Abstract
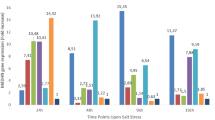
Salinity and drought are important abiotic stress factors whose frequency and severity have increased due to climate change. Today, cultivating plants with increased stress resistance is a global priority. Herein, the morphological and physio-biochemical responses of cactus pear adventitious shoots to different levels of salt and drought stresses were investigated. Sodium chloride (NaCl; 2.5–10 g·L−1) and polyethylene glycol (PEG; 50–150 g·L−1) were added to the culture medium at different growth stages (shoot bud proliferation, and shoot elongation and rooting) and over different periods (1–3 weeks). Salt and drought stresses reduced shoot proliferation, growth, fresh and dry weights. Increasing the concentration of NaCl to 10 g.L−1 and the duration of stress to 3 weeks significantly decreased the survival rate of organogenic cultures (75%) and isolated shoots (62.25%). Drought stress did not significantly affect the survival rate of explants (95.75–100%). Notably, drought stress promoted rhizogenesis of organogenic cultures (31.25–62.50%; no root formation in control group) and isolated shoots (95.75–100%; control group, 91.63%). As regards to the physio-biochemical properties of explants, significant variations were observed. After one week of stress, there was a notable decline in the levels of glycine betaine, proline and carbohydrates. However, increasing the duration of stress promoted their accumulation, which can be taken as an indicator of adaptation to stress. The total protein and chlorophyll a contents decreased under stress conditions, while chlorophyll b showed random variations. The regenerated plants were successfully established in the greenhouse and then transferred to soil where they produced new cladodes.
Key message
In vitro propagation of cactus pear (Opuntia ficus indica) under abiotic stress conditions allows for large-scale production of plants with enhanced tolerance to salt and drought stresses.


Similar content being viewed by others
Data availability
Data available upon reasonable request to the corresponding author.
References
Abbaspour J, Ehsanpour AA (2020) Sequential expression of key genes in proline, glycine betaine and artemisinin biosynthesis of Artemisia aucheri Boiss using salicylic acid under in vitro osmotic stress. Biologia 75:1251–1263. https://doi.org/10.2478/s11756-020-00507-w
Achour A, Bidai Y, Belkhodja K (2015) The impact of salinity on water and metabolic behavior of a variety of Okra (Abelmoschus esculentus L.). Int J Innov Appl Studies 12:943–953
Afzal S, Chaudhary N, Singh NK (2021) Role of soluble sugars in metabolism and sensing under abiotic stress. In: Aftab T, Hakeem KR (eds) Plant growth regulators. Springer, Cham, pp 305–334. https://doi.org/10.1007/978-3-030-61153-8_14
Ahmad W, Ayyub CM, Shehzad MA, Ziaf K, Ijaz M, Sher A, Abbas T, Shafi J (2019) Supplemental potassium mediates antioxidant metabolism, physiological processes, and osmoregulation to confer salt stress tolerance in cabbage (Brassica oleracea L.). Hortic Environ Biotechnol 60:853–869. https://doi.org/10.1007/s13580-019-00172-2
Ahmad Z, Anjum S, Skalicky M, Waraich EA, Tariq RMS, Ayub MA, Hossain A, Hassan MM, Brestic M, Islam MS, Habib-Ur-Rahman M, Wasaya A, Iqbal MA, El Sabagh A (2021) Selenium alleviates the adverse effect of drought in oilseed crops camelina (Camelina sativa L.) and Canola (Brassica napus L.). Molecules 26:1699. https://doi.org/10.3390/molecules26061699
Amghar I, Diria G, Boumlik I, Gaboun F, Iraqi D, Labhilili M, Mentag R, Meziani R, Mazri MA, Ibriz M, Abdelwahd R (2021a) An efficient regeneration pathway through adventitious organogenesis for the endangered Argania spinosa (L.) Skeels. Vegetos 34:355–367. https://doi.org/10.1007/s42535-021-00208-y
Amghar I, Ibriz M, Ibrahimi M, Boudra A, Gaboun F, Meziani R, Iraqi D, Mazri MA, Diria G, Abdelwahd R (2021b) In vitro root induction from argan (Argania spinosa (L.) Skeels) adventitious shoots: influence of ammonium nitrate, auxins, silver nitrate and putrescine, and evaluation of plantlet acclimatization. Plants 10:1062. https://doi.org/10.3390/plants10061062
Ami K, Planchais S, Cabassa C, Guivarc’h A, Very A-A, Khelifi M, Djebbar R, Abrous-Belbachir O, Carol P (2020) Different proline responses of two algerian durum wheat cultivars to in vitro salt stress. Acta Physiol Plant 42:21. https://doi.org/10.1007/s11738-019-3004-9
Annunziata MG, Ciarmiello LF, Woodrow P, Maximova E, Fuggi A, Carillo P (2017) Durum wheat roots adapt to salinity remodeling the cellular content of nitrogen metabolites and sucrose. Front Plant Sci 7:2035. https://doi.org/10.3389/fpls.2016.02035
Azad N, Rezayian M, Hassanpour H, Niknam V, Ebrahimzadeh H (2021) Physiological mechanism of salicylic acid in Mentha pulegium L. under salinity and drought stress. Braz J Bot 44:359–369. https://doi.org/10.1007/s40415-021-00706-y
Bartels D, Sunkar R (2005) Drought and salt tolerance in plants. Crit Rev Plant Sci 24:23–58. https://doi.org/10.1080/07352680590910410
Belfakih M, Ibriz M, Zouahri A (2013) Effet de la salinité sur les paramètres morphophysiologiques de deux variétés de bananier (Musa acuminata L). J App Biosci 70:5652–5662
Bouchiha F, Mazri MA (2022) Micropropagation of cactus pear (Opuntia ficus indica) by organogenesis. Afr Med Agri J Al Awamia 135:1–15. https://doi.org/10.34874/IMIST.PRSM/afrimed-i135.33263
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chen TH, Murata N (2011) Glycinebetaine protects plants against abiotic stress: mechanisms and biotechnological applications. Plant Cell Environ 34:1–20. https://doi.org/10.1111/j.1365-3040.2010.02232.x
Desoky ESM, Ibrahim SA, Merwad ARM (2019) Mitigation of salinity stress effects on growth, physio-chemical parameters and yield of snapbean (Phaseolus vulgaris L.) by exogenous application of glycine betaine. Inter Letter Natural Sci 76:60–71. https://doi.org/10.18052/www.scipress.com/ILNS.76.60
Dobrá J, Vanková R, Havlová M, Burman AJ, Libus J, Štorchová H (2011) Tobacco leaves and roots differ in the expression of proline metabolism-related genes in the course of drought stress and subsequent recovery. J Plant Physiol 168:1588–1597. https://doi.org/10.1016/j.jplph.2011.02.009
Dogan M (2020) Effect of salt stress on in vitro organogenesis from nodal explant of Limnophila aromatica (Lamk.) Merr. and Bacopa monnieri (L.) Wettst. and their physio-morphological and biochemical responses. Physiol Mol Biol Plants 26:803–816. https://doi.org/10.1007/s12298-020-00798-y
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
El Kharrassi Y, Mazri MA, Mabrouk A, Nasser B, El Mzouri EH (2015) Flowering and fruiting phenology, and physico-chemical characteristics of 2-year-old plants of six species of Opuntia from eight regions of Morocco. J Hort Sci Biotechnol 90:682–688. https://doi.org/10.1080/14620316.2015.11668731
El Kharrassi Y, Mazri MA, Benyahia H, Benaouda H, Nasser B, El Mzouri EH (2016) Fruit and juice characteristics of 30 accessions of two cactus pear species (Opuntia ficus indica and Opuntia megacantha) from different regions of Morocco. LWT Food Sci Technol 65:610–617. https://doi.org/10.1016/j.lwt.2015.08.044
El Kharrassi Y, Maata N, Mazri MA, El Kamouni S, Talbi M, El Kebbaj R, Moustaid K, Essamadi AK, Andreoletti P, El Mzouri EH, Cherkaoui-Malki M, Nasser B (2018) Chemical and phytochemical characterizations of argan oil (Argania spinosa L. skeels), olive oil (Olea europaea L. cv. Moroccan picholine), cactus pear (Opuntia megacantha salm-dyck) seed oil and cactus cladode essential oil. J Food Meas Char 12:747–754. https://doi.org/10.1007/s11694-017-9688-x
Fatma F, Kamal A, Srivastava A (2018) Exogenous application of salicylic acid mitigates the toxic effect of pesticides in Vigna radiata (L.) Wilczek. J Plant Growth Regul 37:1185–1194. https://doi.org/10.1007/s00344-018-9819-6
Fu J, Sun P, Luo Y, Zhou H, Gao J, Zhao D, Pubu Z, Liu J, Hu T (2019) Brassinosteroids enhance cold tolerance in Elymus nutans via mediating redox homeostasis and proline biosynthesis. Environ Exp Bot 167:103831. https://doi.org/10.1016/j.envexpbot.2019.103831
Garcia-Saucedo PA, Valdez-Morales M, Valverde ME, Cruz-Hernández A, Paredes-López O (2005) Plant regeneration of three Opuntia genotypes used as human food. Plant Cell Tiss Organ Cult 80:215–219
Ghaffari A, Hasanloo T, Nekouei MK (2013) Micropropagation of tuna (Opuntia ficus–indica) and effect of medium composition on proliferation and rooting. Int J Biosci 3:129–139
Ghassemi-Golezani K, Farhadi N, Nikpour-Rashidabad N (2018) Responses of in vitro-cultured Allium hirtifolium to exogenous sodium nitroprusside under PEG-imposed drought stress. Plant Cell Tissue Organ Cult 133:237–248. https://doi.org/10.1007/s11240-017-1377-2
Ghomari S, Haddad M, Lotmani B, Labdi M, Benjeda M, Bennabi F, Hachem K (2015) Optimization of somatic organogenesis in vitro of Solanum tuberosum in salt stress condition. Adv Environ Biol 9:122–127
Goel R, Suyal DC, Kumar V, Jain L, Soni R (2018) Stress-tolerant beneficial microbes for sustainable agricultural production. In: Panpatte D, Jhala Y, Shelat H, Vyas R (eds) Microorganisms for Green Revolution. Springer, Singapore, pp 141–159. https://doi.org/10.1007/978-981-10-7146-1_8
Gogoi B, Nath T, Kashyap D, Sarma S, Kalita R (2020) Sustainable agriculture, forestry and fishery for bioeconomy. In: Kataki R, Pandey A, Khanal SK, Pant D (eds) Current developments in Biotechnology and Bioengineering. Elsevier, Amsterdam, pp 349–371. https://doi.org/10.1016/B978-0-444-64309-4.00015-5
Grieve CM, Grattan SR (1983) Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 70:303–307. https://doi.org/10.1007/bf02374789
Hai Y, Huang AM, Tang Y (2019) Structure-guided function discovery of an NRPS-like glycine betaine reductase for choline biosynthesis in fungi. Proc Natl Acad Sci 116:10348–10353. https://doi.org/10.1073/pnas.1903282116
Hanana M, Hamrouni L, Cagnac O, Blumwald E (2011) Mécanismes et stratégies cellulaires de tolérance à la salinité (NaCl) chez les plantes. Environ Rev 19:121–140. https://doi.org/10.1139/a11-003
Handa N, Kohli SK, Kaur R, Sharma A, Kumar V, Thukral AK, Arora S, Bhardwaj R (2018) Role of compatible solutes in enhancing antioxidative defense in plants exposed to metal toxicity. In: Hasanuzzaman M, Nahar K, Fujita M (eds) Plants under metal and metalloid stress. Springer, Singapore, pp 207–228. https://doi.org/10.1007/978-981-13-2242-6_7
Hernández-Pérez CA, Gómez-Merino FC, Spinoso-Castillo JL, Bello-Bello JJ (2021) In vitro screening of sugarcane cultivars (Saccharum spp. Hybrids) for tolerance to polyethylene glycol-induced water stress. Agronomy 11:598. https://doi.org/10.3390/agronomy11030598
Honorato-Salazar JA, Aburto J, Amezcua-Allieri MA (2021) Agave and Opuntia species as sustainable feedstocks for bioenergy and byproducts. Sustainability 13:12263. https://doi.org/10.3390/su132112263
Hussain MI, Al-Dakheel AJ (2018) Effect of salinity stress on phenotypic plasticity, yield stability, and signature of stable isotopes of carbon and nitrogen in safflower. Environ Sci Pollut Res 25:23685–23694. https://doi.org/10.1007/s11356-018-2442-z
Inglese P, Liguori G, de la Barrera E (2017) Ecophysiology and reproductive biology of cultivated cacti. In: Inglese P, Mondragon C, Nefzaoui A, Sáenz C (eds) Crop Ecology, Cultivation and uses of Cactus Pear. FAO-ICARDA, Rome, pp 29–41
Kadkhodaei H, Sodaeizadeh H, Arany AM, Zadeh MAH (2016) The role of glycine betain in increasing drought resistance of Sorghum halopens under field condition. Env Stresses Crop Sci. https://doi.org/10.22077/ESCS.2016.360
Kaur G, Asthir B (2019) Water and salt stress metabolomics for wheat genotypes of India. Cereal Res Commun 47:615–625. https://doi.org/10.1556/0806.47.2019.36
Ke W, Yin Y, Chen X, Qiu B (2021) Chlorophylls. In: Gao K, Hutchins DA, Beardall J (eds) Research methods of environmental physiology in aquatic Sciences. Springer, Singapore, pp 95–106. https://doi.org/10.1007/978-981-15-5354-7_11
Koenigshofer H, Loeppert HG (2019) The up-regulation of proline synthesis in the meristematic tissues of wheat seedlings upon short-term exposure to osmotic stress. J Plant Physiol 237:21–29. https://doi.org/10.1016/j.jplph.2019.03.010
Kosová K, Vítámvás P, Urban MO, Prášil IT, Renaut J (2018) Plant abiotic stress proteomics: the major factors determining alterations in cellular proteome. Front Plant Sci 9:122. https://doi.org/10.3389/fpls.2018.00122
Kozminska A, Al Hassan M, Hanus-Fajerska E, Naranjo MA, Boscaiu M, Vicente O (2018) Comparative analysis of water deficit and salt tolerance mechanisms in Silene. South Afr J Bot 117:193–206. https://doi.org/10.1016/j.sajb.2018.05.022
Kumar A, Nagar S, Anand S (2021) Climate change and existential threats. In: Singh S, Singh P, Rangabhashiyam S, Srivastava KK (eds) Global climate change. Elsevier, Amsterdam, pp 1–31. https://doi.org/10.1016/B978-0-12-822928-6.00005-8
La VH, Lee BR, Islam MT, Park SH, Lee H, Bae DW, Kim TH (2019) Antagonistic shifting from abscisic acid- to salicylic acid-mediated sucrose accumulation contributes to drought tolerance in Brassica napus. Environ Exp Bot 162:38–47. https://doi.org/10.1016/j.envexpbot.2019.02.001
Lallouche B, Boutekrabt A, Hadjkouider B (2015) Effect of salt stress on accumulation of proline and soluble sugars in cladodes and roots of two Opuntia species existing in algerian steppe. Int J Biosci 6:43–51
Li SX, Guo J, Wang T, Gong L, Liu F, Brestic M, Liu S, Song F, Li X (2021) Melatonin reduces nanoplastic uptake, translocation, and toxicity in wheat. J Pineal Res 71:e12761. https://doi.org/10.1111/jpi.12761
Lilius G, Holmberg N, Bülow L (1996) Enhanced NaCl stress tolerance in transgenic tobacco expressing bacterial choline dehydrogenase. Nat Biotechnol 14:177–180. https://doi.org/10.1038/nbt0296-177
Liu Y, He Z, Xie Y, Su L, Zhang R, Wang H, Li C, Long S (2021) Drought resistance mechanisms of Phedimus aizoon L. Sci Rep 11:13600. https://doi.org/10.1038/s41598-021-93118-7
Mackinney G (1941) Absorption of light by chlorophyll solutions. J Biol Chem 140:315–322. https://doi.org/10.1016/S0021-9258(18)51320-X
Mazri MA (2018) Cactus Pear (Opuntia spp.) breeding. In: Al-Khayri JM, Jain SM, Johnson DV (eds) Advances in plant breeding strategies: fruits. Springer, Cham, pp 307–341. https://doi.org/10.1007/978-3-319-91944-7_8
Mazri MA (2021) Cactus pear (Opuntia spp.) species and cultivars. In: Ramadan MF, Ayoub TEM, Rohn S (eds) Opuntia spp.: chemistry, bioactivity and industrial applications. Springer, Cham, pp 83–107. https://doi.org/10.1007/978-3-030-78444-7_4
Mazri MA, Meziani R, Elmaataoui S, Alfeddy N, Jaiti F (2019) Assessment of genetic fidelity, biochemical and physiological characteristics of in vitro grown date palm cv. Al-Fayda Vegetos 32:333–344. https://doi.org/10.1007/s42535-019-00034-3
Mazri MA, Meziani R, El Bakouri Z (2021) Cost analysis of date palm (cv. Mejhoul) plantlets produced by organogenesis in Morocco. Plant Cell Tiss Organ Cult 146:409–415. https://doi.org/10.1007/s11240-021-02066-7
Mengesha B, Mekbib F, Abraha E (2016) In vitro screening of cactus [Opuntia ficus-indicia (L.) Mill] genotypes for drought tolerance. Am J Plant Sci 7:1741–1758. https://doi.org/10.4236/ajps.2016.713163
Miri M, Ghooshchi F, Tohidi-Moghadam HR, Larijani HR, Kasraie P (2021) Ameliorative effects of foliar spray of glycine betaine and gibberellic acid on cowpea (Vigna unguiculata L. Walp.) Yield affected by drought stress. Arab J Geosci 14:830. https://doi.org/10.1007/S12517-021-07228-7
Mohamed-Yasseen Y, Barringer SA, Splittstoesser WE, Schnell RJ (1995) Rapid propagation of tuna (Opuntia ficus-indica) and plant establishment in soil. Plant Cell Tiss Organ Cult 42:117–119
Murashige T, Skoog FA (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Phys Planta 15:473–479. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Naliwajski M, Skłodowska M (2021) The relationship between the antioxidant system and proline metabolism in the leaves of cucumber plants acclimated to salt stress. Cells 10:609. https://doi.org/10.3390/cells10030609
Nefzaoui A, Louhaichi M, Ben Salem H (2014) Cactus as a tool to mitigate drought and to combat desertification. J Arid Land Studies 24:121–124
Ozturk M, Unal BT, García-Caparrós P, Khursheed A, Gul A, Hasanuzzaman M (2020) Osmoregulation and its actions during the drought stress in plants. Physiol Plant 172:1321–1335. https://doi.org/10.1111/ppl.13297
Palevitch D, Earon G, Levin I (1993) Treatment of benign prostatic hypertrophy with Opuntia ficus-indica (L.) Miller. J Herbs Spices Med Plants 2:45–49. https://doi.org/10.1300/J044v02n01_06
Paquin R, Lechasseur P (1979) Observations sur une méthode de dosage de la proline libre dans les extraits de plantes. Can J Bot 57:1851–1854. https://doi.org/10.1139/b79-233
Paz-Navarro A, Ojeda-Linares CI, Alvarez-Rios GD, Vallejo M, Casas A (2022) Traditional management and diversity of Opuntia: general panorama in Mexico and a case study in the meridional central plateau. In: Casas A, Blancas Vázquez JJ (eds) Ethnobotany of the Mountain Regions of Mexico. Ethnobotany of Mountain Regions. Springer, Cham, pp 1–32. https://doi.org/10.1007/978-3-319-77089-5_21-1
Pinedo-Espinoza JM, Aguirre-Mancilla CL, Jiménez-Alvarado R, Raya-Pérez JC, Iturriaga G, Ramírez-Pimentel JG, Hernández-Fuentes AD (2017) Bioactive compounds and antioxidant activity evolution during the ripening process of 12 Opuntia spp. fruit accessions. Emir J Food Agric 29:138–148. https://doi.org/10.9755/ejfa.2016-09-1324
Potgieter J, D’Aquino S (2017) Fruit production and post-harvest management. In: Inglese P, Mondragon C, Nefzaoui A (eds) Crop ecology, cultivation and uses of cactus pear. Food and Agriculture Organization of the United Nations and the International Center for Agricultural Research in the Dry Areas, Rome, pp 51–71
Rane J, Singh AK, Kumar M, Boraiah KM, Meena KK, Pradhan A, Prasad PVV (2021) The adaptation and tolerance of major cereals and legumes to important abiotic stresses. Int J Mol Sci 22:12970. https://doi.org/10.3390/ijms222312970
Regalado-Rentería E, Aguirre-Rivera JR, González-Chávez MM, Sánchez-Sánchez R, Martínez-Gutiérrez F, Juárez-Flores BI (2020) Assessment of extraction methods and biological value of seed oil from eight variants of prickly pear fruit (Opuntia spp.). Waste Biomass Valor 11:1181–1189. https://doi.org/10.1007/s12649-018-0409-4
Reyes-Agüero JA, Aguirre RJR, Valiente-Banuet A (2006) Reproductive biology of Opuntia: a review. J Arid Environ 64:549–585. https://doi.org/10.1016/j.jaridenv.2005.06.018
Sáenz C (2017) Processing and utilization of fruit, cladodes and seeds. In: Inglese P, Mondragon C, Nefzaoui A (eds) Crop ecology, cultivation and uses of cactus pear. Food and Agriculture Organization of the United Nations and the International Center for Agricultural Research in the Dry Areas, Rome, pp 135–149
Santos-Díaz MdS, Camarena-Rangel NG (2019) Cacti for production of metabolites: current state and perspectives. Appl Microbiol Biotechnol 103:8657–8667. https://doi.org/10.1007/s00253-019-10125-5
Seleiman MF, Aslam MT, Alhammad BA, Hassan MU, Maqbool R, Chattha MU, Khan I, Gitari HI, Uslu OS, Roy R, Battaglia ML (2022) Salinity stress in wheat: effects, mechanisms and management strategies. Phyton-Int J Exp Bot 91:667–694. https://doi.org/10.32604/phyton.2022.017365
Shedbalkar UU, Adki VS, Jadhav JP, Bapat VA (2010) Opuntia and other cacti: applications and biotechnological insights. Trop Plant Biol 3:136–150. https://doi.org/10.1007/s12042-010-9055-0
Shetty P, Gitau MM, Maróti G (2019) Salinity stress responses and adaptation mechanisms in eukaryotic green microalgae. Cells 8:1657. https://doi.org/10.3390/cells8121657
Shu J, Ma X, Ma H, Huang Q, Zhang Y, Guan M, Guan C (2022) Transcriptomic, proteomic, metabolomic, and functional genomic approaches of Brassica napus L. during salt stress. PLoS ONE 17:e0262587. https://doi.org/10.1371/journal.pone.0262587
Singh D, Kumar A (2021) In vitro screening and characterization of selected elite clones of Eucalyptus tereticornis Sm. For salt stress. J Plant Growth Regul 40:694–706. https://doi.org/10.1007/s00344-020-10138-9
Singh R, Machanuru R, Singh B, Shrivastava M (2021) Climate-resilient agriculture: enhance resilience toward climate change. In: Singh S, Singh P, Rangabhashiyam S, Srivastava KK (eds) Global climate change. Elsevier, Amsterdam, pp 45–61. https://doi.org/10.1016/B978-0-12-822928-6.00016-2
Sobahan MA, Akter N, Rana MM (2022) Polyethylene glycol mediated drought stress impacts on germination, growth and accumulation of proline in rice (Oryza sativa L.). SAARC J Agric 20:107–119. https://doi.org/10.3329/sja.v20i1.60544
Wedeking R, Maucourt M, Deborde C, Moing A, Gibon Y, Goldbach HE, Wimmer MA (2018) ) 1H-NMR metabolomic profiling reveals a distinct metabolic recovery response in shoots and roots of temporarily drought-stressed sugar beets. PLoS ONE 13:e0196102. https://doi.org/10.1371/journal.pone.0196102
Wen W, Timmermans J, Chen Q, Van Bodegom PM (2021) A review of remote sensing challenges for food security with respect to salinity and drought threats. Remote Sens 13:6. https://doi.org/10.3390/rs13010006
Wu B, Ma Z, Yan N (2020) Agricultural drought mitigating indices derived from the changes in drought characteristics. Remote Sens Environ 244:111813. https://doi.org/10.1016/j.rse.2020.111813
Yang A, Dai X, Zhang WH (2012) A R2R3-type MYB gene, OsMYB2, is involved in salt, cold, and dehydration tolerance in rice. J Exp Bot 63:2541–2556. https://doi.org/10.1093/jxb/err431
Yoo SJ, Weon HY, Song J, Sang MK (2019) Induced tolerance to salinity stress by halotolerant bacteria bacillus aryabhattai H19-1 and B. mesonae H20-5 in tomato plants. J Microbiol Biotechnol 29:1124–1136. https://doi.org/10.4014/jmb.1904.04026
Zoghlami N, Bouamama B, Khammassi M, Ghorbel A (2012) Genetic stability of long-term micropropagated Opuntia ficus-indica (L.) Mill. Plantlets as assessed by molecular tools: perspectives for in vitro conservation. Ind Crops Prod 36:59–64
Zulfiqar F, Ashraf M, Siddique KHM (2022) Role of glycine betaine in the thermotolerance of plants. Agronomy 12:276. https://doi.org/10.3390/agronomy12020276
Funding
This research did not receive any specific grant from external funding agencies.
Author information
Authors and Affiliations
Contributions
MAM and SA, study design, conception and supervision. HR, FB, SEM, EO and MAM, plant material collection, preparation, in vitro culture and morphological analyses. HR, FB and SA, salt stress experiments and biochemical analyses. HR, FB and MNA, drought stress experiments and biochemical analyses. IR and HR, plantlet acclimatization, transplantation and ex vitro evaluation. HR and FB, data collection. MAM, statistical analysis. HR and MAM, manuscript writing. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Additional information
Communicated by Christell van der Vyver.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Radi, H., Bouchiha, F., El Maataoui, S. et al. Morphological and physio-biochemical responses of cactus pear (Opuntia ficus indica (L.) Mill.) organogenic cultures to salt and drought stresses induced in vitro. Plant Cell Tiss Organ Cult 154, 337–350 (2023). https://doi.org/10.1007/s11240-023-02454-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02454-1