Abstract
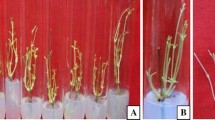
Santalum album L. (Indian sandalwood) is an economically important but vulnerable tropical tree species. Cultures were established via direct shoot regeneration from axillary buds on Murashige and Skoog (MS) medium supplemented with 2.5 mg L−1 6-benzylaminopurine (BAP). The shoots were multiplied using MS medium containing 1.0 mg L−1 BAP and 0.5 mg L−1 indole-3 acetic acid and rooted on half strength MS medium containing 1.0 mg L−1 indole-3 butyric acid. The rooted plantlets were hardened and acclimatized in greenhouse using soilrite® and cocopeat (1:1) mixture. The concentrations of photosynthetic pigments were analyzed and detected less under in vitro conditions (6.05 μg g−1 FW) as compared to the 4 weeks old hardened (6.91 μg g−1 FW) and 12 weeks old acclimatized plantlets (7.8 μg g−1 FW) under greenhouse (ex vitro) environment. The anatomical evaluation of plantlets at subsequent stages of propagation suggested that the in vitro raised plantlets possessed structural abnormalities such as underdeveloped cuticle, unorganized tissue systems, reduced mesophyll tissues, fewer vascular elements and mechanical tissues, and loosely arranged thin walled paranchymatous ground tissues, which were slowly repaired during ex vitro hardening and acclimatization process to validate the developmental adaptation of micropropagated plantlets for maximum survival in the field (98.0% survival rate). The findings could help in the optimization of high-frequency commercial micropropagation of S. album for year-round production, and supply of this economically prominent vulnerable plant species to the farmers and the industries that rely on it.
Key Message
The anatomical and physiological features at subsequent stages in micropropagation of Santalum album elucidated the in vitro anomalies and ex vitro repair mechanism for improved survival in field.





Similar content being viewed by others
References
Agrawal AA (2001) Phenotypic plasticity in the interactions and evolution of species. Science 294:321–326. https://doi.org/10.1126/science.1060701
Arunkumar AN, Joshi G, Mohan Ram HY (2012) Sandalwood: history, uses, present status and the future. Curr Sci 103:1408–1416
Arunkumar AN, Dhyani A, Joshi G (2019) Santalum album. IUCN Red List Threatened Species. https://doi.org/10.2305/IUCN.UK.2019-1.RLTS.T31852A2807668.en
Athanasiou K, Dyson BC, Websterm RE, Johnson GN (2010) Dynamic acclimation of photosynthesis increases plant fitness in changing environments. Plant Physiol 152:366–373. https://doi.org/10.1104/pp.109.149351
Baldovini N, Delasalle C, Joulain D (2011) Phytochemistry of the heartwood from fragrant Santalum species: a review. Flavour Fragr J 26:7–26. https://doi.org/10.1002/ffj.2025
Barros J, Serk H, Granlund I, Pesquet E (2015) The cell biology of lignifications in higher plants. Ann Bot 115(7):1053–1074. https://doi.org/10.1093/aob/mcv046
Bhargava P, Ravindra N, Singh G (2018) A modified and improved protocol development for in vitro clonal propagation of Santalum album L. from internodal explants. Trop Plant Res 5:193–199. https://doi.org/10.22271/tpr.2018.v5.i2.024
Bhattacharyya P, Kumaria S, Job N, Tandon P (2016) En-masse production of elite clones of Dendrobium crepidatum: a threatened, medicinal orchid used in traditional Chinese medicine (TCM). J Appl Res Med Aromat Plants 3:168–176. https://doi.org/10.1016/j.jarmap.2016.04.001
Bidabadi SS, Jain MS (2020) Cellular, molecular and physiological aspects of in vitro plant regeneration. Plants 9:702. https://doi.org/10.3390/plants9060702
Burdock GA, Carabin IG (2008) Safety assessment of sandalwood oil (Santalum album L.). Food Chem Toxicol 46:421–432. https://doi.org/10.1016/j.fct.2007.09.092
Chen L, Wang RZ (2009) Anatomical and physiological divergences and compensatory effects in two Leymus chinensis (Poaceae) ecotypes in Northeast China. Agric Ecosyst Environ 134:46–52. https://doi.org/10.1016/j.agee.2009.05.015
Deb CR, Imchen T (2010) An efficient in vitro hardening technique of tissue culture raised plants. Biotechnology 9:79–83
Dobranszki J, Mendler-Drienyovszki N (2014) Cytokinin-induced changes in the chlorophyll content and fuorescence of in vitro apple leaves. J Plant Physiol 171:1472–1478. https://doi.org/10.1016/j.jplph.2014.06.015
Dutt S, Verma KS (2005) Effect of collection of time, pre-sowing treatments and sowing time on the germinability of sandal (Santalum album L.) seeds under nursery conditions. J Non-Timber Prod 12:205–208
Gairola S, Aggarwal PS, Ravikumar GS (2008) Status of production and marketing of sandalwood (Santalum album L.). In: Gairola et al. (eds) Proceedings of national seminar on conservation, improvement, cultivation and management of sandal. Institute of Wood Science and Technology, Bangalore. pp 1–8
Gentile A, Frattarelli A, Nota P, Condello E, Caboni E (2017) The aromatic cytokinin meta-topolin promotes in vitro propagation, shoot quality and micrografting in Corylus colurna L. Plant Cell Tiss Org Cult 128:693–703
Harbaugh DT, Baldwin BG (2007) Phylogeny and biogeography of the sandalwoods (Santalum, Santalaceae): repeated dispersals throughout the Pacific. Am J Bot 94:1028–1040. https://doi.org/10.3732/ajb.94.6.1028
Hatfield JL, Boote KJ, Kimball BA, Ziska LH, Izaurralde RC, Ort D, Thomson AM, Wolfe DW (2011) Climate impacts on agriculture: implications for crop production. Agron J 103:351–370
Hazarika BN (2003) Acclimatization of tissue-cultured plants. Curr Sci 85:1704–1712
Hirai G, Okumura T, Takeuchi S, Tanaka O, Chujo H (2000) Studies on the effect of the relative humidity of the atmosphere on the growth and physiology of rice plants. Plant Prod Sci 3:129–133
Hronkova M, Zahradnickova H, Simkova M, Simek P, Heydova A (2003) The role of abscisic acid in acclimation of plants cultivated in vitro to ex vitro conditions. Biol Plant 46:535–541
IUCN (2012) Asian Regional Workshop (Conservation & Sustainable Management of Trees, Viet Nam, August 1996) Santalum album. In: IUCN 2012. IUCN red list of threatened species. Version 2012.2. www.iucnredlist.org. Accessed 18 Feb 2021
Jeon MW, Ali MB, Hahn EJ, Paek KY (2006) Photosynthetic pigments, morphology and leaf gas exchange during ex vitro acclimatization of micropropagated CAM Doritaenopsis plantlets under relative humidity and air temperature. Environ Exp Bot 55:183–194
Khande BP, Burondkar MM, Sawardekar SV, Chavan SS, Gokhale NB, Bhave SG, Shinde AK, Devmore JP (2017) Effect of media combination on in vitro callus induction in sandalwood (Santalum album L.). Adv Agri Res Technol J 1:98–105
Kozai T (2010) Photoautotrophic micropropagation-environmental control for promoting photosynthesis. Propag Ornam Plants 10:188–204
Krishnakumar N, Parthiban KT (2018) Micropropagation (in vitro) techniques for sandal wood (Santalum album L.). J Pharmacog Phytochem 7:620–627
Kulus D, Muhire JD, Aksoy B (2020) Growth regulation and validation of homogeneity in in vitro-derived bleeding heart by molecular markers and spectral analysis of pigments. J Plant Growth Regul. https://doi.org/10.1007/s00344-020-10204-2
Kushalapa KA (1998) Trade liberalisation in sandalwood. In: Radomiljac AM, Ananthapadmanabho HS, Welbourn RM, Rao KS (eds) Sandal and its Products. Proceedings of an international seminar held on 18–19 December 1997 organised by the Institute of Wood Science and Technology (ICFRE) and Karnataka State Forest Department, Bangalore, lndia. Australian Centre for International Agricultural Research, Canberra, Australia, pp 24–26
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592. https://doi.org/10.1042/bst0110591
Manokari M, Priyadharshini S, Shekhawat MS (2021) Microstructural and histochemical variations during in vitro to in vivo plant developments in Aloe vera (L.) Burm.f (Xanthorrhoeaceae). Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2020.113162
Martins JPR, Rodrigues LCA, Conde LT, Gontijo ABPL, Falqueto AR (2020) Anatomical and physiological changes of in vitro-propagated Vriesea imperialis (Bromeliaceae) in the function of sucrose and ventilated containers. Plant Biosyst. https://doi.org/10.1080/11263504.2019.1635223
Mathur A, Mathur AK, Verma P, Yadav S, Gupta ML, Darokar MP (2008) Biological hardening and genetic fidelity testing of micro-cloned progeny of Chlorophytum borivilianum Sant. et Fernand. Afr J Biotechnol 7:1046–1053
Mishra B, Chakraborty S, Sandhya MC, Viswanath S (2018) Sandalwood farming in India: problems and prospects. Indian J Trop Biodiv 26:1–12
Moy RL, Levenson C (2017) Sandalwood ablum oil as a botanical therapeutic in dermatology. J Clin Aesthet Dermatol 10:34–39
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nunez-Ramos JE, Quiala E, Posada L, Mestanza S, Sarmiento L, Daniels D, Arroyo CR, Naranjo B, Vizuete K, Noceda C, Gomez-Kosky R (2020) Morphological and physiological responses of tara (Caesalpinia spinosa (Mol.) O. Kuntz) microshoots to ventilation and sucrose treatments. In Vitro Cell Dev Biol. https://doi.org/10.1007/s11627-020-10104-w
Ora YANR (2012) An integrated management and conservation strategy for sandalwood (Santalum album L.) in West Timor, Indonesia. In: Thomson L, Padolina C, Sami R, Prasad V, Doran J (eds) Paper presented at the regional workshop on sandalwood resource development, research and rrade in the Pacific and Asian region, Port Vila, Vanuatu, 22–25 November 2010, pp 65–89
Poorter H, Niklas KJ, Reich PB, Oleksyn J, Poot P, Mommer L (2012) Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol 193:30–50
Porra RJ (2002) The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynth Res 73:149–156. https://doi.org/10.1023/A:1020470224740
Pospisilova J, Ticha I, Kadlecek P, Haisel D, Plzakova S (1999) Acclimatization of micropropagated plants to ex vitro conditions. Biol Plant 42:481–497
Premkumar A, Mercado JA, Quesada MA (2001) Effects of in vitro tissue culture conditions and acclimatization on the contents of Rubisco, leaf soluble proteins, photosynthetic pigments, and C/N ratio. J Plant Physiol 158:835–840
Rao PS, Bapat VA (1992) Micropropagation of sandalwood (Santalum album L.). In Bajaj YPS (ed) High-tech and micropropagation II Biotechnology in agriculture and forestry, vol 18. Springer, Berlin
Rashkow ED (2014) Perfumed the axe that laid it low: the endangerment of sandalwood in southern India. Indian Econ Soc Hist Rev 51:41–70. https://doi.org/10.1177/0019464613515553
Sandeep C, Manohara TN (2019) Sandalwood in India: historical and cultural significance of Santalum album L. as a basis for its conservation. NeBIO 10:235–241
Santos KR, Pereira MP, Ferreira ACG, Rodrigues LCA, Castro EM, Correa FF, Pereira FJ (2015) Typha domingensis Pers. growth responses to leaf anatomy and photosynthesis as influenced by phosphorus. Aquat Bot 122:47–53. https://doi.org/10.1016/j.aquabot.2015.01.007
Schuldt B, Leuschner C, Brock N, Horna V (2013) Changes in wood density, wood anatomy and hydraulic properties of the xylem along the root-to-shoot flow path in tropical rainforest trees. Tree Physiol 33(2):161–174. https://doi.org/10.1093/treephys/tps122
Shekhawat MS, Manokari M (2016) Optimization of in vitro and ex vitro regeneration and micromorphological studies in Basella alba L. Physiol Mol Biol Plants 22:605–612
Shin KS, Park SY, Paek KY (2013) Sugar metabolism, photosynthesis and growth of in vitro plantlets of Doritaenopsis under controlled microenvironmental conditions. Vitro Cell Dev Biol - Plant 49:445–454. https://doi.org/10.1007/s11627-013-9524-x
Solle HRL, Semiarti E (2016) Micropropagation of Sandalwood (Santalum album L.) Endemic plant from east Nusa Tenggara, Indonesia. AIP Conf Proc 1744(020026):1–5. https://doi.org/10.1063/1.4953500
Teixeira da Silva JA, Kher MM, Soner D, Page T, Zhang X, Nataraj M, Ma G (2016) Sandalwood: basic biology, tissue culture, and genetic transformation. Planta 243:847–887. https://doi.org/10.1007/s00425-015-2452-8
Ticha I, Cap F, Pacovska D, Hofman P, Haisel D, Capkova V, Schafer C (1998) Culture on sugar medium enhances photosynthetic activity and high light resistance of plantlets grown in vitro. Physiol Plant 102:155–162
Tyree MT, Zimmermann MH (2002) Xylem structure and the ascent of sap. Springer, Berlin
Walters RG (2005) Towards an understanding of photosynthetic acclimation. J Exp Bot 56:435–447
Xiao Y, Niu G, Kozai T (2011) Development and application of photoautotrophic micropropagation plant system. Plant Cell Tiss Org Cult 105:149–158
Yan ZB, Guan HY, Han WX, Han TS, Guo YL, Fang JY (2016) Reproductive organ and young tissues show constrained elemental composition in Arabidopsis thaliana. Ann Bot 117:431–439
Yeats TH, Rose JKC (2013) The formation and function of plant cuticles. Plant Physiol 163(1):5–20. https://doi.org/10.1104/pp.113.222737
Acknowledgements
Authors MSS and PS are grateful to the National Medicinal Plants Board, Ministry of AYUSH, Government of India for providing financial support to their laboratory (Grant Number NMPB/IFD/GIA/NR/PL/2018-19/187).
Author information
Authors and Affiliations
Contributions
MSS, SRM, MM, SP, MKB and AD: Conceptualization, investigation, methodology. BSR: Data compilation and hardening of the plants. MSS, MM, and PJ: Writing of original draft and revision of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Human and animal rights
This research did not involve experiments with human or animal participants.
Additional information
Communicated by Klaus Eimert.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shekhawat, M.S., Mehta, S.R., Manokari, M. et al. Morpho-anatomical and physiological changes of Indian sandalwood (Santalum album L.) plantlets in ex vitro conditions to support successful acclimatization for plant mass production. Plant Cell Tiss Organ Cult 147, 423–435 (2021). https://doi.org/10.1007/s11240-021-02136-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-021-02136-w