Abstract
The role of the A>G polymorphism at position 19911 in the prothrombin gene (factor [F] 2 at rs3136516) as a risk factor for venous thromboembolism [VTE] is still unclear. To evaluate the presence of the F2 polymorphism in VTE patients compared to healthy blood donors and to adjust the results for common inherited thrombophilias [IT], age at onset and blood group [BG], and to calculate the risk of VTE recurrence. We investigated 1012 Caucasian patients with a diagnosis of VTE for the presence of the F2 rs3136516 polymorphism and compared these with 902 healthy blood donors. Odds ratios [OR] together with their 95% confidence intervals were calculated adjusted for F5 at rs6025, F2 at rs1799963, blood group, age and gender. In addition, we evaluated the risk of recurrent VTE during patient follow-up calculating hazard ratios [HR] together with their 95% CI. Compared with the AA wildtype, the F2 GG and AG genotypes (rs3136516) were associated with VTE (OR 1.48 and 1.45). The OR in F5 carriers compared to controls was 5.68 and 2.38 in patients with F2 (rs1799963). BG “non-O” was significantly more often diagnosed in patients compared to BG “O” (OR 2.74). VTE recurrence more often occurred in males (HR 2.3) and in carriers with combined thrombophilia (HR 2.11). Noteworthy, the rs3136516 polymorphism alone was not associated significantly with recurrence. In Caucasian patients with VTE the F2 GG/GA genotypes (rs3136516) were moderate risk factors for VTE. Recurrence was associated with male gender and combined thrombophilia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acquired risk factors such as surgery, immobilization, pregnancy, smoking, oral contraceptives and obesity, as well as inherited risk factors simultaneously contribute to the onset of venous thromboembolism [VTE] [1,2,3]. Rare inherited risk factors such as antithrombin-, protein C- and protein S deficiency are considered as high risk factors for VTE, whereas the factor [F] 5 [rs6025] and F2 [rs1799963] mutations are considered as more commonly diagnosed but being rather mild risk factors [1,2,3,4,5]. The A>G polymorphism at position 19911 of the prothrombin gene, e.g. F2 at rs3136516, located in intron 13 of the prothrombin gene is associated with increased plasma prothrombin levels, but its role as a risk factor for VTE is still unclear [6,7,8,9,10,11]. There is a small number of case control studies investigating the role of the F2 at rs3136516 polymorphism and its association with VTE. Of these, two reports showed a moderate risk for VTE [8, 9], whereas another study showed no statistically significance [10].
Therefore, the objectives of this study were to (i) evaluate the presence of the prothrombin 19911 A>G polymorphism in adolescent and younger adult Caucasian VTE in- and outpatients compared to healthy blood donors and to adjust the results obtained for common inherited thrombophilias [IT], age at onset and blood group [BG]. The second objective of this study was to (ii) calculate in the patient cohort the risk of VTE recurrence independently of gender, common mild ITs including the F2 at rs3136516 polymorphism, age at first VTE onset and blood group.
Material and methods
Study population
Consecutive patients with a first symptomatic VTE were recruited between December 2008 and 2018 whether or not prothrombotic risk factors were present. Screening of thrombophilia was performed in 1012 adolescent and adult patients [14 to < 60 years] and 902 healthy blood donors [18 to < 61 years] from the same catchment area.
Inclusion and exclusion criteria
Corresponding to the following criteria patients were included into the study: (i) age from puberty status > Tanner 2 [12] to 60 years at first VTE onset, (ii) objectively confirmed thrombosis and re-thrombosis with established imaging procedures such as Duplex- or Doppler- ultrasonography, computed tomography or magnetic resonance tomography for venous thrombosis and spiral computed pulmonary angiography or lung perfusion scintigraphy for pulmonary embolism. Patients were excluded from the study when they suffer from (i) central line-associated VTE, VTE linked to malignancy, antiphospholipid syndrome, inherited antithrombin-, protein C- or protein S-deficiency or (ii) when they were lost to follow-up or did not abide by the follow-up visits.
Study aims
The primary study objective was to determine the individual VTE risk of the F2 mutation at rs3136516 in Caucasian VTE patients [n = 1012] compared to healthy blood donors [n = 902] adjusted for (i) common ITs, e.g. F5 at rs6025 and F2 at rs1799963, age at onset and blood group [1,2,3, 13, 14]. The secondary study objective was to investigate the time to VTE recurrence after withdrawal of antithrombotic therapy, adjust for the above listed potential confounders.
Clinical procedures
Following a first VTE onset (in- and outpatients) and during regular follow-up visits [3–6 months (adolescents), 9–12 months, 2 years (adults)] we evaluated and re-evaluated patient’s disease history and possible risk factors causing VTE, such as the use of oral contraceptives, pregnancy, obesity, immobilization, surgery and smoking. Clinical data collection also included laboratory test results, antithrombotic therapy, including adherence to anticoagulation and duration and family history of VTE. Adolescent and adult patients were treated independently from the underlying IT risk factors according to the latest antithrombotic VTE therapy guideline. (Online Supplement) [15,16,17].
Details on blood sample collection are depicted in detail in the online supplement.
Laboratory analyses
With written or oral parental consent mutations in F2 at rs3136516, F5 at rs6025 and F2 at rs1799963 as well as circulating levels of antithrombin, coagulation inhibitors, d-dimer concentration as well as lupus anticoagulants and antiphospholipid antibodies were investigated with standard laboratory techniques at VTE onset and were repeated during routine follow up visits [12, 14]. Especially, the analyses of protein C and protein S were performed at least three months after the index event and/or withdrawal of vitamin-K-antagonists and deficiency states of antithrombin, protein C or protein S were confirmed if values repeatedly persisted below the age-related reference ranges [14]. Criteria for the hereditary nature of a hemostatic defect were its presence in at least one further first or second-degree family member and/or the identification of a causative gene mutation.
Single nucleotide polymorphism [SNP] Genotyping for F2 A19911G (LRG_551t1:c.1726-59G>A, rs3136516) was performed by TaqMan PCR. The assay includes two allele specific TaqMan probes containing distinct fluorescent dyes and highly specific PCR primers to detect the target (Assay ID: C__11661574_10, ThermoFisher Scientific, RRID:SCR_018060). ABI 7900HT Fast Real-Time PCR System was used for analysis. Of note, the specific snip applied (rs3136516) does not show a significant deviation from Hardy Weinberg equilibrium in the healthy cohort of blood donors (p = 0.55).
Statistics
Statistical analyses were performed using the MedCalc® software bvba (version 16.4.3, Ostend, Belgium, RRID:SCR_015044) and StatView 5 software packages (SAS Institute Inc., RRID:SCR_017411). Continuous variables were expressed as mean (± standard deviation) or median [minimum–maximum] values and categorical data were expressed as counts and percentages. The Wilcoxon–Mann–Whitney-U-test investigated differences in continuous non-normal distributed variables between two independent groups. The Chi-square-test was used to analyze differences in binary and categorical data between for the primary study aim Odds ratios [OR] and their corresponding 95% confidence intervals [CI] were calculated using an univariable logistic regression model comparing patients with healthy controls (blood donors). The homozygous GG genotype and the heterozygous AG were compared with the AA wildtype (F2 at rs3136516). In order to evaluate an independent contribution to the risk of VTE and to adjust for further potential confounders (common IT, age at onset, blood group) [1,2,3, 13, 14] these variables were entered into a backward multivariable logistic regression model. In addition, to test for possible statistical interactions between F5 rs6025 and F2 rs313616 genotypes a multiplicative scale was incorporated in the model. Per “a priori” definition this backward model included variables with a p value of ≤ 0.2 and, vice versa, removed variables from the model if p was > 0.21. For the secondary study objective, i.e. the time to recurrence, we calculated the probability of VTE-free survival as a function of time utilizing the method of Kaplan and Meier (univariable analysis). The log rank test was used to test for differences in recurrence-free survival between groups. On the basis of previous reports (data presented in the Online Suppl. Tables 1 and 2) minimum sample size calculation for the comparison of two proportions was performed assuming a type I error of 0.05 and a type II error of 0.20 (sampling comparison of proportions) [18]. Patients were withdrawn from the survival analysis (censored cases) either at death unrelated to VTE recurrence or at loss to follow-up using data of the last clinical follow-up visit. In order to evaluate an independent contribution to the risk of recurrent VTE (dichotomous variables: recurrence “yes” versus “no recurrence”) and to adjust for further potential confounders (gender, common IT, age at first VTE onset, blood group) the hazard ratio (HR) together with 95% confidence intervals (CI) were estimated from Cox’s proportional hazards model. To further test the relationship between independent and dependent variables, the likelihood ratio test was performed. To test the proportional hazard assumption, one of the prerequisites for applying the Log-rank test or the Cox regression model fit, we used the cox.zph procedure in the package ‘survival’ (v3.1.8; Therneau TM, 2019) in R 3.4.3 [19]. A p value > 0.05 indicates no violation of the proportional hazard assumption. The recurrence rates were calculated as the number of recurrent events per 100 person-years.
Ethics
The underlying multicenter cohort study was approved by the medical ethics committee of the University of Münster & Kiel [B304/16], Germany and written informed consent was provided in all cases prior to study participation.
Results
Demographic data and thrombotic locations of patients (n = 1012) and healthy controls (n = 902) are depicted in Table 1. Frequency distribution of single & combined thrombophilic risk factors are given in Table 2 and in the online supplement.
Primary study aim (univariable analysis: Table 3)
Compared with the AA wildtype the GG genotype (rs3136516) increases the risk of VTE with an OR of 1.39 (CI 1.04–1.73) and 1.21 (CI 0.97–1.51) in carriers of the AG variant. In addition, in the cohort investigated the OR of F5 carriers compared to healthy blood donors was 6.35 (CI 4.59–8.77) and 2.44 (1.47–4.05) in patients with F2 (rs1799963). Combinations between the homozygous F2 GG genotype at rs3136516 and F5 at rs6025 did not significantly influence the thrombotic risk when comparing patient with controls (OR 0.56; CI 0.29–1.09) and combinations between the factor 2 rs1799963 GA/AA variants and the homozygous GG (rs3136516) genotype were neither detected in cases nor in healthy controls, respectively. BG “non-O” was significantly more often found in patients with VTE compared to BG “O” (OR 3.77, CI 3.04–4.69). Furthermore, increasing age per year at first VTE onset enhances the VTE risk significantly by an OR of 1.01 (CI 1.02–1.04).
Multivariable analysis (Table 3)
Backward logistic regression showed that compared with the AA wildtype the GG genotype (rs3136516) was independently associated with a diagnosis of VTE with an OR of 1.48 (95% CI 1.06–2.06) and with the OR of 1.45 (95% CI 1.1–1.92) in carriers in which the AG variant was genotyped. In addition, the OR of F5 in patients compared to healthy blood donors was 5.68 (95% CI 3.94–8.20) and 2.38 (95% CI 1.35–4.20) in patients with F2 (rs1799963). BG “non-O” was significantly more often found in patients with VTE compared to BG “O” (OR 2.74; 95% CI 2.11–3.55). Again, as depicted in univariable analysis increasing age per year at first onset retained its significance also in multivariate analysis (OR 1.03; 95% CI 1.02–1.04). Due to the “a priori” defined statistical cut-off p-values combinations between F5 rs6025 and F2 rs3135616 (multiplicative scale) were removed from the multivariable model and were not included in the final analysis.
Secondary study aim
Within a mean time of 51.2 months (standard deviation: 43.5), recurrent VTE events (provoked 77%; locations depicted in Table 1) occurred in 178 out of 1012 patients (17.6%) following the first VTE onset (Fig. 1): 26.5% in men and 13.1% in women (p < 0.001). Gender associated incidence rates per 100 patient years (Table 4) were 4.3 (95% CI 3.4–5.3) in men and 1.9 (95% CI 1.6–2.4) in women (p < 0.0001). Further details of thrombotic locations and AC prior VTE recurrence are described in the online supplement material.
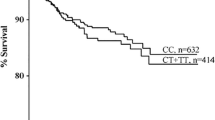
Whereas no significant associations were found between the risk of recurrent VTE and individuals classified as carriers of (i) the F2 mutation at rs3136516 “GG” subjects (OR 1.41; 95% CI 0.9–2.16) or in patients carrying (ii) the F2 rs179963 “GA” genotype (OR 1.81; 95% CI 0.87–3.76), results of the logistic regression model demonstrates that (iii) heterozygous F5 carriers had an increased OR of 1.86 (95% CI 1.22–2.83) to suffer from recurrent VTE with a further increase in patients additionally carrying the F2 GG/GA variant (rs3136516): OR 3.34 (95% CI 1.62–6.89). Patients with at least any IT compared to patients with no IT showed an increased odds ratio of 1.7 (95% CI 1.2–2.3) for a recurrence of VTE, while in patients with combined ITs the OR increased to 2.4 (95% CI 1.7–3.4). Of note, there was no significant association between the presence or absence of provoking risk factors and inherited thrombophilia (p = 0.93). The recurrence-free survival is depicted in Fig. 2: 32.7% of patients carrying combined ITs compared to 15.6% without IT developed a second thromboembolic event within 36 months (min–max: 1–120; log rank test: p < 0.0001). Multivariable analysis including time to recurrence demonstrates that male gender [hazard ratio (HR) 2.3; 95% CI 1.7–3.0] and combined ITs (HR 2.11; 95% CI 1.5–3.0) showed a significant impact on the risk of VTE recurrence. Noteworthy, in this specific data set, gender, F5, F2 rs3136516, F2 rs179963 and blood group did not violate the proportional hazard assumption. Remarkably, age, both as continuous term or grouped into three equal sized groups did violate the proportional hazard assumption and was thus not included in the multivariable analysis. Due to the “a priori” defined statistical cut-off p values the comparison between BG O and non-O was removed from the multivariate model. Incidence rates of recurrence per 100 patient years are depicted in detail in Table 4.
Probability of recurrence-free survival (y-axis) following a first symptomatic VTE onset is depicted: 32.7% of patients carrying combined thrombophilia variants compared to 15.6% without thrombophilia developed a second thrombotic event (log rank value: p < 0.0001). In addition, recurrence-free survival in patients with any thrombophilia is depicted
Discussion
In the present longitudinal study in German patients with VTE we have shown that the presence of the F2 mutation at rs3136516 either in its GG or its GA genotype compared to the AA variant plays a moderate but independent role at first VTE onset with an adjusted odds ratio of 1.48 (GG genotype) and 1.45 (GA genotype), respectively. These findings were in agreement to study results described earlier in 2006 in an Italian cohort of patients (OR 1.5) and in line with data derived from the Netherlands with a 1.3-fold increased OR to suffer from VTE. As in our study, in the latter two cohorts the F2 rs3136516 GG genotypes were compared to AA carriers [8, 9]. The observation of a moderately enhanced association with VTE reported by us and by both cohorts published in 2006, however, is in contrast to findings reported by Perez-Ceballos et al. in 2002 and by Aradjanski et al. in 2014 [7, 10]. A summary of pooled data of previous reports [7,8,9,10] including our patients is depicted in the online supplement. Pooling results of overall four studies with 6150 patients with VTE and 6588 population-based controls, a modest increased association with VTE in carriers of the F2 (rs3136516) GG genotype of 1.3 (95% CI 1.2–1.4; p < 0.0001; heterogeneity [I2] 0.0%) was calculated. Interestingly, when investigating the role of this F2 intron polymorphism in a genome-wide association study the risk association with VTE was also modestly increased with an OR of 1.08 (1.06–1.11) [20].
In our cohort, combinations between the homozygous F2 GG genotype at rs3136516 and F5 at rs6025 additionally incorporated in the statistical model in a multiplicative scale did not significantly influence the thrombotic risk at VTE onset. This observation was in contrast to both cohort studies reported in 2006 [8, 9]. Combinations of the GG genotype with the F2 variant at rs1799963 GA/AA genotypes were not found, in accordance to previously reported data [8].
Of note, data of the multivariable analysis (primary study objective) confirmed findings by others that blood group “non-O” is an independent risk factor for a first VTE onset [21].
The contribution of male gender, age at first VTE onset and IT risk factors on VTE events were additionally investigated as secondary study aim in the patient follow-up study. During the patient-follow up after the first withdrawal of therapeutic AC we could demonstrate a significant risk increase of recurrent VTE in males (annual incidence rate of 4.3) compared to females (annual incidence rate of 1.9). Male gender remained an independent risk factor for recurrent VTE in multivariable analysis. The findings of our study, where VTE events were associated in the majority of cases with provoking risk factors, are in line with the higher recurrence risk in men following unprovoked VTE [22,23,24]. In contrast to findings from 2011 derived from a patient level meta-analysis in which the risk of recurrent VTE in men with a first provoked vascular occlusion was similar to that in women, the meta-analysis by McRae [23] found an enhanced risk for VTE recurrence in men. In our study the aforementioned increased risk of recurrence in men was neither affected by age at first VTE onset nor by underlying IT risk factors. Additionally, in the follow-up study, probably due to the small number of patients affected, the increased risk of blood group “non-O” and vice versa the protective role of BG O lost its significance in multivariable Cox regression analysis. Apart from possible explanations for the higher VTE recurrence in males compared to females discussed in more detail by Douketis [22], another explanation may be male gender per se: endogenous testosterone levels in males compared to females are clearly higher. Enhanced testosterone levels are associated with a hypo-fibrinolytic state possibly contributing to the onset of symptomatic thrombosis. The latter is not only described for endogenous elevated testosterone levels but also in patients receiving testosterone substitution therapy [25, 26].
As recently evaluated in a large international cohort of children and adolescents with VTE leading to thromboembolic and vascular stroke, the presence of more than one IT is an important risk factor for recurrent VTE [27]. Carriers of the F5 mutation showed a further risk increase in patients additionally carrying the F2 GG/GA variant (rs3136516) with the Odds ratio of 3.34 compared to 1.86 found in F5 carriers alone.
This study has several limitations: apart from (i) selection bias, i.e. age at onset, exclusion of subjects with central line- or malignancy-associated VTE, the non-enrollment of patients with antiphospholipid syndrome, limitations of the present study include (ii) the patient population ascertained with subjects of white race only. Thus, the results presented here cannot be directly transferred to other races, other age groups, to patients with VTE suffering from basic diseases not included in our cohort, such as malignancies. (iii) Continuously admitted patients were ascertained from 2008 onwards and were referred as in- and outpatients within a critical period in which improvement of imaging methods and clinical practice was noted [15, 16]. Results obtained at first thrombotic onset (case–control setting), however, are independent from change in imaging methods or clinical practice and the distribution of F5 rs605 GA/AA and F2 rs3136516 GA/AA is in line with previous reports: thus referral bias caused by inclusion of in- and outpatients seems to be unlikely. Since in- and outpatients were followed according to a similar study protocol a possible referral bias did not play a role with respect to recurrent VTE. It has to be mentioned here that the introduction and uptake of new therapeutic approaches such as DOACs might have influenced referral and treatment patterns. (iv) Although we observed a large number of patients with common mild IT risk factors this study might still be too small to show statistically significance: Although our findings in a clinical setting are in agreement with previously reported population based studies [8, 9], higher patient numbers are needed to rule out statistical errors. (v) Finally, we may have missed recurrent thrombosis in the patients investigated: whereas we may have overlooked asymptomatic VTE recurrence it is unlikely, however, that we would have missed symptomatic recurrences. This statement is underlined by the fact that the recurrence rates detected in this investigation are within the range reported by others [28, 29].
In conclusion in Caucasian adolescent and adult patients with a first VTE onset, the F2 GG/GA genotypes (rs3136516) contribute to the first onset of the disease. Findings of this study gave evidence that in patients with provoked VTEs (i) male gender and (ii) combined ITs, including the F2 mutation at rs3136516 may be regarded as important risk factors for VTE recurrence. These should be included in decision-making in regard to length of AC treatment. Whether (iii) blood group non-O should be included into a predictive scoring system, however, is part of an ongoing prospective study.
Change history
15 April 2023
A Correction to this paper has been published: https://doi.org/10.1007/s11239-023-02809-7
References
Rosendaal FR (1999) Venous thrombosis: a multicausal disease. Lancet 353:1167–1173. https://doi.org/10.1016/S0140-6736(98)10266-0
Dahlbäck B (2000) Blood coagulation. Lancet 355:1627–1632. https://doi.org/10.1016/S0140-6736(00)02225-X
Martinelli I (2001) Risk factors in venous thromboembolism. Thromb Haemost 86:395–403
Bertina RM, Koeleman BP, Koster T et al (1994) Mutation in blood coagulation factor V associated with resistance to activated protein C. Nature 369:64–67. https://doi.org/10.1038/369064a0
Poort SR, Rosendaal FR, Reitsma PH, Bertina RM (1996) A common genetic variation in the 3′-untranslated region of the prothrombin gene is associated with elevated plasma prothrombin levels and an increase in venous thrombosis. Blood 88:3698–3703
Ceelie H, Bertina RM, van Hylckama VA et al (2001) Polymorphisms in the prothrombin gene and their association with plasma prothrombin levels. Thromb Haemost 85:1066–1070
Pérez-Ceballos E, Corral J, Alberca I et al (2002) Prothrombin A19911G and G20210A polymorphisms’ role in thrombosis. Br J Haematol 118:610–614. https://doi.org/10.1046/j.1365-2141.2002.03624.x
Martinelli I, Battaglioli T, Tosetto A et al (2006) Prothrombin A19911G polymorphism and the risk of venous thromboembolism. J Thromb Haemost 4:2582–2586. https://doi.org/10.1111/j.1538-7836.2006.02216.x
Chinthammitr Y, Vos HL, Rosendaal FR, Doggen CJM (2006) The association of prothrombin A19911G polymorphism with plasma prothrombin activity and venous thrombosis: results of the MEGA study, a large population-based case-control study. J Thromb Haemost 4:2587–2592. https://doi.org/10.1111/j.1538-7836.2006.02257.x
Aradjanski M, Djordjevic V, Pruner I et al (2014) The 3′ end prothrombin gene variants in Serbian patients with idiopathic thrombophilia. Balkan J Med Genet 17:43–48. https://doi.org/10.2478/bjmg-2014-0073
Soria JM, Almasy L, Souto JC et al (2000) Linkage analysis demonstrates that the prothrombin G20210A mutation jointly influences plasma prothrombin levels and risk of thrombosis. Blood 95(9):2780–2785
Holzhauer S, Goldenberg NA, Junker R et al (2012) Inherited thrombophilia in children with venous thromboembolism and the familial risk of thromboembolism: an observational study. Blood 120:1510–1515. https://doi.org/10.1182/blood-2012-01-405514
Anderson FA, Wheeler HB, Goldberg RJ et al (1991) A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism: The Worcester DVT study. Arch Intern Med 151:933–938. https://doi.org/10.1001/archinte.1991.00400050081016
Nowak-Göttl U, Limperger V, Kenet G et al (2017) Developmental hemostasis: a lifespan from neonates and pregnancy to the young and elderly adult in a European white population. Blood Cells Mol Dis. https://doi.org/10.1016/j.bcmd.2016.11.012
Kearon C, Akl EA, Ornelas J et al (2016) Antithrombotic Therapy for VTE Disease. Chest 149:315–352. https://doi.org/10.1016/j.chest.2015.11.026
Monagle P, Chan AKC, Goldenberg NA et al (2012) Antithrombotic therapy in neonates and children: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141:e737S–801S. https://doi.org/10.1378/chest.11-2308
Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (2015) S3 Leitlinie Prophylaxe der venösen Thrombembolie (VTE). 1–238
Machin D, Campbell MJ, Tan SB, Tan SH (2008) Sample size tables for clinical studies. Wiley-Blackwell, Oxford
Therneau TM, Grambsch PM (2000) Modeling survival data: extending the Cox model. Springer, New York
Lindström S, Wang L, Smith EN et al (2019) Genomic and transcriptomic association studies identify 16 novel susceptibility loci for venous thromboembolism. Blood 134:1645–1657. https://doi.org/10.1182/blood.2019000435
Franchini M, Mannucci PM (2014) ABO blood group and thrombotic vascular disease. Thromb Haemost 112:1103–1109. https://doi.org/10.1160/th14-05-0457
Douketis J, Tosetto A, Marcucci M et al (2011) Risk of recurrence after venous thromboembolism in men and women: patient level meta-analysis. BMJ 342:d813. https://doi.org/10.1136/bmj.d813
McRae S, Tran H, Schulman S et al (2006) Effect of patient’s sex on risk of recurrent venous thromboembolism: a meta-analysis. Lancet 368:371–378. https://doi.org/10.1016/S0140-6736(06)69110-1
Kearon C, Parpia S, Spencer FA et al (2019) Long-term risk of recurrence in patients with a first unprovoked venous thromboembolism managed according to d-dimer results; A cohort study. J Thromb Haemost 17:1144–1152. https://doi.org/10.1111/jth.14458
Normann S, de Veber G, Fobker M et al (2009) Role of endogenous testosterone concentration in pediatric stroke. Ann Neurol 66:754–758. https://doi.org/10.1002/ana.21840
Freedman J, Glueck CJ, Prince M et al (2015) Testosterone, thrombophilia, thrombosis. Transl Res 165:537–548. https://doi.org/10.1016/j.trsl.2014.12.003
DeVeber G, Kirkham F, Shannon KK et al (2019) Recurrent stroke: the role of thrombophilia in a large international pediatric stroke population. Haematologica. https://doi.org/10.3324/haematol.2018.211433
Vossen CY, Walker ID, Svensson P et al (2005) Recurrence rate after a first venous thrombosis in patients with familial thrombophilia. Arterioscler Thromb Vasc Biol 25:1992–1997. https://doi.org/10.1161/01.ATV.0000174806.76629.7b
Heit JA (2012) Predicting the risk of venous thromboembolism recurrence. Am J Hematol 87:S63. https://doi.org/10.1002/ajh.23128
Acknowledgements
Open Access funding provided by Projekt DEAL.
Funding
The study was supported by grants from the Förderverein “Schlaganfall und Thrombosen im Kindesalter e.V.”, Interdisziplinäres Zentrum für Klinische Forschung (IZKF: CRA01-09: University of Münster) and the “Gesellschaft für Thrombose und Hämostaseforschung/ Society of Thrombosis and Hemostasis Research” [GTH (Grant 2016)]. None of the aforementioned funding agencies had a role in study design, conduct, interpretation, or reporting.
Author information
Authors and Affiliations
Contributions
U.N.G., B.K. and V.L. designed the study and analyzed the data. V.L., B.K., R.J., B. and U.N.G. wrote the paper, G.K., M.K., M.S., F.L., D.J., M.Sch., A.F., U.C.K., R.M., F.R., M.S., D.S., D.K., A.R., P.K., T.B., A.T. and B.N. had full access to the data and took part in the design, execution and data analysis, discussion, and in writing the report. The manuscript has been read and approved for submission to the Journal of Thrombosis and Haemostasis by all authors.
Corresponding authors
Ethics declarations
Conflict of interest
Verena Limperger: personal fees from Bristol-Myers Squibb, Amedes holding GmbH, CSL Behring, NovoNordisk, non-financial support from Octapharma, NovoNordisk, outside the submitted work. Gili Kenet: personal fees from Bayer, personal fees from Boehringer Ingelheim, personal fees from Daiichi Sankyo, other from Pfizer, outside the submitted work. Bettina Kiesau: personal fees and other from Sobi, outside the submitted work. Max Köther, Malin Schmeiser, David Juhl, Andre Franke, Ulrich K. Klostermeier, Rolf Mesters, Frank Rühle, Monika Stoll, Dagmar Steppat, Antje Torge, Bruno Neuner, and Ralf Junker declared that they have no conflict of interest. Florian Langer: personal fees from Bayer, personal fees from Bristol-Myers Squibb, personal fees from Daiichi Sankyo, personal fees from LEO Pharma, grants and personal fees from Pfizer, personal fees from Chugai, personal fees from Roche, personal fees from SOBI, personal fees from Takeda, personal fees from CSL Behring, personal fees from SOBI, personal fees from Werfen, personal fees from Siemens, personal fees from Grifols, personal fees from Novo Nordisk, personal fees from Octapharma, personal fees from Ablynx, outside the submitted work. Maria Shneyder: personal fees from NovoNordisk, Octapharma and Takeda, outside the submitted work. Dorothee Kowalski: non-financial support from Octapharma, NovoNordisk and LFB GmbH outside the submitted work. Angela Rocke: personal fees from Octapharm, CSL Behring outside the submitted work. Piotr Kuta: personal fees from CSL Behring, NovoNordisk, Octapharma, Stago, Aspen outside the submitted work. Tido Bajorat: personal fees from BFSH e.V. (Bündnis zur Förderung der Sicherheit von Hämophilen), personal fees from Octapharma, outside the submitted work. Ulrike Nowak-Göttl: grants from Octapharma, grants from LFB, other from Bayer Vital, other from Boehringer-Ingelheim, outside the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Limperger, V., Kenet, G., Kiesau, B. et al. Role of prothrombin 19911 A>G polymorphism, blood group and male gender in patients with venous thromboembolism: Results of a German cohort study. J Thromb Thrombolysis 51, 494–501 (2021). https://doi.org/10.1007/s11239-020-02169-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-020-02169-6