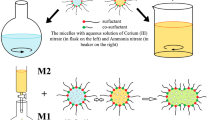
A series of CeO2 nanoparticles was synthesized by precipitation in reverse microemulsions in the presence of mixed aqueous–alcoholic solutions at variable and constant medium pH. It is shown that these methods promote the formation of non-agglomerated CeO2 particles with a size less than 15 nm. TEM and XPS methods showed that the ratio of Ce3+ /Ce4+ ions increases on the surface when the size of CeO2 nanoparticles is decreased. The synthesized CeO2 nanoparticles show high stability in aqueous suspensions without using additional stabilizers (𝜁 > +35 mV), which is important for medical and biological application.


Similar content being viewed by others
References
L. Junheng, Y. Jun, S. Ping, et al., Energy, 161, 17-27 (2018).
S. Putla, M. H. Amin, B. M. Reddy, et al., ACS Appl. Mater. Interfaces, 7, 16525-16535 (2015).
S. B. Khan, M. Faisal, M. M. Rahman, and A. Jama, Sci. Total Environ., 409, 2987-2992 (2011).
B. Choudhury, P. Chetri, and A. Choudhury, J. Exp. Nanosci., 10, 103-114 (2015).
A. Gupta, S. Das, C. J. Nealaand, and S. Seal, J. Mater. Chem. B, 4, 3195-3202 (2016).
A. Y. Estevez and J. S. Erlichman, Nanomedicine, 9, 1437-1440 (2014).
A. Datta, S. Mishra, K. Manna, et al., ACS Omega, 5, 9714-9723 (2020).
A. L. Popov, S. I. Zaichkina, N. R. Popova, et al., RSC Adv., 6, 106141-106149 (2016).
D. L. Trimm, The Design of Industrial Catalysts, Elsevier Scientific Pub. Co., Amsterdam (1980).
J. Mingxia, Z. Peisen, M. Junli, et al., Biomater. Sci., 6, 726-745 (2018).
Z. Ji, X. Wang, H. Zhang, et al., ACS Nano., 6, 5366-5380 (2012).
M. Sendraa, M. Vollanda, T. Balbib, et al., Aquat. Toxicol., 200, 13-20 (2018).
I. Celardo, J. Z. Pedersen, E. Traversa, and L. Ghibelli, Nanoscale, 3, 1411-1420 (2011).
J. Malleshappa, H. Nagabhushana, B. Daruka, et al., Optik (Stuttg.), 127, 855-861 (2016).
N. S. Ferreira, R. S. Angelica, V. B. Marques, et al., Mater. Lett., 165, 139-142 (2016).
Y. Shlapa, V. Sarnatskaya, I. Timashkov, et al., Appl. Phys. A, 125, 412-422 (2019).
L. Yin, Y. Wang, G. Pang, et al., J. Colloid Interf. Sci., 246, 78-84 (2002).
A. Khorsand Zak, W. H. A. Majid, M. E. Abrishami, and R. Yousefi, Solid State Sci., 13, 251-256 (2011).
F. Zhang, P. Wang, J. Koberstein, et al., Surf. Sci., 563, Nos. 1-3, 74-82 (2004).
T. S. Sreeremya, K. M. Thulasi, A. Krishnan, and S. Ghosh, Ind. Eng. Chem. Res., 51, No. 1, 318-326 (2012).
S. Deshpande, S. Patil, S.V.N.T. Kuchibhatla, and S. Seal, Appl. Phys., 87, 133113 (2005).
K. Siposova, V. Huntosova, Yu. Shlapa, et al., ACS Appl. Bio Mater., 2, No. 5, 1884-1896 (2019).
The work was performed within the framework of the project of the Target Program of Scientific Research of the NAS of Ukraine “Grants of the NAS of Ukraine for research laboratories/groups of young scientists of the NAS of Ukraine for research in priority areas of science and technology” No. 0121U110363 (2021-2022). The authors express their gratitude to the CERIC-ERIC consortium for access to the equipment and opportunity to conduct XPS measurements under grant No. 20197153. The authors also express ratitude to M. S. Kharchuk and the Center for Collective Use of D. K. Zabolotny Institute of Microbiology and Virology of the NAS of Ukraine for the opportunity to conduct research by electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Teoretychna ta Eksperymentalna Khimiya, Vol. 57, No. 4, pp. 241-246, July-August, 2021.
Rights and permissions
About this article
Cite this article
Timashkov, I.P., Shlapa, Y., Veltruska, K. et al. Physical-Chemical Properties of Nanosized Cerium Dioxide Synthesized via Different Methods for Biomedical Application. Theor Exp Chem 57, 282–288 (2021). https://doi.org/10.1007/s11237-021-09696-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11237-021-09696-0