Abstract
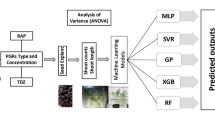
In this study, in vitro regeneration protocol of sorghum (Sorghum bicolor) was successfully established by using direct organogenesis from a mature zygotic embryo explant. The used basal medium encompassed Murashige and Skoog medium (MS) supplemented with 2–4 mg/L Benzylaminopurine (BAP) alone or with 0.25 mg/L Indole butyric acid (IBA) or Naphthalene acetic acid (NAA). Results demonstrated a significant impact of cytokinin-auxin on shoot count (1.24–3.46) and shoot length (2.80–3.47 cm). Maximum shoot count (3.46) and shoot length (3.97 cm) were achieved on the MS medium enriched with 2 mg/L BAP + 0.25 mg/L NAA and 2.0 mg/L BAP, respectively. To ascertain the impact of BAP alone, BAP + IBA, and BAP + NAA, the data were also analyzed by using a factorial regression model. Pareto chart and normal plots were used to check either the positive or negative impact of input variables on output variables. To further explore the association between BAP + IBA and BAP + NAA on shoot count and shoot length, contour and surface plots were also built. Three different artificial intelligence-based models along with four different performance metrics were utilized to validate the predicted results. Multilayer perceptron (MLP) model performed more efficiently (R2 = 0.799 for shoot count and R2 = 0.831 for shoot length) as compared to the decision tree-based algorithms of random forest (RF) – (R2 = 0.779 for shoot count and R2 = 0.786 for shoot length) and extreme gradient boost (XGBoost) – (R2 = 0.768 for shoot count and R2 = 0.781 for shoot length). As plant tissue culture protocol is a powerful tool for genetic engineering and genome editing of crops, integration of different artificial intelligence-based models can lead to improvement of sorghum with the aid of biotechnological tools.
Graphical abstract

Key message
-
Optimization of In vitro whole plant regeneration of sorghum using mature zygotic embryo explant and cytokinin-auxin combination
-
Data analysis through ANOVA, factorial regression anlaysis and machine learning
-
Presentation of data via Pareto charts, standarized plots, contour plots and surface plots
-
Better performance of ANN-based MLP model compared to decision tree based RF and XGBoost model.






Similar content being viewed by others
Data availability
The whole datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ANNs:
-
Artificial neural networks
- ANOVA:
-
Analysis of variance
- XGBoost:
-
Extreme Gradient Boosting
- LOO-CV:
-
Leave-one-out cross validation
- ML:
-
Machine learning
- MLP:
-
Multilayer Perceptron
- MS:
-
Murashige and Skoog Medium
- RF:
-
Random Forest
References
Aasim M, Akin F, Ali SA et al (2023a) Artificial neural network modeling for deciphering the in vitro induced salt stress tolerance in chickpea (Cicer arietinum L). Physiol Mol Biol Plants 29:289–304
Aasim M, Ali SA, Aydin S et al (2023b) Artificial intelligence–based approaches to evaluate and optimize phytoremediation potential of in vitro regenerated aquatic macrophyte Ceratophyllum demersum L. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-022-25081-3
Aasim M, Ali SA, Bekiş P, Nadeem MA (2022a) Light-emitting diodes induced in vitro regeneration of Alternanthera reineckii mini and validation via machine learning algorithms. Vitr Cell Dev Biol 58:816–825
Aasim M, Katirci R, Baloch F et al (2022b) Innovation in the breeding of common bean through a combined approach of in vitro regeneration and machine learning algorithms. Front Genet 13:897696
Aasim M, Katırcı R, Akgur O et al (2022c) Machine learning (ML) algorithms and artificial neural network for optimizing in vitro germination and growth indices of industrial hemp (Cannabis sativa L). Ind Crops Prod 181:114801
Aasim M, Sahin-Demirbag N, Khawar KM et al (2011) Direct axillary shoot regeneration from the mature seed explant of the hairy vetch (Vicia villosa Roth). Arch Biol Sci 63:757–762. https://doi.org/10.2298/ABS1103757A
Abbas F, Afzaal H, Farooque AA, Tang S (2020) Crop yield prediction through proximal sensing and machine learning algorithms. Agronomy 10:1046
Aggarwal CC (2018) Neural networks and deep learning. Springer, Cham. https://doi.org/10.1007/978-3-319-94463-0
Ahmad FI, Wagiran A, Abd Samad A et al (2016) Improvement of efficient in vitro regeneration potential of mature callus induced from malaysian upland rice seed (Oryza sativa cv. Panderas). Saudi J Biol Sci 23:S69–S77
Ahmar S, Gill RA, Jung K-H et al (2020) Conventional and molecular techniques from simple breeding to speed breeding in crop plants: recent advances and future outlook. Int J Mol Sci 21:2590
Ahmed RI, Rehman SU, Akhtar HL, Khan AM, Mahmood K, Ahmad RT, Anum W (2022) Optimization of in vitro responses of various explants sources in sorghum (Sorghum bicolor). Asian J Agric & Biol. https://doi.org/10.35495/ajab.2021.02.102
Alanagh EN, Garoosi G, Haddad R et al (2014) Design of tissue culture media for efficient prunus rootstock micropropagation using artificial intelligence models. Plant Cell, Tissue Organ Cult 117:349–359
Andiku C, Shimelis H, Laing M et al (2021) Assessment of sorghum production constraints and farmer preferences for sorghum variety in Uganda: implications for nutritional quality breeding. Acta Agric Scand Sect B—Soil Plant Sci 71:620–632
Arab MM, Yadollahi A, Eftekhari M et al (2018) Modeling and optimizing a new culture medium for in vitro rooting of G× N15 Prunus rootstock using artificial neural network-genetic algorithm. Sci Rep 8:1–18
Arab MM, Yadollahi A, Shojaeiyan A, Ahmadi H (2016) Artificial neural network genetic algorithm as powerful tool to predict and optimize in vitro proliferation mineral medium for G× N15 rootstock. Front Plant Sci 7:1526
Baskaran P, Jayabalan N (2005) In vitro plant regeneration and mass propagation system for Sorghum bicolor-a valuable major cereal crop. J Agric Technol 1:345–363
Baskaran P, Rajeswari B, Jayabalan N (2006) Development of an in vitro regeneration system in Sorghum [Sorghum bicolor (L.) Moench] using root transverse thin cell layers (tTCLs). Turk J Botany 30:1–9
Chen T, Guestrin C (2016) XGBoost. In: Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining. ACM, New York, NY, USA, pp 785–794
Dreger M, Mól R, Deja A et al (2019) Improved plant regeneration in callus cultures of Sorghum bicolor (L.) Moench. Vitr Cell Dev Biol 55:190–198
Dutta N (2021) Use of Sorghum/Jowar for various purposes in different states of India. Int J Mod Agric 10:2792–2801
Earl C, Belitz MW, Laffan SW et al (2021) Spatial phylogenetics of butterflies in relation to environmental drivers and angiosperm diversity across North America. Iscience 24:102239
Elango D, Sandoya G, Chopra S (2021) Techniques and tools of modern plant breeding. In: Ricroch A, Chopra S, Kuntz M (eds) Plant Biotechnology. Springer, Cham, pp 17–26
Farhadi S, Salehi M, Moieni A et al (2020) Modeling of paclitaxel biosynthesis elicitation in Corylus avellana cell culture using adaptive neuro-fuzzy inference system-genetic algorithm (ANFIS-GA) and multiple regression methods. PLoS ONE 15:1–16. https://doi.org/10.1371/journal.pone.0237478
Grootboom AW, OKennedy MM, Mkhonza NL et al (2008) In vitro culture and plant regeneration of sorghum genotypes using immature zygotic embryos as explant source. Int J Bot 4:450–455
Hesami M, Condori-Apfata JA, Valencia MV, Moham M (2020) Application of artificial neural network for modeling and studying in vitro genotype-independent shoot regeneration in wheat. Appl Sci 10:1–10. https://doi.org/10.3390/APP10155370
Hesami M, Jones AMP (2021) Modeling and optimizing callus growth and development in Cannabis sativa using random forest and support vector machine in combination with a genetic algorithm. Appl Microbiol Biotechnol 105:5201–5212
Hesami M, Naderi R, Tohidfar M (2019) Modeling and optimizing in vitro sterilization of chrysanthemum via multilayer perceptron-non-dominated sorting genetic algorithm-II (MLP-NSGAII). Front Plant Sci 10:1–13. https://doi.org/10.3389/fpls.2019.00282
Hodges HE, Walker HJ, Cowieson AJ et al (2021) Latent anti-nutrients and unintentional breeding consequences in australian Sorghum bicolor varieties. Front Plant Sci 12:625260
Katirci R (2015) S statistical approach to optimizing a Zn–Ni bath containing ED and TEA. Surf Rev Lett 22:1550015
Katırcı R, Yılmaz EK, Kaynar O, Zontul M (2021) Automated evaluation of Cr-III coated parts using Mask RCNN and ML methods. Surf Coat Technol 422:127571
Keijok WJ, Pereira RHA, Alvarez LAC et al (2019) Controlled biosynthesis of gold nanoparticles with Coffea arabica using factorial design. Sci Rep 9:1–10
Kendir H, Sahin-Demirbag N, Aasim M, Khawar KM (2009) In vitro plant regeneration from turkish Narbon Vetch (Vicia narbonensis L. var. Narbonensis L). Afr J Biotechnol 8:614–618
Kiondo F, Feyissa T, Ndakidemi PA et al (2014) Micropropagation of Dalbergia melanoxylon Guill. & Perr.: a threatened tree species. Am J Res Comm 2:43–52
Kirtis A, Aasim M, Katırcı R (2022) Application of artificial neural network and machine learning algorithms for modeling the in vitro regeneration of chickpea (Cicer arietinum L.). Plant Cell. Tissue Organ Cult 150:141–152
Kul M, Oskay KO, Erden F et al (2020) Effect of process parameters on the electrodeposition of zinc on 1010 steel: Central composite design optimization. Int J Electrochem Sci 15:9779–9795
Kumlay AM, Arslan N, Kaya C (2014) Farklı Fotoperiyot Şartlarında in vitro olarak Yetiştirilen patates (Solanum tuberosum L.)’lerde BAP’ın NAA ve IBA ile Birlikte Mikro Yumru Oluşturma Üzerine Etkileri. Iğdır Üni Fen Bilim Enst Der/Iğdır. Univ J Inst Sci Tech 4:73–82
Lewis CD (1982) Industrial and business forecasting methods: a practical guide to exponential smoothing and curve fitting. Butterworth-Heinemann, London
Malik SI, Rashid H, Yasmin T, Minhas NM (2004) Plant regeneration by somatic embryogenesis from callus of mature seed explants of bread wheat (Triticum aestivum L.). Pakistan J Bot 36:629–634
Mallu TS, Mutinda S, Githiri SM et al (2021) New pre-attachment Striga resistant sorghum adapted to african agro‐ecologies. Pest Manag Sci 77:2894–2902
Mirza K, Aasim M, Katırcı R et al (2022) Machine learning and artificial neural networks-based approach to model and optimize ethyl methanesulfonate and sodium azide induced in vitro regeneration and morphogenic traits of water hyssops (Bacopa monnieri L). J Plant Growth Regul. https://doi.org/10.1007/s00344-022-10808-w
Mishra A, Khurana P (2003) Genotype dependent somatic embryogenesis and regeneration from leaf base cultures of Sorghum bicolor. J Plant Biochem Biotechnol 12:53–56
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Myo EM, Ge B, Ma J et al (2019) Indole-3-acetic acid production by Streptomyces fradiae NKZ-259 and its formulation to enhance plant growth. BMC Microbiol 19:1–14
Niazian M, Niedbała G (2020) Machine learning for plant breeding and biotechnology. Agriculture 10:436
Niazian M, Noori SAS, Galuszka P et al (2017) Genetic stability of regenerated plants via indirect somatic embryogenesis and indirect shoot regeneration of Carum copticum L. Ind Crops Prod 97:330–337
Niazian M, Sadat-Noori SA, Abdipour M et al (2018) Image processing and artificial neural network-based models to measure and predict physical properties of embryogenic callus and number of somatic embryos in ajowan (Trachyspermum ammi (L.) Sprague). Vitr Cell Dev Biol 54:54–68
Niazian M, Shariatpanahi ME (2020) In vitro-based doubled haploid production: recent improvements. Euphytica 216:1–21
Omer RA, Suliman S, Beshir MM (2021) Regeneration of sorghum through tissue culture techniques. Int J Genet Eng 9:16–20
Pacher M, Puchta H (2017) From classical mutagenesis to nuclease-based breeding–directing natural DNA repair for a natural end‐product. Plant J 90:819–833
Paterson AH, Bowers JE, Bruggmann R et al (2009) The Sorghum bicolor genome and the diversification of grasses. Nature 457:551–556
Pavlov YL (2019) Random forests. In: Text mining with machine learning: Principles and Techniques (Eds) Žižka J, Dařena F, Svoboda A. Crc Press. https://doi.org/10.1201/9780429469275-8
Pedregosa F, Varoquaux G, Gramfort A et al (2011) Scikit-learn: machine learning in {P}ython. J Mach Learn Res 12:2825–2830
Pennells J, Cruickshank A, Chaléat C et al (2021) Sorghum as a novel biomass for the sustainable production of cellulose nanofibers. Ind Crops Prod 171:113917
Pola S, Saradamani N, Ramana T (2007) Enhanced shoot regeneration in tissue culture studies of Sorghum bicolor. J Agric Technol 3:275–286
Qureshi MA, Shafqat W, ud Din S, et al (2021) Effect of different plant growth regulators on in vitro regeneration of mango: a report. J Glob Innov Agric Sci 9:95–98
Ramazan K, Esma S, Belkıs U (2015) Statistical optimisation of organic additives for maximum brightness and brightener analysis in a nickel electroplating bath. Trans IMF 93:89–96
Riaz A, Kanwal F, Börner A et al (2021) Advances in genomics-based breeding of barley: molecular tools and genomic databases. Agronomy 11:894
Sai NK, Visarada K, Lakshmi YA et al (2006) In vitro culture methods in sorghum with shoot tip as the explant material. Plant Cell Rep 25:174–182
Salehi M, Farhadi S, Moieni A et al (2020) Mathematical modeling of growth and paclitaxel biosynthesis in corylus avellana cell culture responding to fungal elicitors using multilayer perceptron-genetic algorithm. Front Plant Sci 11:1–12. https://doi.org/10.3389/fpls.2020.01148
Salehi M, Farhadi S, Moieni A et al (2021) A hybrid model based on general regression neural network and fruit fly optimization algorithm for forecasting and optimizing paclitaxel biosynthesis in Corylus avellana cell culture. Plant Methods 17:1–13
Sattar MN, Iqbal Z, Al-Khayri JM, Jain SM (2021) Induced genetic variations in fruit trees using new breeding tools: food security and climate resilience. Plants 10:1347
Stamenković OS, Siliveru K, Veljković VB et al (2020) Production of biofuels from sorghum. Renew Sustain Energy Rev 124:109769
Sudhakararao P (2011) Leaf discs as a source material for plant tissue culture studies of Sorghum bicolor (L.) Moench. Not Sci Biol 3:70–78
Van Rossum G, Drake FL (2009) Python 3 reference manual. CreateSpace, Scotts Valley, CA
Visarada K, Sai Kishore N, Balakrishna D, Rao SV (2003) Transient GUS expression studies in sorghum to develop a simple protocol for Agrobacterium mediated genetic transformation [Sorghum bicolor (L.) Moench]. J Genet Breed 57:147–154
Webb GI, Sammut C, Perlich C et al (2011) Leave-one-out cross-validation. Encyclopedia of machine learning. Springer US, Boston, MA, pp 600–601
Weiss T, Zhao J, Hu R et al (2022) Production of distilled spirits using grain sorghum through liquid fermentation. J Agric Food Res 9:100314
Zapata C, Srivatanakul M, Park S-H et al (1999) Improvements in shoot apex regeneration of two fiber crops: cotton and kenaf. Plant Cell Tissue Organ Cult 56:185–191
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
MA: Conceptualization, Methodology; Formal analysis; Writing - Original Draft, Supervision, Project administration, SAA: Formal analysis, Writing - Review & Editing, Visualization, AA: Investigation, Data Curation, MTA: Investigation, Data Curation, MAN: Original Draft Writing - Review & Editing, FSB: Material, Article control, Review & Editing.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest. All authors read the manuscript and showed their willingness to publish the manuscript.
Additional information
Communicated by Alison M.R. Ferrie.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aasim, M., Ali, S.A., Altaf, M.T. et al. Artificial neural network and decision tree facilitated prediction and validation of cytokinin-auxin induced in vitro organogenesis of sorghum (Sorghum bicolor L.). Plant Cell Tiss Organ Cult 153, 611–624 (2023). https://doi.org/10.1007/s11240-023-02498-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02498-3