Abstract
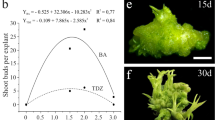
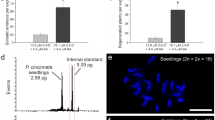
The cellular and biochemical features associated with expression of pluripotency in endosperm cells during de novo shoot organogenesis, as well as the cytogenetic and morpho-agronomic features of regenerated Passiflora foetida triploid plants has been recently reported. Endosperms were cultured on Murashige and Skoog medium supplemented with 0.5, 0.75, 1.0, 1.5, and 2.0 mg L−1 of 6-benzyladenine, thidiazuron, or kinetin. No plant growth regulators were added to the control treatments. Morphogenetic responses were observed only in the presence of cytokinins, especially thidiazuron. The highest percentage of morphogenetic responses (27%) and number of adventitious shoots per explant (68.2) were observed with 1.5 and 2.0 mg L−1 thidiazuron, respectively. The regeneration of adventitious shoots occurred mostly from the outer face of the endosperm by cells that acquired meristematic-like features. Lipids and proteins were consumed rapidly, whereas carbohydrates increased considerably throughout the organogenic process. Cytogenetic analyses confirmed the triploid genotype of endosperm-derived plants, whose vegetative and floral structures were nearly all larger than their diploid counterparts. This study provides a complete morphological characterization of P. foetida endosperm-derived triploid plants and opens new possibilities for genetic breeding of passion fruit.
Key Message
Triploid plants of Passiflora foetida originating from in vitro endosperm culture are morphologically distinct from their diploid counterparts.




Similar content being viewed by others
References
Abreu PP, Souza MM, Santos EA, Pires MV, Pires MM, Almeida AAF (2009) Passion flower hybrids and their use in the ornamental plant market: perspectives for sustainable development with emphasis on Brazil. Euphytica 166:307–315. https://doi.org/10.1007/s10681-008-9835-x
Antoniazzi CA, Faria RB, Carvalho PP, Mikovski AI, Carvalho IF, Matos EM, Reis AC, Viccini LF, Paim Pinto DL, Rocha DI, Otoni WC, Silva ML (2018) In vitro regeneration of triploid plants from mature endosperm culture of commercial passionfruit (Passiflora edulis Sims). Sci Hortic 238:408–415. https://doi.org/10.1016/j.scienta.2018.05.001
AOAC (1990) Ofcial methods of analysis of the association of ofcial analytical chemists. In: Horwitz W (ed) Method 989.05, 15th edn. Association of Official Analytical Chemists, Arlington
Aversano R, Ercolano MR, Caruso I, Fasano C, Rosellini D, Carputo D (2012) Molecular tools for exploring polyploid genomes in plants. Int J Mol Sci 13:10316–10335. https://doi.org/10.3390/ijms130810316
Barker MS, Arrigo N, Baniaga AE, Li Z, Levin DA (2016) On the relative abundance of autopolyploids and allopolyploids. New Phytol 210:391–398. https://doi.org/10.1111/nph.13698
Basumatari M, Das BN (2017) Karyomorphological studies in two species of Bauhinia Linn. and induction of polyploidy in Bauhinia acuminata Linn. Int J Life Sci Sci Res 3:1223–1229. https://doi.org/10.21276/ijlssr.2017.3.4.20
Bewley D, Bradford KJ, Hilhorst HWM, Nonogaki H (2013) Seeds - Physiology of development, germination and dormancy, 3rd edn. Springer, New York
Bowden WM (1945) A list of chromosome numbers in higher plants. I Acanthaceae to Myrtaceae. Am J Bot 32:81–92
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Carvalho CRDE, Saraiva LS (1993) A new heterochromatin banding pattern revealed by modified HKG banding technique in maize chromosomes. Heredity 70:515–519. https://doi.org/10.1038/hdy.1993.74
Cerqueira-Silva CBM, Jesus ON, Santos ESL, Corrêa RX, Souza AP (2014) Genetic breeding and diversity of the genus Passiflora: progress and perspectives in molecular and genetic studies. Int J Mol Sci 15:14122–14152. https://doi.org/10.3390/ijms150814122
Chaturvedi R, Razdan MK, Bhojwani SS (2003) An efficient protocol for the production of triploid plants from endosperm callus of neem, Azadirachta indica A. Juss. J Plant Physiol 160:557–564. https://doi.org/10.1078/0176-1617-00884
Chen ZJ (2010) Molecular mechanisms of polyploidy and hybrid vigor. Trends Plant Sci 15:57–71. https://doi.org/10.1016/j.tplants.2009.12.003
Comai L, Tyagi AP, Winter K, Holmes-Davis R, Reynolds SH, Stevens Y, Byers B (2000) Phenotypic instability and rapid gene silencing in newly formed Arabidopsis allotetraploids. Plant Cell 12:1551–1568. https://doi.org/10.1105/tpc.12.9.1551
Corneillie S, Storme N, Van Acker R, Fangel JU, Bruyne M, Rycke R, Geelen D, Willats WGT, Vanholme B, Boerjan W (2019) Polyploidy affects plant growth and alters cell wall composition. Plant Physiol 179:74–87. https://doi.org/10.1104/pp.18.00967
Corner JH (1976) The seeds of dicotyledons. Cambridge University Press, Cambridge
Crochemore ML, Molinari HB, Stenzel NMC (2003) Caracterização agromorfológica do maracujazeiro (Passiflora spp.). Rev Bras Frutic 25:5–10. https://doi.org/10.1590/s0100-29452003000100004
Dubois M, Gilles K, Hamilton JK, Rebers PA, Smith F (1951) A colorimetric method for the determination of sugars. Nature 168:167. https://doi.org/10.1038/168167a0
Eeckhaut T, Samyn G, Van BE (2002) In vitro polyploidy induction in Rhododendron simsii hybrids. Acta Hortic 572:43–49. https://doi.org/10.17660/ActaHortic.2002.572.4
Faleiro FG, Tadeu N, Junqueira V, Junghans TG, Jesus ON, Miranda D, Otoni WC (2019) Advances in passion fruit (Passiflora spp.) propagation. Rev Bras Frutic 41:1–17. https://doi.org/10.1590/0100-29452019155
Faria RB, Carvalho IF, Rossi AB, Matos EM, Rocha DI, Paim Pinto DL, Otoni WC, Silva ML (2018) High responsiveness in de novo shoot organogenesis induction of Passiflora cristalina (Passifloraceae), a wild Amazonian passion fruit species. In Vitro Cell Dev Biol-Plant 54:166–174. https://doi.org/10.1007/s11627017-9881y
Faleiro FG, Junqueira NTNV, Braga MF, Peixoto JR, Borges RS, Araújo SCB, Andrade SEM, Costa AM, Castellen MS, Vaz APA, Soares-scott MD, Bernacci LC, Andrade GA (2009) BRS Estrela do Cerrado, BRS Rubiflorae, BRS Roseflora: híbridos de maracujazeiro para uso como plantas ornamentais. In: Faleiro FG, Farias Neto AL, Ribeiro Junior WQ (eds) Livros e Cultivares Apresentados no II Encontro da Sociedade Brasileira de Melhoramento de Plantas - Regional - DF. Embrapa Cerrados, Planaltina, DF
Ferreira DAT, Praça-Fontes MM, Vieira AT, Nunes ACP, Clarindo WR (2020) Karyotype and nuclear DNA content variation in Passiflora L. Sci Hortic 272:109532. https://doi.org/10.1016/j.scienta.2020.109532
Ferreira DF (2011) Sisvar: a computer statistical analysis system. Ciênc Agrotec 35:1039–1042. https://doi.org/10.1590/s1413-70542011000600001
Fischer E (2004) Hybrids and hybridization. In: Ulmer T, MacDougal J (eds) Passiflora. Passionflowers of the world. Timber, Portland, pp 362–376
Fonseca KG, Faleiro FG, Junqueira NTV, Barth M, Feldberg NP (2017) Morphoagronomic and molecular characterization of ornamental passion fruit cultivars. Pesq Agropec Bras 52:849–860. https://doi.org/10.1590/S0100-204X2017001000004
Galbraith D, Harkins K, Maddox J, Ayres N, Sharma D, Firoozabady E (1983) Flow cytometric analysis of the cell cycle. Lab Proced Appl 220:1049–1051. https://doi.org/10.1016/b978-0-12-715001-7.50090-1
Góralski G, Popielarska M, Ślesak H, Siwińska D, Batycka M (2005) Organogenesis in endosperm of Actinidia deliciosa cv. Hayward cultured in vitro. Acta Biol Cracov Ser Bot 47:121–128
Guerra M (1986) Citogenética de angiospermas coletadas em Pernambuco, I. Rev Bras Gen 9:21–40
Gui Y, Hong S, Ke S, Skirvin RM (1993) Fruit and vegetative characteristics of endosperm-derived kiwifruit (Actinidia chinensis F.) plants. Euphytica 71:57–62. https://doi.org/10.1007/BF00023467
Harvey MJ (1966) IOPB - chromosome number reports VII. Taxon 15:155–163
Hoshino Y, Miyashita T, Thomas TD (2011) In vitro culture of endosperm and its application in plant breeding: approaches to polyploidy breeding. Sci Hortic 130:1–8. https://doi.org/10.1016/j.scienta.2011.06.041
Husband BC, Sabara HA (2004) Reproductive isolation between autotetraploids and their diploid progenitors in fireweed, Chamerion angustifolium (Onagraceae). New Phytol 161:703–713. https://doi.org/10.1046/j.1469-8137.2004.00998.x
Jesus ON, Oliveira EJ, Soares TL, Faleiro FG (2015) Aplicação de descritores morfoagronômicos utilizados em ensaios de DHE de cultivares de maracujazeiro-doce, ornamental, medicinal, incluindo espécies silvestres e híbridos interespecíficos (Passiflora spp.): manual prático. Embrapa, Brasília, p 45
Julião SA, Ribeiro CV, Lopes JML, Matos EM, Reis AC, Peixoto PHP, Machado MA, Azevedo ALS, Grazul RM, Campos JMS, Viccini LF (2020) Induction of synthetic polyploids and assessment of genomic stability in Lippia alba. Front Plant Sci 11:1–11. https://doi.org/10.3389/fpls.2020.00292
Junqueira AH, Peetz M (2017) Brazilian consumption of flowers and ornamental plants. Ornam Hortic 23:178–184. https://doi.org/10.14295/oh.v23i2.1070
Levan A, Fredga K, Sandberg AA (1964) Nomenclature for centromeric position on chromosomes. Hereditas 52:201–220
Li J, Berger F (2012) Endosperm: food for humankind and fodder for scientific discoveries. New Phytol 195:290–305. https://doi.org/10.1111/j.1469-8137.2012.04182.x
Liao P, Hemmerlin A, Bach TJ, Chye ML (2016) The potential of the mevalonate pathway for enhanced isoprenoid production. Biotechnol Adv 34:697–713. https://doi.org/10.1016/j.biotechadv.2016.03.005
Liqin G, Jianguo Z, Xiaoxia L, Guodong R (2019) Polyploidy-related differential gene expression between diploid and synthesized allotriploid and allotetraploid hybrids of Populus. Mol Breed 39:69. https://doi.org/10.1007/s11032-019-0975-6
Lopes MA, Larkins BA (1993) Endosperm origin, development, and function. Plant Cell 5:1383–1399. https://doi.org/10.1105/tpc.5.10.1383
Loureiro J, Rodriguez E, Doležel J, Santos C (2007) Two new nuclear isolation buffers for plant DNA flow cytometry: a test with 37 species. Ann Bot 100:875–888. https://doi.org/10.1093/annbot/mcm152
Manzoor T, Ahmad T, Bashir MA, Hafiz IA, Silvestri C (2019) Studies on colchicine induced chromosome doubling for enhancement of quality traits in ornamental plants. Plants 8:194. https://doi.org/10.3390/plants8070194
Meletti LMM, Soares-Scott MD, Bernacci LC, Alvares V, Azevedo Filho JA (2011) Caracterização de Passiflora mucronata Lam.: nova alternativa de maracujá ornamental. Rev Bras Hortic Ornam 17:87. https://doi.org/10.14295/rbho.v17i1.721
Melo NF, Cervi AC, Guerra M (2001) Karyology and cytotaxonomy of the genus Passiflora L. (Passifloraceae). Plant Syst Evol 226:6984. https://doi.org/10.1007/s006060170074
Mikosvki AI, Silva NT, Souza CS, Machado MD, Otoni WC, Carvalho IF, Rocha DI, Silva ML (2019) Tissue culture and biotechnological techniques applied to passion fruit with ornamental potential: an overview. Ornam Hortic 25:189–195. https://doi.org/10.14295/oh.v25i2.2036
Miyashita T, Ohashi T, Shibata F, Araki H, Hoshino Y (2009) Plant regeneration with maintenance of the endosperm ploidy level by endosperm culture in Loniceracaerulea var. emphyllocalyx. Plant Cell Tiss Organ Cult 98:291–301. https://doi.org/10.1007/s11240-009-9562-6
Mohamed ME, Hicks RGT, Blakesley D (1996) Shoot regeneration from mature endosperm of Passiflora foetida. Plant Cell Tiss Organ Cult 46:161–164. https://doi.org/10.1007/BF00034851
Muñoz-Concha D (2016) Culture of triploid tissue from the endosperm of an endangered chilean tree species Gomortega keule. J Hortic Sci Biotechnol 91:80–87. https://doi.org/10.1080/14620316.2015.1110995
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakano A, Mii M, Hoshino Y (2021) Simultaneous production of triploid and hexaploid plants by endosperm culture with colchicine treatment in diploid Haemanthus albiflos. Plant Cell Tiss Organ Cult 144:661–669. https://doi.org/10.1007/s11240-020-01974-4
Nishiyama I, Kondo N (1942) Report of the Kihara Institute for. Biol Res 1:29
O’Brien TP, McCully ME (1981) The study of plant structure principles and selected methods. Termarcarphy Pty. Ltd., Melbourne
Oliveira EJ, Koehler AD, Rocha DI, Vieira LM, Pinheiro MVM, Matos EM, Cruz ACF, Silva TCR, Tanaka FAO, Nogueira FTS, Otoni WC (2017) Morpho-histological, histochemical, and molecular evidences related to cellular reprogramming during somatic embryogenesis of the model grass Brachypodium distachyon. Protoplasma 254:2017–2024. https://doi.org/10.1007/s00709-017-1089-9
Otoni WC, Pinto DPL, Rocha DI, Vieira LM, Dias LLC, Silva ML, Silva CVE, Lani RG, Silva LC, Tanaka FAO (2013) Organogenesis and somatic embryogenesis in passionfruit (Passiflora spp.). In: Aslam J, Srivastava OS, Sharma MP (eds) Somatic embryogenesis and gene expression. Narosa Publishing House, New Delhi, pp 1–17
Popielarska M, Ślesak H, Goralski G (2006) Histological and studies on organogenesis in endosperm-derived callus of kiwifruit (Actinidia deliciosa cv. Hayward). Acta Biol Cracov Ser Bot 48:97–104
Rabobank Industry (2016). World Floriculture Map. 2016: Equator Countries Gathering Speed. https://research.rabobank.com/far/en/sectors/regionalfoodagri/worldfloriculturemap-2019.
Randolph LF (1941) Genetic characteristics of the B chromosomes in maize. Genetics 26:608–631
Ramsey J, Schemske DW (1998) Pathways, mechanisms, and rates of polyploid formation in flowering plants. Annu Rev Ecol Syst 29:467–501. https://doi.org/10.1146/annurev.ecolsys.29.1.467
Rangan P (2020) Endosperm variability: from endoreduplication within a seed to higher ploidy across species and its competence. Seed Sci Res 30:173–185. https://doi.org/10.1017/S0960258520000148
Regalado JJ, Carmona-Martín E, Querol V, Veléz CG, Encina CL, Pitta-Alvarez SI (2017) Production of compact petunias through polyploidization. Plant Cell Tiss Organ Cult 129:61–71. https://doi.org/10.1007/s11240-016-1156-5
Rocha DI, Batista DS, Faleiro FG, Rogalski M, Ribeiro LM, Mercadante-Simões MO, Yockteng R, Silva ML, Soares WS, Pinheiro MVM, Pacheco TG, Lopes AS, Viccini LF, Otoni WC (2020) Passion fruit: Passiflora spp. In: Litz RA, Pliego-Alfaro F, Hormaza JI (eds) Biotechnology of fruit and nut crops, 2nd edn. CABI, Wallingford, UK, pp 381–408
Rocha DI, Monte-Bello CC, Dornelas MC (2015) Alternative induction of de novo shoot organogenesis or somatic embryogenesis from in vitro cultures of mature zygotic embryos of passion fruit (Passiflora edulis Sims) is modulated by the ratio between auxin and cytokinin in the medium. Plant Cell Tiss Organ Cult 120:1087–1098. https://doi.org/10.1007/s00709-015-0837-y
Rocha DI, Pinto DLP, Vieira LM, Tanaka FAO, Dornelas MC, Otoni WC (2016) Cellular and molecular changes associated with competence acquisition during passion fruit somatic embryogenesis: ultrastructural characterization and analysis of SERK gene expression. Protoplasma 252:249–257. https://doi.org/10.1007/s00709-015-0837-y
Rocha DI, Vieira LM, Tanaka FAO, Silva LC, Otoni WC (2012) Somatic embryogenesis of a wild passion fruit species Passiflora cincinnata masters: histocytological and histochemical evidence. Protoplasma 249:747–758. https://doi.org/10.1007/s00709-011-0318-x
Sajjad A, Jaskani M, Mehmood A, Ahmad I (2013) Effect of colchicine on in vitro polyploidy induction in African marigold (Tagetes erecta). Pak J Bot 45:1255–1258
Sattler MC, Carvalho CR, Clarindo WR (2015) The polyploidy and its key role in plant breeding. Planta 243:281–296. https://doi.org/10.1007/s00425-015-2450-x
Satyaki PRV, Gehring M (2019) Paternally acting canonical RNA-directed DNA methylation pathway genes sensitize arabidopsis endosperm to paternal genome dosage. Plant Cell 31:1563–1578. https://doi.org/10.1105/tpc.19.00047
Silva-Cardoso IMA, Meira FS, Gomes ACMM, Scherwinski-Pereira JE (2020) Histology, histochemistry and ultrastructure of pre-embryogenic cells determined for direct somatic embryogenesis in the palm tree Syagrus oleracea. Physiol Plant 168:845–875. https://doi.org/10.1111/ppl.13026
Silva GM, Cruz ACF, Otoni WC, Pereira TNS, Rocha DI, Silva ML (2015) Histochemical evaluation of induction of somatic embryogenesis in Passiflora edulis Sims (Passifloraceae). In Vitro Cell Dev Biol-Plant 51:539–545. https://doi.org/10.1007/s11627-015-9699-4
Silva NT, Silva LAS, Reis AC, Machado MD, Matos EM, Viccini LF, Otoni WC, Carvalho IF, Rocha DI, Silva ML (2020) Endosperm culture: a facile and efficient biotechnological tool to generate passion fruit (Passiflora cincinnata Mast.) triploid plants. Plant Cell Tiss Organ Cult 142:613–624. https://doi.org/10.1007/s11240-020-01887-2
Snow N, MacDougal JM (1993) New chromosome reports in Passiflora (Passifloraceae). Syst Bot 18:261. https://doi.org/10.2307/2419402
Soltis PS, Soltis DE (2000) The role of genetic and genomic attributes in the success of polyploids. Proc Natl Acad Sci USA 97:7051–7057. https://doi.org/10.1073/pnas.97.13.7051
Spoelhof JP, Soltis PS, Soltis DE (2017) Pure polyploidy: closing the gaps in autopolyploid research. J Syst Evol 55:340–352. https://doi.org/10.1111/jse.12253
Stacklies W, Redestig H, Scholz M, Walther D, Selbig J (2007) pcaMethods - A bioconductor package providing PCA methods for incomplete data. Bioinformatics 23:1164–1167. https://doi.org/10.1093/bioinformatics/btm069
Stebbins GL (1971) Chromosomal evolution in higher plants. In: G. Ledyard Stebbins (eds.) Edward Arnold Ltd., London
Storey WB (1950) Chromosomes numbers of some species of Passiflora occurring in Hawaii. Pac Sci 4:37–42
Su YH, Liu YB, Bai B, Zhang XS (2015) Establishment of embrionic shoot-root axis in involved in auxin and cytokinin response during Arabidopsis somatic embryogenesis. Front Plant Sci 14:792. https://doi.org/10.3389/fpls.2014.00792
Suda J, Krahulcová A, TrávníčekP RR, Peckert T, Krahulec F (2007) Genome size variation and species relationships in Hieracium sub-genus Pilosella (Asteraceae) as inferred by flow cytometry. Ann Bot 100:1323–1335. https://doi.org/10.1093/aob/mcm218
te Beest M, Le Roux JJ, Richardson DM, Brysting AK, Suda J, Kubešová M, Pyšek P (2012) The more the better? The role of polyploidy in facilitating plant invasions. Ann Bot 109:19–45. https://doi.org/10.1093/aob/mcr277
Thomas TD, Chaturvedi R (2008) Endosperm culture: a novel method for triploid plant production. Plant Cell Tiss Organ Cult 93:1–14. https://doi.org/10.1007/s11240-008-9336-6
Tian Y, Luo C, Lu Y, Tang C, Ouyang Q (2012) Cell cycle synchronization by nutrient modulation. Integr Biol 4:328–334. https://doi.org/10.1039/c2ib00083k
Tiku AR, Razdan MK, Raina SN (2014) Production of triploid plants from endosperm cultures of Phlox drummondii. Biol Plant 58:153–158. https://doi.org/10.1007/s10535-013-0372-7
Tozzi H, Takaki M (2011) Histochemical analysis of seed reserve mobilization in Passiflora edulis Sims f. flavicarpa O. Deg. (yellow passion fruit) during germination. Braz J Biol 71:701–708. https://doi.org/10.1590/s1519-69842011000400015
Van De Peer Y, Mizrachi E, Marchal K (2017) The evolutionary significance of polyploidy. Nat Rev Genet 18:411–424. https://doi.org/10.1038/nrg.2017.26
Vichiato MRM, Vichiato M, Pasqual M, Rodrigues FA, Castro DM (2014) Morphological effects of induced polyploidy in Dendrobium nobile Lindl. (Orchidaceae). Crop Breed Appl Biotechnol 14:154–159. https://doi.org/10.1590/1984-70332014v14n3a23
Wang X, Cheng ZMM, Zhi S, Xu F (2016) Breeding triploid plants: a review. Czech J Genet Plant Breed 52:41–54. https://doi.org/10.17221/151/2015-CJGPB
Worley B, Powers R (2013) Multivariate analysis in metabolomics. Curr Metabolomics 1:92–107. https://doi.org/10.2174/2213235x11301010092
Yan D, Duermeyer L, Leoveanu C, Nambara E (2014) The functions of the endosperm during seed germination. Plant Cell Physiol 55:1521–1533. https://doi.org/10.1093/pcp/pcu089
Yotoko KSC, Dornelas MC, Togni PD, Fonseca TC, Salzano FM, Bonatto SL, Freitas LB (2011) Does variation in genome sizes reflect adaptive or neutral processes? New clues from the Passiflora. PLoS ONE 6:18212. https://doi.org/10.1371/journal.pone.0018212
Acknowledgements
This work was supported by the Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT) (Cuiabá, MT), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) (Belo Horizonte, MG; Grant APQ-00772-19), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) – (Brasília, DF; Finance Code 001), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Brasília, DF; Grant 420913/2018-1). We also thank CNPq for granting a scholarship to MM (DCR-314905/2018-9). We would like to thank Editage (www.editage.com) for English language editing.
Funding
This work was supported by Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT) (Cuiabá, MT), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) (Belo Horizonte, MG; Grant APQ-00772-19), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (Brasília, DF; Finance Code 001), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Brasília, DF; Grant 420913/2018-1).
Author information
Authors and Affiliations
Contributions
MLS, AIM, and DIR designed the study; AIM and NTS established organogenic cultures; AIM, LASS, and LCSB assisted with histological evaluations; MM and NTS performed biochemical analyses; ACR, EMM, and LFV performed cytogenetics and flow cytometry analyses; NTS, CSS, MLS, and MDM performed morphometric evaluations; AIM, DIR, MLS, and WCO wrote the manuscript; MM, IFC, and LFV revised the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Consent for publication
All co-authors have approved the final version of the manuscript.
Additional information
Communicated by M. I. Beruto.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mikovski, A.I., da Silva, N.T., Silva, L.A.S. et al. From endosperm to triploid plants: a stepwise characterization of the de novo shoot organogenesis and morpho-agronomic aspects of an ornamental passion fruit (Passiflora foetida L.). Plant Cell Tiss Organ Cult 147, 239–253 (2021). https://doi.org/10.1007/s11240-021-02120-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-021-02120-4