compounds in callus culture of Cnidium officinale Makino. J Photochem Photobiol B 196:111509. https://doi.org/10.1016/j.jphotobiol.2019.05.006
Afshari RT, Angoshtari R, Kalantari S (2011) Effects of light and different plant growth regulators on induction of callus growth in Rapeseed (‘Brassica napus L.’) genotypes. Plant Omics 4:60–67. https://doi.org/10.1016/j.jphotobiol.2019.05.006
Ahanger MA, Gul F, Ahmad P, Akram NA (2018) Environmental stresses and metabolomics deciphering the role of stress responsive metabolites. In: Ahmad P, Ahanger MA, Singh VP, Tripathi DK, Alyemeni MN (eds) Plant metabolites and regulation under environmental stress. Academic press, Cambridge, pp 53–67. https://doi.org/10.1016/B978-0-12-812689-9.00003-0
Ahmad MA, Javed R, Adeel M, Rizwan M, Yang Y (2020) PEG 6000-stimulated drought stress improves the attributes of in vitro growth, steviol glycosides production, and antioxidant activities in Stevia rebaudiana Bertoni. Plants 9:1552. https://doi.org/10.3390/plants9111552
Al Maghrabi AM, Rogers HJ, Francis D, Ochatt SJ (2017) PEG induces high expression of the cell cycle checkpoint gene WEE1 in embryogenic callus of Medicago truncatula: potential link between cell cycle checkpoint regulation and osmotic stress Front Plant Sci 81479. https://doi.org/10.3389/fpls.2017.01479
Ali H, Khan MA, Ullah N, Khan RS (2018) Impacts of hormonal elicitors and photoperiod regimes on elicitation of bioactive secondary volatiles in cell cultures of Ajuga bracteosa. J Photochem Photobiol B 183:242–250. https://doi.org/10.1016/j.jphotobiol.2018.04.044
Amer A, Omar H (2019) In-vitro propagation of the multipurpose egyptian medicinal plant Pimpinella anisum. Egypt Pharm J 18(3):25
Anderson MD, Prasad TK, Stewart CR (1995) Changes in isozyme profiles of catalase, peroxidase, and glutathione reductase during acclimation to chilling in mesocotyls of maize seedlings. Plant Physiol 109:1247–1257. https://doi.org/10.1104/pp.109.4.1247
André CM, Larondelle Y, Eversa D (2010) Dietary antioxidants and oxidative stress from a human and plant perspective: a review. Curr Nutr Food Sci 6:2–12. https://doi.org/10.2174/157340110790909563
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Arafa NM, Aly UI (2022) Induced biosynthesis of Acephenanthrylene in Callus Culture of Pimpinella anisum L., by yeast and phenylalanine application. Egypt J Chem. https://doi.org/10.21608/ejchem.2022.124157.5541
Assaha DVM, Liu L, Ueda A, Nagaoka T (2016) Effect of drought stress on growth, solute accumulation and membrane stability of leafy vegetable, huckleberry (Solanum scabrum Mill). J Environ Biol 37(1):107–114
Attree SM, Moore D, Sawhney VK, Fowke LC (1991) Enhanced maturation and desiccation tolerance of white spruce [Picea glauca (Moench) Voss] somatic embryos: effects of a non-plasmolysing water stress and abscisic acid. Ann Bot 68:519–525. https://doi.org/10.1093/oxfordjournals.aob.a088291
Bahadur A, Chatterjee A, Kumar R, Singh M, Naik PS (2011) Physiological and biochemical basis of drought tolerance in vegetables. Veg Sci 38:1–16 Online ISSN
Barba-Epsin G, Diaz-Vivancos P, Clemente-Moreno MJ et al (2010) Interaction between hydrogen peroxide and plant hormones during germination and the early growth of pea seedlings. Plant Cell Environ 33:981–994. https://doi.org/10.1111/j.1365-3040.2010.02120.x
Behbahani M, Shanehsazzadeh M, Hessami MJ (2011) Optimization of callus and cell suspension cultures of Barringtonia racemosa (Lecythidaceae family) for lycopene production. Sci Agric 68:69–76. https://doi.org/10.1590/S0103-90162011000100011
Bhatt D, Negi M, Sharma P et al (2011) Responses to drought induced oxidative stress in five finger millet varieties differing in their geographical distribution. Physiol Mol Biol Plant 17:347–353. https://doi.org/10.1007/s12298-011-0084-4
Boucelha L, Djebbar R, Abrous-Belbachir O (2019) The involvement of guaiacol peroxidase in embryo development of Vigna unguiculata (L.) Walp. during germination. Survival 5:39
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bressan RA, Hasegawa PM, Handa AK (1981) Resistance of cultured higher plant cells to Polyethylene glycol-induced water stress. Plant Sci Lett 21:23–30. https://doi.org/10.1016/0304-4211(81)90065-1
Bruc RC, Thayer WS (1983) Differential effect of lipid peroxidation on membrane fluidity as determined by electron spin resonance probes. Biochim Biophys Acta Biomembr 733(2):216–222. https://doi.org/10.1016/0005-2736(83)90525-4
Bueno P, Piqueras A, Kurepa J, Savoure A, Verbruggen N, Van Montagu M, Inze D (1998) Expression of antioxidant enzymes in response to abscisic acid and high osmoticum in tobacco BY-2 cell cultures. Plant Sci 138:27–34. https://doi.org/10.1016/S0168-9452(98)00154-X
Caversan A, Casassola A, Brammer SP (2016) Antioxydant responses of wheat plants under stress. Genet Mol Biol 39(1):1–6. https://doi.org/10.1590/1678-4685-GMB-2015-0109
Chand S, Sahrawat AK, Prakash D (1997) In vitro culture of Pimpinella anisum L. (Anise). J Plant Biochem Biotechnol 6:91–95. https://doi.org/10.1007/BF03263017
Chaves MM, Pereira JS, Maroco J (2003) Understanding plant responses to drought from genes to the whole plant. Funct Plant Biol 30:239–264. https://doi.org/10.1071/FP02076
Chen YM, Huang JZ, Hou TW, Pan IC (2019) Effects of light intensity and plant growth regulators on callus proliferation and shoot regeneration in the ornamental succulent Haworthia. Bot Stud 60(1):10. https://doi.org/10.1186/s40529-019-0257-y
Choudhury FK, Rivero RM, Blumwald E, Mittler R (2017) Reactive oxygen species, abiotic stress and stress combination. Plant J 90(5):856–867. https://doi.org/10.1111/tpj.13299
Comparot SM, Graham CM, Reid DM (2002) Methyl jasmonate elicits a differential antioxidant response in light-and dark-grown canola (Brassica napus) roots and shoots. Plant Growth Regul 38:21–30. https://doi.org/10.1023/A:1020970319190
De-Eknamkul W, Ellis BE (1985) Effects of auxins and cytokinins on growth and rosmarinic acid formation in cell suspension cultures of Anchusa officinalis. Plant Cell Rep 4:50–53. https://doi.org/10.1007/BF00269204
Dobisova T, Hrdinova V, Cuesta C et al (2017) Light controls cytokinin signaling via transcriptional regulation of constitutively active sensor histidine kinase CKI1. Plant physiol 174:387–404. https://doi.org/10.1104/pp.16.01964
Duran-Encalada JA, Paucar-Caceres A, Bandala ER, Wright GH (2017) The impact of global climate change on water quantity and quality: a system dynamics approach to the US–Mexican transborder region. Eur J Oper Res 256:567–581. https://doi.org/10.1016/j.ejor.2016.06.016
Easwar Rao D, Viswanatha CK (2020) Changes in the antioxidant intensities of seven different soybean (Glycine max (L.) Merr.) Cultivars during drought. J Food Biochem 44(2):e13118. https://doi.org/10.1111/jfbc.13118
El Dawayati MM, El Sharabasy S, Gantait S (2020) Light intensity-induced morphogenetic response and enhanced β-sitosterol accumulation in date palm (Phoenix dactylifera L. cv. Hayani) callus culture. Sugar Tech 22:1122–1129. https://doi.org/10.1007/s12355-020-00844-9
Feierabend J, Schaan C, Hertwig B (1992) Photoinactivation of catalase occurs under both high-and low-temperature stress conditions and accompanies photoinhibition of photosystem II. Plant Physiol 100:1554–1561. https://doi.org/10.1104/pp.100.3.1554
FAO (2018) The impact of disasters and crises on agriculture and food security. Food and Agriculture Organization of the United Nations, Rome, p 168
George EF, Hall MA, Klerk GJ (2008) Effects of the physical environment. Plant Propag Tissue Cult 1:423–464. https://doi.org/10.1007/978-1-4020-5005-3
Gratão PL, Prasad MNV, Cardoso PF, Lea PJ, Azevedo RA (2005) Phytoremediation: green technology for the clean-up of toxic metals in the environment. Braz J Plant Physiol 17:53–64. https://doi.org/10.1590/S1677-04202005000100005
Guo Z, Ou W, Lu S, Zhong Q (2006) Differential responses of anti-oxidative system to chilling and drought in four rice cultivars differing in sensitivity. Plant Physiol Biochem 44(11–12):828–836. https://doi.org/10.1016/j.plaphy.2006.10.024
Gupta A, Rico Medina A, Caño-Delgado AI (2020) The physiology of plant responses to drought. Science 368(6488):266–269. https://doi.org/10.1126/science.aaz7614
Hajihashemi S, Rajabpoor S, Djalovic I (2018) Antioxidant potential in Stevia rebaudiana callus in response to polyethylene glycol, paclobutrazol and gibberellin treatments. Physiol Mol Biol Plant 24:335–341. https://doi.org/10.1007/s12298-017-0498-8
Halliday KJ, Fankhauser C (2003) Phytochrome-hormonal signalling networks. New Phytol 157:449–463. https://doi.org/10.1046/j.1469-8137.2003.00689.x
Halliwell B, Gutteridge JMC (1984) Lipid peroxidation, oxygen radicals, cell damage and antioxidant therapy. Lancet 1:1396–1398. https://doi.org/10.1016/S0140-6736(84)91886-5
Harris D, Tripathi RS, Joshi A (2002) On-farm seed priming to improve crop establishment and yield in dry direct-seeded rice. Direct seeding Res. Strategy Opportumities International Research Institute, Manila, pp 231–240
Hasegawa PM, Bressan RA, Handa S, Handa AK (1984) Cellular mechanisms of tolerance to water stress [in tomatoes]. HortScience 19:371–377. https://doi.org/10.21273/HORTSCI.19.3.371
Hassanpour H (2021) Potential impact of red-blue LED light on callus growth, cell viability, and secondary metabolism of Hyoscyamus reticulatus. Vitr Cell Dev Biol 1–10. https://doi.org/10.1007/s11627-021-10232-x
Heidari N, Pouryousef M, Tavakkoli A, Saba J (2012) Effect of drought stress and harvesting date on yield and essential oil production of anise (Pimpinella anisum L.). Iran J Med Aromat Plant 28(1):121–130
Hoseini A, Salehi A, Sayyed RZ, Balouchi H, Moradi A, Piri R, Bahman FN, Poczai P, Ansari MJ, Al Obeid S, Datta R (2022) Efficacy of biological agents and fillers seed coating in improving drought stress in anise. Front Plant Sci 13:955512. https://doi.org/10.3389/FPLS.2022.955512
Hosseini NS, Ghasimi Hagh Z, Khoshghalb H (2020) Morphological, antioxidant enzyme activity and secondary metabolites accumulation in response of polyethylene glycol-induced osmotic stress in embryo-derived plantlets and callus cultures of Salvia leriifolia. Plant Cell Tissue Organ Cult 140:143–155. https://doi.org/10.1007/s11240-019-01718-z
Hussien FA, Osman MA, Idris TIM (2014) The influence of liquid media support, gelling agents and liquid overlays on performance of in vitro cultures of ginger (Zingiber officinale). Int J Sci Res Publ 4:2250–3153
Jan N, Qazi HA, Ramzan S, John R (2018) Developing stress-tolerant plants through in vitro tissue culture: family Brassicaceae. In: Gosal SS, Wani SH (eds) Biotechnologies of crop improvement. Springer, New York, pp 327–372. https://doi.org/10.1007/978-3-319-78283-6_10
Jazi ZG, Etemadi N, Aalipour H (2019) The physiological responses of four turfgrass species to drought stress. Adv Hortic Sci 33:381–390. https://doi.org/10.13128/ahs-23830
Judi M, Dehghani H, Janmohammadi M, Ebadi A (2004) The reaction of Pimpinella anisum to drought and salinity stress at germination stage. Shahed University, Iran, The proceedings of the second conference on herbal plants, p p77
Kamarudin ZS, Yusop MR, Tengku Muda Mohamed M, Ismail MR, Harun AR (2018) Growth performance and antioxidant enzyme activities of advanced mutant rice genotypes under drought stress condition. Agronomy 8:279: 1–15. https://doi.org/10.3390/agronomy8120279
Kaya MD, Okçu G, Atak M et al (2006) Seed treatments to overcome salt and drought stress during germination in sunflower (Helianthus annuus L). Eur J Agron 24:291–295. https://doi.org/10.1016/j.eja.2005.08.001
Kesavulu MM, Rao BK, Giri R et al (2001) Lipid peroxidation and antioxidant enzyme status in type 2 diabetics with coronary heart disease. Diabet Res Clin Pract 53:33–39. https://doi.org/10.1016/S0168-8227(01)00238-8
Kevers C, Bisbis B, Le Dily F, Billard JP, Huault C, Gaspar Th (1995) Darkness improves growth and delays necrosis in a nonchlorophyllous habituated sugarbeet callus: biochemical changes. In Vitro Cell Dev Biol 31:122–126. https://doi.org/10.1007/BF02632249
Kreis W (2006) In-vitro culturing techniques of medicinal plants. In: Kayser O, Quax W (eds) Medicinal plant biotechnology. From basic research to industrial application Wiley, Hoboken, p 157–185. https://doi.org/10.1002/9783527619771.ch8
Kumar A, Kumar VA (2000) Biotechnology for fruit crop improvement (International Book Distributing company Crop improvement, Lucknow, India), pp. 71-85.
Larson RA (1988) The antioxidants of higher plants. Phytochemistry 27:969–978. https://doi.org/10.1016/0031-9422(88)80254-1
Lei C, Bagavathiannan M, Wang H, Sharpe S, Meng W, Yu J (2021) Osmopriming with polyethylene glycol (PEG) for abiotic stress tolerance in germinating crop seeds: a review. Agronomy 11(11):2194. https://doi.org/10.3390/agronomy11112194
Li L, Van Staden J (1998) Effects of plant growth regulators on the antioxidant system in callus of two maize cultivars subjected to water stress. Plant Growth Regul 24:55–66. https://doi.org/10.1023/A:1005954532397
Lin H, Lie Z, Lin KT (2022) Editorial: light-nanomaterial interactions for energy efficient nanophotonic devices. Front Mater 9:1–3. https://doi.org/10.3389/fmats.2022.865312
Lum MS, Hanafi MM, Rafii YM, Akmar ASN (2014) Effect of drought stress on growth, proline and antioxidant enzyme activities of upland rice. J Anim Plant Sci 24:1487–1493
Luna CM, Pastori GM, Driscoll S et al (2005) Drought controls on H2O2 accumulation, catalase (CAT) activity and CAT gene expression in wheat. J Exp Bot 56:417–423. https://doi.org/10.1093/jxb/eri039
MacAdam JW, Nelson CJ, Sharp RE (1992) Peroxidase activity in the leaf elongation zone of tall fescue: I. spatial distribution of ionically bound peroxidase activity in genotypes differing in length of the elongation zone. Plant Physiol 99:872–878. https://doi.org/10.1104/pp.99.3.872
Mahmood I, Razzaq A, Hafiz IA et al (2012) Interaction of callus selection media and stress duration for in vitro selection of drought tolerant callus of wheat. Afr J Biotechnol 11(17):4000–4006. https://doi.org/10.5897/AJB11.2610
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Maurel C, Boursiac Y, Luu DT, Santoni V, Shahzad Z, Verdoucq L (2015) Aquaporins in plants. Physiol Rev 95:1321–1358. https://doi.org/10.1152/physrev.00008.2015
McClung CR (1997) Regulation of catalases in Arabidopsis. Free Radic Biol Med 23:489–496. https://doi.org/10.1016/S0891-5849(97)00109-3
McKersie BD, Bowley SR, Jones KS (1999) Winter survival of transgenic alfalfa over expressing superoxide dismutase. Plant Physiol 119:839–847. https://doi.org/10.1104/pp.119.3.839
Mehravi S, Ranjbar GA, Najafi-Zarrini H, Mirzaghaderi G (2020) Investigation of drought stress tolerance and adaptation in iranian endemic anise (Pimpinella anisum L.) genotypes. Plant Genet Res 7(1):77–102
Mohamed MH, Harris PJC, Henderson J (2000) In vitro selection and characterisation of a drought tolerant clone of Tagetes minuta. Plant Sci 159:213–222. https://doi.org/10.1016/S0168-9452(00)00339-3
Morison JIL, Baker NR, Mullineaux PM, Davies WJ (2008) Improving water use in crop production. Philos Trans R Soc B Biol Sci 363:639–658. https://doi.org/10.1098/rstb.2007.2175
Moubarak M, Sekhi YS, Al-furaiji AMMAR, Almehemdi A, Almarie AA (2021) Potential of hormones combination on callugensis of anise seeds and its antioxidant activity. Plant Cell Biotechnol Mol Biol 22(31):96–106
Munir N, Aftab F (2009) The role of poly ethylene glycol (PEG) pretreatment in improving sugarcane’s salt (NaCl) tolerance. Turk J Bot 33(6):407–415. https://doi.org/10.3906/bot-0806-6
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Ncise W, Daniels CW, Nchu F (2020) Effects of light intensities and varying watering intervals on growth, tissue nutrient content and antifungal activity of hydroponic cultivated Tulbaghia violacea L. under greenhouse conditions. Heliyon 6:1–7. https://doi.org/10.1016/j.heliyon.2020.e03906
Newton RJ, Sen S, Puryear JD (1989) Solute contributions to osmotic potential in loblolly pine (Pinus taeda L.) callus. J Plant Physiol 134:746–750. https://doi.org/10.1016/S0176-1617(89)80038-0
Obidiegwu JE, Bryan GJ, Jones HG, Prashar A (2015) Coping with drought: stress and adaptive responses in potato and perspectives for improvement. Front Plant Sci 6:542. https://doi.org/10.3389/fpls.2015.00542
Okla MK, Abdel-Mawgoud M, Alamri SA, Abbas ZK, Al-Qahtani WH, Al-Qahtani SM, Al Harbi NA, Hassan NHA, Selim S, Alruhaili MH et al (2021) Developmental stages-specific response of anise plants to laser-induced growth, nutrients accumulation, and essential oil metabolism. Plants 10:2591. https://doi.org/10.3390/plants10122591
Oneto CD, Otegui ME, Baroli I et al (2016) Water deficit stress tolerance in maize conferred by expression of an isopentenyltransferase (IPT) gene driven by a stress-and maturation-induced promoter. J Biotechnol 220:66–77. https://doi.org/10.1016/j.jbiotec.2016.01.014
Ozkur O, Ozdemir F, Bor M, Turkan I (2009) Physiochemical and antioxidant responses of the perennial xerophyte Capparis ovata desf. to drought. Environ Exp Bot 66:487–492. https://doi.org/10.1016/j.envexpbot.2009.04.003
Park WT, Kim YK, Kim YS et al (2011) In vitro plant regeneration and micropropagation of Liriope platyphylla. Plant Omics J 4(4):199–203
Park J, Jeong BG, Kim SI et al (2021) All-solid-state spatial light modulator with independent phase and amplitude control for three-dimensional lidar applications. Nat Nanotechnol 16:69–76. https://doi.org/10.1038/s41565-020-00787-y
Patade VY, Bhargava S, Suprasanna P (2011) Salt and drought tolerance of sugarcane under iso-osmotic salt and water stress: growth, osmolytes accumulation and antioxidant defense. J Plant Interact 6(4):275–282. https://doi.org/10.1080/17429145.2011.557513
Patade VY, Bhargava S, Suprasanna P (2012) Effects of NaCl and iso-osmotic PEG stress on growth, osmolytes accumulation and antioxidant defense in cultured sugarcane cells. Plant Cell Tissue Organ Cult 108:279–286. https://doi.org/10.1007/s11240-011-0041-5
Peever T, Higgins VJ (1989) Electrolyte leakage, lipoxygenase, and lipid peroxidation induced in tomato leaf tissue by specific elicitors from Cladosporium fulvum. Plant Physiol 90(3):867–875. https://doi.org/10.1104/pp.90.3.867
Pérez-Clemente RM, Gómez-Cadenas A (2012) In vitro tissue culture, a tool for the study and breeding of plants subjected to abiotic stress conditions. Recent Adv Plant Vitr Cult. https://doi.org/10.5772/50671
Prado K, BoursiacY, Tournaire-Roux C et al (2013) Regulation of Arabidopsis leaf hydraulics involves light-dependent phosphorylation of aquaporins in veins. Plant Cell 25:1029–1039. https://doi.org/10.1105/tpc.112.108456
Queiroz MS, Oliviera CES, Steiner F et al (2019) Drought stresses on seed germination and early growth of maize and sorghum. J Agric Sci 11:310–318. https://doi.org/10.5539/jas.v11n2p310
Rahim D, Kalousek P, Tahir N et al (2020) In vitro assessment of kurdish rice genotypes in response to PEG-induced drought stress. Appl Sci 10:4471. https://doi.org/10.3390/app10134471
Rao S, Jabeen F (2013) In-vitro selection and characterization of polyethylene glycol (PEG) tolerant callus lines and regeneration of plantlets from the selected callus lines in sugarcane (Saccharum officinarum L.). Physiol Mol Biol Plant 19:261–268. https://doi.org/10.1007/s12298-013-0162-x
Razavizadeh R, Adabavazeh F, Rostami F, Teimouri A (2017) Comparative study of osmotic stress effects on the defense mechanisms and secondary metabolites in Carum copticum seedling and callus. J Plant Process Funct 5:23–33
Reddy AR, Chaitanya KV, Jutur PP, Sumithra K (2004) Differential antioxidative responses to water stress among five mulberry (Morus alba L.) cultivars. Environ Exp Bot 52:33–42. https://doi.org/10.1016/j.envexpbot.2004.01.002
Rishla K, Sithara A, Deepa P (2017) Effect of NaCl stress on callus induction in Centella asiatica (L.) Urban. Eur J Pharm Sci 4(9):527–531
Ross IA (2001) Medicinal plants of the world. chemical constitutes, traditional and modern medicinal uses. Humana press, Totowa, pp 363–374. https://doi.org/10.1021/jm068020k
Sahoo MR, Devi TR, Dasgupta M, Nongdam P, Prakash N (2020) Reactive oxygen species scavenging mechanisms associated with polyethylene glycol mediated osmotic stress tolerance in chinese potato. Sci Rep 10:1–9. https://doi.org/10.1038/s41598-020-62317-z
Salisbury FJ, Hall A, Grierson CS, Halliday KJ (2007) Phytochrome coordinates Arabidopsis shoot and root development. Plant J 50:429–438. https://doi.org/10.1111/j.1365-313X.2007.03059.x
Sarker U, Oba S (2018) Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant Biol 18:258. https://doi.org/10.1186/s12870-018-1484-1
Sarmadi M, Karimi N, Palazón J, Ghassempour A, Mirjalili MH (2019) Improved effects of polyethylene glycol on the growth, antioxidative enzymes activity and taxanes production in a Taxus baccata L. callus culture. Plant Cell Tissue Organ Cult 137:319–328. https://doi.org/10.1007/s11240-019-01573-y
Shahrajabi̇an MH, Khoshkharam M, Wenli S, ̇Qi̇ C (2019) The effects of pretreatment factors on seed germination and seedling growth of anise (Pimpinella anisum L). Middle East J Sci 5:86–93. https://doi.org/10.23884/mejs.2019.5.1.09
Sirisha KB, Sujathamma P (2018) Pharmacognostic standardization of Pimpinella tirupatiensis bal. & Subr. An endemic to eastern ghats, Tirumala hills, Andhra Pradesh. J Pharmacogn Phytochem 7:1676–1682
Sofo A, Scopa A, Nuzzaci M, Vitti A (2015) Ascorbate peroxidase and catalase activities and their genetic regulation in plants subjected to drought and salinity stresses. Int J Mol Sci 16(6):13561–13578. https://doi.org/10.3390/ijms160613561
Soussi M, El Yaagoubi W, Nekhla H, El Hanafi L, Squalli W, Benjelloun M, El Ghadraoui L (2023) A multidimensional review of Pimpinella anisum and recommendation for future research to face adverse climatic conditions. Chemistry Africa. https://doi.org/10.1007/s42250-023-00633-3
Stark G (2005) Functional consequences of oxidative membrane damage. J Membr Biol 205:1–16. https://doi.org/10.1007/s00232-005-0753-8
Stasolla C, Zyl LV, Egertsdotter U et al (2003) The Effects of Polyethylene Glycol on Gene expression of developing White Spruce somatic embryos. Plant Physiol 131(1):49–60. https://doi.org/10.1104/pp.015214
Stefanello R, Garcia DC, Menezes NL, Castilhos G (2008) Efeito do estresse hídrico na germinação e no vigor de sementes de anis (Pimpinella anisum L.), funcho (Foeniculum vulgare Miller) e endro (Anethum graveolens L). Rev Bras Plant Med 10:68–74
Sun W, Shahrajabian MH, Cheng Q (2019) Anise (Pimpinella anisum L.), a dominant spice and traditional medicinal herb for both food and medicinal purposes. Cogent Biol 5:1673688. https://doi.org/10.1080/23312025.2019.1673688
Suran D, Tumendalai B, Bayarmaa GA (2016) In vitro seed germination and callus induction of Ferula ferulaeoides (Steud.) Korov (Apiaceae). Mong J Biol Sci 14(1–2):53–58. https://doi.org/10.22353/mjbs.2016.14.07
Tariq U, Ali M, Abbasi BH (2014) Morphogenic and biochemical variations under different spectral lights in callus cultures of Artemisia absinthium L. J Photochem Photobiol B Biol 130:264–271. https://doi.org/10.1016/j.jphotobiol.2013.11.026
Ungureanu N, Vlăduț V, Voicu G (2020) Water scarcity and waste water reuse in crop irrigation. Sustainability 12:9055. https://doi.org/10.3390/su12219055
Verslues PE, Ober ES, Sharp RE (1998) Root growth and oxygen relations at low water potentials. Impact of oxygen availability in polyethylene glycol solutions. Plant Physiol 116(4):1403–1412. https://doi.org/10.1104/pp.116.4.1403
Viji M, Maheswari P, Karuppanapandian T, Manoharan K (2012) Effect of polyethylene glycol and mannitol on somatic embryogenesis of pigeonpea, Cajanus cajan (L) Millsp. Afr J Biotechnol 11(45):10340–10349. https://doi.org/10.5897/AJB12.368
Wang J, Zhang L, Cao Y et al (2018) CsATAF1 positively regulates Drought stress tolerance by an ABA-Dependent pathway and by promoting ROS scavenging in Cucumber. Plant Cell Physiol 59:930–945. https://doi.org/10.1093/pcp/pcy030
Wu G, Ma L, Sayre RT, Lee CH (2020) Identification of the optimal light harvesting antenna size for highlight stress mitigation in plants. Front Plant Sci 11:1–11. https://doi.org/10.3389/fpls.2020.00505
Yang Y, Han C, Liu Q, Lin B, Wang J (2008) Effect of drought and low light on growth and enzymatic antioxidant system of Picea asperata seedlings. Acta Physiol Plant 30:433–440. https://doi.org/10.1007/s11738-008-0140-z
Yoshimura K, Yabuta Y, Ishikawa T, Shigeoka S (2000) Expression of spinach ascorbate peroxidase isoenzymes in response to oxidative stresses. Plant Physiol 123:223–234. https://doi.org/10.1104/pp.123.1.223
Zehtab-Salmasi S, Javanshir A, Omidbaigi R, Alyari H, Ghassemi-Golezani K (2001) Effects of water supply and sowing date on performance and essential oil production of anise (Pimpinella anisum L.). Acta Agron Hung 49:75–81. https://doi.org/10.1556/AAgr.49.2001.1.9
Zhu H, Xiaofeng L, Zhai W, Liu W (2017) Effects of low light on photosynthetic properties, antioxidant enzyme activity and anthocyanin accumulation in purple pak-choi (Brassica campestris ssp. Chinensis Makino). PLoS ONE 12:e0179305. https://doi.org/10.1371/journal.pone.0179305
Funding
This work was supported by the Ministry of Higher Education and Scientific Research (Algeria). No fund, grant were received. We express our gratitude for all members of both laboratories for their help.
Author information
Authors and Affiliations
Contributions
CD and DR were contributed to the study conception and design. CD, DR, LS and BL carried out experiments Callus establishment without and with PEG treatment were performed by LS and CD Physiological and enzymes activities were carried out by LS and BL with DR. All authors contributed to data analysis, reading and approved final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Patricia Marconi.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lamara, S., Boucelha, L., Djebbar, R. et al. Antioxidant activities in Pimpinella anisum L. callus responding to PEG-6000-induced water stress under light (12 h-photoperiod) and dark conditions. Plant Cell Tiss Organ Cult 154, 399–413 (2023). https://doi.org/10.1007/s11240-023-02530-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02530-6