Abstract
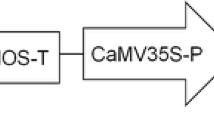
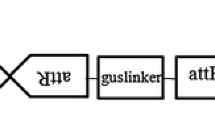
Zoysia tenuifolia Willd. ex Trin. is one of the most popularly cultivated turfgrass. This is the first report of successful plant regeneration and genetic transformation protocols for Z. tenuifolia using Agrobacterium tumefaciens. Initial calli was induced from stem nodes incubated on a Murashige and Skoog (1962) (MS) medium supplemented with 2 mg l−1 2,4-dichlorophenoxyacetic acid (2,4-D) and 1 mg l−1 6-benzyladenine (BA), with a frequency of 53%. Compact calli were selected and subcultured monthly on the fresh medium. Sixty-nine percent of the calli could be induced to regenerate plantlets when the calli incubated on a MS medium supplemented with 0.2 mg l−1 BA under darkness. For genetic transformation, calli were incubated with A. tumefaciens strain EHA105 harboring the binary vector pCAMBIA 1301 which contains the hpt gene as a selectable marker for hygromycin resistance and an intron-containing β-glucuronidase gene (gus-int) as a reporter gene. Following co-cultivation, about 12% of the callus explants produced hygromycin resistant calli on MS medium supplemented with 2 mg l−1 2,4-D, 1 mg l−1 BA, 50 mg l−1 hygromycin, 500 mg l−1 cefotaxime after 8 weeks. Shoots were regenerated following transfer of the resistant calli to shoot induction medium containing 0.2 mg l−1 BA, 50 mg l−1 hygromycin, and 250 mg l−1 cefotaxime, and about 46% of the resistant calli differentiated into shoots. Finally, all the resistant shoots were rooted on 1/2 MS media supplemented with 50 mg l−1 hygromycin, 250 mg l−1 cefotaxime. The transgenic nature of the transformants was demonstrated by the detection of β-glucuronidase activity in the primary transformants and by PCR and Southern hybridization analysis. About 5% of the total inoculated callus explants produced transgenic plants after approximately 5 months. The procedure described will be useful for both, the introduction of desired genes into Z. tenuifolia and the molecular analysis of gene function.


Similar content being viewed by others
References
Al-Khayari JM, Huang FH, Thomson LF, King JW (1989) Plant regeneration of zoysia grass from embryo-derived callus. Crop Sci 29:123–1324
Asano Y (1989) Somatic embryogenesis and protoplast culture in Japanese lawngrass (Zoysia japonica). Plant Cell Rep 8(3):141–143
Asano Y, Katsumoto H, Inokuma C, Kaneko S, Ito Y, Fujiie A (1996) Cytokinin and thiamine requirements and stimulative effects of riboflavin and α-ketoglutarrc acid on embrogenic callus induction from the seeds of Zoysia japonica Steud. J Plant Physiol 149(3–4):413–417
Chai ML, Kim DH (2000) Agrobacterium-mediated transformation of Korean lawngrass (Zoysia japonica). J Kor Soci Hort Sci 41(5):455–458
Chai ML, Lee JM, Kim DH (1998) High efficiency of plant regeneration from seed derived callus of zoysia grass cv. Zenith Kor J Turf Sci 12:195–202
Chen H, Nelson RS, Sherwood JL (1994) Enhanced recovery of transformants of Agrobacterium tumefaciens after freeze–thaw transformation and drug selection. Biotech 16:664–670
Chu CC (1978) The N6 medium and its application to anther culture of cereal crops. In: Proceedings of the symposium on plant tissue culture, Science Press, Beijing, pp 45–50
Compton ME (1999) Dark pretreatment improves adventitious shoot organogenesis from cotyledons of diploid watermelon. Plant Tiss Organ Cult 58:185–188
Dhandapani M, Hong SB, Aswath CR, Kim DH (2008) Regeneration of zoysia grass (Zoysia matrella L. Merr.) cv. Konhee from young inflorescences and stem nodes. In Vitro Cell Dev Biol Plant 44:8–13
Duble RL (1996) Turfgrasses: their management and use in the southern zone. Texas A&M University Press, College Station
Evans DA, Sharp WR, Flick CE (1981) Plant regeneration from cell cultures. Hort Rev 3:214–314
Ge Y, Norton T, Wang ZY (2006) Transgenic zoysiagrass (Zoysia japonica) plants obtained by Agrobacterium-mediated transformation. Plant Cell Rep 25:792–798
Hartmann HT, Kester DE, Davies FT Jr, Geneve RL (1997) Plant propagation: principles and practices, 6th edn. Prentice-Hall Inc, Englewood Cliffs
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J 6:271–282
Inokuma C, Sugiura K, Cho C, Okawara R, Kaneko S (1996) Plant regeneration from protoplasts of Japanese lawngrass. Plant Cell Rep 15(12):737–741
Inokuma C, Sugiura K, Imaizumi N, Cho C (1998) Japanese lawngrass (Zoysia japonica Steud.) plants regenerated from protoplasts. Plant Cell Rep 17(5):334–338
Jefferson RA (1987) Assaying chimeric genes in plants: the Gus gene fusion system. Plant Mol Biol Rep 5:387–405
Jun J, Yae B, Yang M (1996) Influence of cultivar, light condition and pretreatment on adventitious shoot regeneration from leaves, internodes and petioles of Malus domestica Borkh in vitro. J Kor Soc Hort Sci 37(5):700–703
Li MR, Li HQ (2003) A simple and highly efficient Agrobacterium-mediated rice transformation system. Acta Biol Exp Sin 36:289–294
Liu J, Zhang X, Poudyal BK, Zhang Y, Jiao Z, Qi J (2009a) Adventitious shoot regeneration from the leaves of in vitro grown ‘Zhongli 1’ pear (Pyrus spp.). Front Agric China 3(1):60–61
Liu L, Fan X, Zhang J, Yan M, Bao M (2009b) Long-term cultured callus and the effect factor of high-frequency plantlet regeneration and somatic embryogenesis maintenance in Zoysia japonica. In Vitro Cell Dev Biol 45:673–680
Muhammad AL, Ye GN, Weeden NF, Reisch BI (1994) A simple and efficient method for DNA extraction from grapevine cultivars, Vitis species and Ampelopsis. Plant Mol Biol Rep 12:6–13
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497
Rim TW, Kim KY, Choi GJ (2002) Callus induction and plant regeneration from seeds of Zoysia japonica Steud. J Kor Soci Hort Sci 21(2):49–52
Toriyama K, Hinata K (1985) Panicle culture in liquid media for obtaining anther calli and protoplasts in rice. Japan J Breed 35(Suppl 1): 449–452
Toyama K, Bae CH, Kang JG, Lim YP, Adachi T, Riu KZ, Song PS, Lee HY (2003) Production of herbicide-tolerant zoysiagrass by Agrobacterium-mediated transformation. Mol Cells 16(1):19–27
Zhang L, Wu DX, Hu FR, Ma CX (2004) Optimization on major factors for tissue culture and genetic transformation of Japanese lawngrass (Zoysia japonica Steud.). Acta Pratac Sinica 4:100–105
Zhang L, Wu D, Zhang L, Yang C (2007) Agrobacterium-mediated transformation of Japanese lawngrass (Zoysia japonica Steud.) containing a synthetic cryIA(b) gene from Bacillus thuringiensis. Plant Breeding 126:428–432
Acknowledgments
This research was supported by the CAS/SAFEA International Partner-ship Program for Creative Research Teams.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, M., Li, H., Hu, X. et al. An Agrobacterium tumefaciens-mediated transformation system using callus of Zoysia tenuifolia Willd. ex Trin. Plant Cell Tiss Organ Cult 102, 321–327 (2010). https://doi.org/10.1007/s11240-010-9736-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-010-9736-2