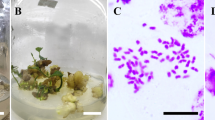
Abstract
A protocol for in vitro induction of tetraploids of the grape rootstock 101-14 Millardet et de Grasset (Vitis riparia × V. rupestris) (101-14 Mgt) (2n = 2x = 38) × Muscadinia rotundifolia cv. Trayshed (2n = 2x = 40) hybrids was established. Different explant materials were exposed to the antimitotic chemical agents colchicine and oryzalin, at three concentrations, for 24, 48 and 72 h. Treated in vitro shoot tips, anthers and pre-embryogenic cultures were regenerated, and their ploidy levels were confirmed by chromosome counts and flow cytometry. It is generally considered that anatomical characteristics of leaves, including leaf stomatal size and number of chloroplasts in guard cells, correlate with ploidy levels of grape plants. Chromosome counts and flow cytometry are labor intensive and difficult, while the number of chloroplasts in stomatal guard cells and stomatal size are easily measured and can be stable and reliable markers. Analysis of stomata revealed clear differences between regenerated diploid and tetraploid plants, and these measurements were used as a preliminary screen, followed by chromosome counts and flow cytometry for confirmation. This study presents the results obtained by various combinations of explants, antimitotic agents, exposure time, concentration and ploidy analysis to evaluate in vitro chromosome doubling. These results provide an easy and effective method for production of tetraploid, potentially fertile Vitis × Muscadinia hybrids for utilization in rootstock breeding programs.




Similar content being viewed by others
References
Agüero C, Meredith CP, Dandekar A (2006) Genetic transformation of Vitis vinifera L. cvs Thompson Seedless and Chardonnay with the pear PGIP and GFP encoding genes. Vitis 45:1
Allum J, Bringloe D, Roberts A (2007) Chromosome doubling in a Rosa rugosa Thunb. hybrid by exposure of in vitro nodes to oryzalin: the effects of node length, oryzalin concentration and exposure time. Plant Cell Rep 26:1977–1984
Awoleye F, Van Duren M, Dolezel J, Novak F (1994) Nuclear DNA content and in vitro induced somatic polyploidization cassava (Manihot esculenta Crantz) breeding. Euphytica 76:195–202
Bartish IV, Korkhovoy VI, Fomina YL, Lim YK (1999) A new approach to obtain polyploid forms of apple. Acta Hort 484:561–564
Chakraborti S, Vijayan K, Roy B, Qadri S (1998) In vitro induction of tetraploidy in mulberry (Morus alba L.). Plant Cell Rep 17:799–803
Chauvin J, Label A, Kermarrec M (2005) In vitro chromosome-doubling in tulip (Tulipa gesneriana L.). J Hortic Sci Biotechnol 80:693–698
Cohen D, Yao J-L (1996) In vitro chromosome doubling of nine Zantedeschia cultivars. Plant Cell, Tissue Organ Cult 47:43–49
de Carvalho JFRP, de Carvalho CRdP, Otoni WC (2005) In vitro induction of polyploidy in annatto (Bixa orellana). Plant Cell, Tissue Organ Cult 80:69–75
Dhooghe E, Grunewald W, Leus L, Van Labeke M-C (2009) In vitro polyploidisation of Helleborus species. Euphytica 165:89–95
Dhooghe E, Van Laere K, Eeckhaut T, Leus L, Van Huylenbroeck J (2011) Mitotic chromosome doubling of plant tissues in vitro. Plant Cell, Tissue Organ Cult 104:359–373
Dolezel J, Greilhuber J, Suda J (2007) Flow cytometry with plants: an overview. In: Dolezel JGJ, Suda J (eds) Flow cytometry with plant cells. Wiley, Weinheim, pp 41–65
Dwivedi N (1986) Induced tetraploidy in mulberry I. Morphological, anatomical and cytological investigations in cultivar RFS-b. Cytologia 51:393–401
Eeckhaut TG, Werbrouck SP, Leus LW, Van Bockstaele EJ, Debergh PC (2004) Chemically induced polyploidization in Spathiphyllum wallisii Regel through somatic embryogenesis. Plant Cell, Tissue Organ Cult 78:241–246
Eeckhaut T, Leus L, Van Huylenbroeck J (2005) Exploitation of flow cytometry for plant breeding. Acta Physiol Plant 27:743–750
Ewald D, Ulrich K, Naujoks G, Schröder M-B (2009) Induction of tetraploid poplar and black locust plants using colchicine: chloroplast number as an early marker for selecting polyploids in vitro. Plant Cell, Tissue Organ Cult 99:353–357
Franks T, He DG, Thomas M (1998) Regeneration of transgenic shape Vitis vinifera L. Sultana plants: genotypic and phenotypic analysis. Mol Breed 4:321–333
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220:1049–1051
Ganga M, Chezhiyan N (2002) Influence of the antimitotic agents colchicine and oryzalin on in vitro regeneration and chromosome doubling of diploid bananas (Musa spp.). J Hortic Sci Biotechnol 77:572–575
Greplova M, Polzerova H, Domkářová J (2009) Intra-and inter-specific crosses of Solanum materials after mitotic polyploidization in vitro. Plant Breed 128:651–657
Hamill S, Smith M, Dodd W (1992) In vitro induction of banana autotetraploids by colchicine treatment of micropropagated diploids. Aust J Bot 40:887–896
Hancock JF (1997) The colchicine story. HortScience 32:1011–1012
Hanson B, Engler D, Moy Y, Newman B, Ralston E, Gutterson N (1999) A simple method to enrich an Agrobacterium-transformed population for plants containing only T-DNA sequences. Plant J 19:727–734
Hebert CJ, Touchell DH, Ranney TG, LeBude AV (2010) In vitro shoot regeneration and polyploid induction of Rhododendron ‘Fragrantissimum Improved’. HortScience 45(5):801–804
Khosravi P, Kermani MJ, Nematzadeh GA, Bihamta MR, Yokoya K (2008) Role of mitotic inhibitors and genotype on chromosome doubling of Rosa. Euphytica 160:267–275
Luckett D (1989) Colchicine mutagenesis is associated with substantial heritable variation in cotton. Euphytica 42:177–182
Murashige T, Nakano R (1966) Tissue culture as a potential tool in obtaining polyploid plants. J Hered 57:115–118
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Notsuka K, Tsuru T, Shiraishi M (2000) Induced polyploid grapes via in vitro chromosome doubling. J Jpn Soc Hortic Sci 69:543–551
Patel G, Olmo H (1955) Cytogenetics of Vitis: I. The hybrid V. vinifera × V. rotundifolia. Am J Bot 42:141–159
Patel G, Olmo H (1957) Interspecific triploid hybrid in grape: (with 10 figures). Caryologia 9:340–352
Petersen KK, Hagberg P, Kristiansen K (2003) Colchicine and oryzalin mediated chromosome doubling in different genotypes of Miscanthus sinensis. Plant Cell, Tissue Organ Cult 73:137–146
Roy A, Leggett G, Koutoulis A (2001) In vitro tetraploid induction and generation of tetraploids from mixoploids in hop (Humulus lupulus L.). Plant Cell Rep 20:489–495
Shi Y, Wang Q, Zhou G, Wang J (1990) Genome engineering breeding of apple in vitro. Fruit Breed Genet 317:13–22
Stebbins GL (1971) Chromosomal evolution in higher plants. Edward Arnold, London
Tal M (1980) Physiology of polyploids. In: Lewis WH (ed) Polyploidy, vol 13. Plenum Press, New York, pp 61–75
Thao NTP, Ureshino K, Miyajima I, Ozaki Y, Okubo H (2003) Induction of tetraploids in ornamental Alocasia through colchicine and oryzalin treatments. Plant Cell, Tissue Organ Cult 72:19–25
Väinölä A (2000) Polyploidization and early screening of Rhododendron hybrids. Euphytica 112:239–244
Van Duren M, Morpurgo R, Dolezel J, Afza R (1996) Induction and verification of autotetraploids in diploid banana (Musa acuminata) by in vitro techniques. Euphytica 88:25–34
Van Tuyl JM, Meijer B, van Diën MP (1992) The use of oryzalin as an alternative for colchicine in in vitro chromosome doubling of Lilium and Nerine. Acta Hort 325:625–630
Viehmannová I, Cusimamani EF, Bechyne M, Vyvadilová M, Greplová M (2009) In vitro induction of polyploidy in yacon (Smallanthus sonchifolius). Plant Cell, Tissue Organ Cult 97:21–25
Xu C, Zhang Y, Cao L, Lu J (2010) Phenolic compounds and antioxidant properties of different grape cultivars grown in China. Food Chem 119:1557–1565
Yahata S, Sato S, Ohara H, Matsui H (2004) Induction of tetraploid in loquat [Eriobotrya japonica] with amiprofos-methyl and colchicine. Hortic Res (Jpn) 3:339–344
Yang X, Cao Z, An L, Wang Y, Fang X (2006) In vitro tetraploid induction via colchicine treatment from diploid somatic embryos in grapevine (Vitis vinifera L.). Euphytica 152:217–224
Zeng S, Chen C, Hong L, Liu J, Deng X (2006) In vitro induction, regeneration and analysis of autotetraploids derived from protoplasts and callus treated with colchicine in Citrus. Plant Cell, Tissue Organ Cult 87:85–93
Zhang Z, Dai H, Xiao M, Liu X (2008) In vitro induction of tetraploids in Phlox subulata L. Euphytica 159:59–65
Zhang Q, Luo F, Liu L, Guo F (2010) In vitro induction of tetraploids in crape myrtle (Lagerstroemia indica L.). Plant Cell, Tissue Organ Cult 101:41–47
Zlesak DC, Thill CA, Anderson NO (2005) Trifluralin-mediated polyploidization of Rosa chinensis minima (Sims) Voss seedlings. Euphytica 141:281–290
Acknowledgments
The authors are grateful to Dr. Changmou Xu for his help with the statistical analysis. This work was supported by the China Scholarship Council (CSC) scholarship in conjunction with the assistance of the Department of Viticulture and Enology at University of California, Davis. Research funding for supplies was received from the California Grape Rootstock Improvement Commission.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, X., Agüero, C.B., Wang, Y. et al. In vitro induction of tetraploids in Vitis × Muscadinia hybrids. Plant Cell Tiss Organ Cult 122, 675–683 (2015). https://doi.org/10.1007/s11240-015-0801-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-015-0801-8