Abstract
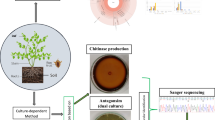
In this article we describe the identification of endophytic bacteria belonging to three groups isolated from shoot tip cultures of banana cv. Grand Naine in a recent study (Thomas et al. 2008) based on partial 16S rRNA gene sequence homology analysis. The first group included banana stocks that displayed obvious colony growth on MS based tissue culture medium during the first in vitro passage. The second group constituted stocks that were tissue index-negative for cultivable bacteria initially but turned index-positive after a few to several (4–8) in vitro passages while the third group formed one sub-stock that turned index-positive after about 18 passages. The organisms belonged to about 20 different genera comprising of α, β, γ-proteobacteria, Gram-positive firmicutes and actinobacteria. Visibly expressing easily cultured organisms during the first in vitro passage included Enterobacter, Klebsiella, Ochrobactrum, Pantoea, Staphylococcus and Bacillus spp. Organisms of second group that were not detected or non-culturable originally constituted Brevundimonas, Methylobacterium, Alcaligenes, Ralstonia, Pseudomonas, Corynebacterium, Microbacterium, Staphylococcus, Oceanobacillus and Bacillus spp. while the third group that turned cultivable after extended in vitro culturing included mostly non-filamentous actinobacteria (Brachybacterium, Brevibacterium, Kocuria and Tetrasphaera spp.). The identification results suggested that the endophytes of second and third groups were not strictly obligate or fastidious microbes but those surviving in viable but-non-culturable (VBNC) state and displaying gradual activation to cultivable form during continuous tissue culturing. Several of the organisms isolated are known as beneficial ones in agriculture while some organisms have possible implications in human health. The use of tissue cultures for isolating uncommon endophytes is discussed.

Similar content being viewed by others
Abbreviations
- BIM:
-
Bacteriological indexing medium
- NA:
-
Nutrient agar
- OC:
-
Original culture
- TCM:
-
Tissue culture medium
- TSA:
-
Trypticasein soy agar
- VA:
-
Viss et al. (1991) agar
- VBNC:
-
Viable but non-culturable
References
Alexander E, Pham D, Steck TR (1999) The viable-but-nonculturable condition is induced by copper in Agrobacterium tumefaciens and Rhizobium leguminosarum. Appl Environ Microbiol 65:3754–3756
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (2005) Current protocols in molecular biology. vol 1. Wiley, New York, pp 2.4.1–2.4.2
Azevedo JL, Maccheroni W Jr, Pereira JO, Araújo WL (2000) Endophytic microorganisms: a review on insect control and recent advances on tropical plants. Electronic J Biotech 3:40–65
Basaglia M, Povolo S, Casella S (2007) Resuscitation of viable but not culturable Sinorhizobium meliloti 41 pRP4-luc: effects of oxygen and host plant. Curr Microbiol 54:167–174
Bell CR, Dickie GA, Harvey WLG, Chan JWYF (1995) Endophytic bacteria in grapevine. Can J Microbiol 41:46–63
Cao L, Qiu Z, You J, Tan H, Zhou (2005) Isolation and characterization of endophytic streptomycete antagonists of fusarium wilt pathogen from surface sterilized banana roots. FEMS Microbiol Lett 247:147–152
Cappuccino JG, Sherman N (1996) Microbiology—a laboratory manual, 4th edn. Addison Wesley Longman Inc., Harlow
Coenye T, Goris J, Spilker T, Vandamme P, LiPuma JJ (2002) Characterization of unusual bacteria isolated from respiratory secretions of cystic fibrosis patients and description of Inquilinus limosus gen. nov., sp. nov. J Clin Microbiol 40:2062–2069
Colwell RR, Brayton PR, Grimes DJ, Roszak DR, Huq SA, Palmer LM (1985) Viable, but non-culturable, Vibrio cholerae and related pathogens in the environment: implications for release of genetically engineered microorganisms. Biotechnol 3:817–820
Conn VM, Franco CMM (2004) Effect of microbial inoculants on the indigenous actinobacterial endophyte population in the roots of wheat as determined by terminal restriction fragment length polymorphism. Appl Environ Microbiol 70:6407–6413
Drancourt M, Bollet C, Carlioz A, Martelin R, Gayral JP, Rauult D (2000) 16S ribosomal DNA sequence analysis of a large collection of environmental and clinical unidentifiable bacterial isolates. J Clin Microbiol 38:3623–3630
Frank DN, Wysocki A, Specht-Glick DD, Rooney A, Feldman RA, St Amand AL, Pace NR, Trent J (2005) Comparison of culture and molecular-based identification of microbial constituents of chronic human wounds (NCBI acc no. DQ170425)
Goldenberger D, Grubenmann M, Dutly F (2002) Corynebacterium amycolatum isolated from urine of a 27-year-old female with recurrent urinary tract infection (NCBI acc no. AY831726)
Habiba U, Reza S, Saha ML, Khan MR, Hadiuzzaman S (2002) Endogenous bacterial contamination during in vitro culture of banana: identification and prevention. Plant Tiss Cult 12:117–124
Hallmann J (2001) Plant interactions with endophytic bacteria. In: Jeger MJ, Spence NJ (eds) Biotic interactions in plant-pathogen associations, CABI Publishing, Oxon, Wallingford, pp 87–119
Herman EB (2004) Recent advances in plant tissue culture viii. Microbial contaminants in plant tissue cultures: solutions and opportunities 1996–2003. Agritech Consultants, Inc., Shrub Oak, USA
Ko KS, Oh WS, Song J-H (2005) Two novel Bacillus species isolated from a patient with neonatal sepsis: Bacillus neonatiensis sp. nov. and Bacillus koguryoae sp. nov. (NCBI acc no. AY904032)
Kulkarni AA, Kelkar SM, Watwe MG, Krishnamurthy MV (2007) Characterization and control of endophytic bacterial contaminants in in vitro cultures of Piper spp., Taxus baccata subsp. wallichiana, and Withania somnifera. Can J Microbiol. 53:63–74
Leifert C, Cassells AC (2001) Microbial hazards in plant tissue and cell cultures. In vitro Cell Dev Biol Plant 37:133–138
Mehnaz S, Mirza MS, Haurat J, Bally R, Normand P, Bano A, Malik KA (2001) Isolation and 16S rRNA sequence analysis of the beneficial bacteria from the rhizosphere of rice. Can J Microbiol 47:110–1177
Nunes C, Usall J, Texido N, Fons E, Vinas I (2002) Post-harvest biological control by Pantoea agglomerans (CPA-2) on Golden Delicious apples. J Appl Microbiol 92:247–255
Panicker B, Thomas P, Janakiram T, Venugopalan R (2007) Influence of cytokinin levels on in vitro propagation of shy suckering chrysanthemum ‘Arka Swarna’ and activation of endophytic bacteria. In vitro Cell Dev Biol Plant 43:614–622
Reiter B, Sessitsch A (2006) Bacterial endophytes of the wildflower Crocus albiflorus analyzed by characterization of isolates and by a cultivation-independent approach. Can J Microbiol 52:140–149
Reiter B, Pfeifer U, Schwab H, Sessitsch A (2002) Response of endophytic bacterial communities in potato plants to infection with Erwinia carotovora subsp. atroseptica. Appl Environ Microbiol 68:2261–2268
Rosenblueth M, Martínez-Romero E (2006) Bacterial endophytes and their interaction with hosts. Mol Plant Microbe Interact 8:827–837
Strosse H, Van den Houwe I, Panis B (2004) Banana cell and tissue culture—a review. In: Jain SM, Swennen R (eds) Banana improvement: Cellular, molecular biology, and induced mutations, (http://www.fao.org/docrep/007/ae216e/ae216e00.html)
Suslow TV, Schroth MN, Isaka M (1982) Application of a rapid method for Gram differentiation of plant pathogenic and saprophytic bacteria without staining. Phytopathol 72:917–918
Thomas P (2004a) A three-step screening procedure for detection of covert and endophytic bacteria in plant tissue cultures. Curr Sci 87:67–72
Thomas P (2004b) In vitro decline in plant cultures: detection of a legion of covert bacteria as the cause for degeneration of long-term micropropagated triploid watermelon cultures. Plant Cell Tiss Org Cult 77:173–179
Thomas P (2006) Isolation of an ethanol-tolerant endospore-forming Gram-negative Brevibacillus sp. as a covert contaminant in grape tissue cultures. J Appl Microbiol 101:764–774
Thomas P, Prabhakara BS, Pitchaimuthu M (2006) Cleansing the long-term micropropagated triploid watermelon cultures from covert bacteria and field testing the plants for clonal fidelity and fertility during the 7–10 year period in vitro. Plant Cell Tiss Org Cult 85:317–329
Thomas P, Kumari S, Swarna GK, Gowda TKS (2007a) Papaya shoot tip associated endophytic bacteria isolated from in vitro cultures and host-endophyte interaction in vitro and in vivo. Can J Microbiol 53:380–390
Thomas P, Kumari S, Swarna GK, Prakash DP, Dinesh MR (2007b) Ubiquitous presence of fastidious endophytic bacteria in field shoots and index-negative apparently clean shoot-tip cultures of papaya. Plant Cell Rep 26:1491–1499
Thomas P, Swarna GK, Patil P (2008) Ubiquitous presence of normally non-cultivable endophytic bacteria in field shoot tips of banana and their gradual activation to quiescent cultivable form in tissue cultures. Plant Cell Tiss Org Cult, doi:10.1007/s11240-008-9340-x
Van den Houwe I, Swennen R (2000) Characterization and control of bacterial contaminants in in vitro cultures of banana (Musa spp.). Acta Hort 530:69–79
Van den Houwe I, Gun J, Swennen R (1998) Bacterial contamination in Musa shoot tip cultures. Acta Hort 490:485–492
Viss PR, Brooks EM, Driver JA (1991) A simplified method for the control of bacterial contamination in woody plant tissue culture. In vitro Cell Dev Biol Plant 27:42
Xu HS, Roberts N, Singleton FL, Attwell RW, Grimes DJ, Colwell RR (1982) Survival and viability of nonculturable E. coli and V. cholerae in the estuarine and marine environment. Microbial Ecol 8:313–323
Acknowledgments
This study was supported by a grant from the Department of Biotechnology, Govt. of India under the project ‘Identification of covert endophytic microbes in plant tissue cultures and their management and control’. Contributions from D. P. Prakash and V. Bidari during their brief association with the project and laboratory assistance by N. Shivrudriah and B. Hanumanthraju are acknowledged. This publication bears IIHR Contribution No. 10/2007.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supply of live bacterial cultures or genetic material for research purpose is subject to their revival from glycerol stocks (as some of the organisms showed poor tolerance) and the requestor obtaining written permission from the Director General, Indian Council of Agricultural Research, New Delhi-110001.
Rights and permissions
About this article
Cite this article
Thomas, P., Swarna, G.K., Roy, P.K. et al. Identification of culturable and originally non-culturable endophytic bacteria isolated from shoot tip cultures of banana cv. Grand Naine. Plant Cell Tiss Organ Cult 93, 55–63 (2008). https://doi.org/10.1007/s11240-008-9341-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-008-9341-9