Abstract
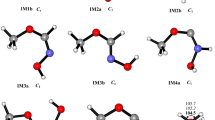
The oxidation mechanism of CH3CHCO initiated by OH is systematically studied using quantum theoretical methods. According to thermodynamic research, the dominant channel is the addition of CH3CHCO with OH generating C-IM1 (CH3CHOHCO) and C-IM2 (CH3CHCOOH), and then dissociate to the dominant products P1 (CH3CHO + CO) from C-IM1 with the lowest barrier. The rate constants for CH3CHCO + OH → C-IM1 → P1/CH3CHCO + OH → C-IM2 channels are computed through RRKM theory at 200–2000 K. The obtained overall rate constant at 298 K (7.60 × 10−11 cm3 molecule−1 s−1) is well consistent with the reported experimental value. The atmospheric lifetime of CH3CHCO is estimated to be around 1.83 h.






Similar content being viewed by others
Code availability
Gaussian 09 package program.
References
Atkinson R (1986) Kinetics and mechanisms of the gas-phase reactions of the hydroxyl radical with organic compounds under atmospheric conditions. Chem Rev 86:69–201
Atkinson R, Pitts JN Jr (1975) Rate constants for the reaction of OH radicals with propylene and the butenes over the temperature range 297–425° K. J Chem Phys 63:3591–3595
Atkinson R, Perry RA, Pitts JN Jr (1977) Absolute rate constants for the reaction of OH radicals with allene, 1,3-butadiene, and 3-methyl-1-butene over the temperature range 299–424 °K. J Chem Phys 67:3170–3174
Howard CJ (1976) Rate constants for the gas–phase reactions of OH radicals with ethylene and halogenated ethylene compounds. J Chem Phys 65:4771–4777
Finlayson-Pitts BJ, Pitts JN Jr (1986) Atmospheric chemistry. Wiley, New York
Nicholas J (1976) Chemical kinetics Harper and Row London
Blake GA, Sutton EC, Masson CR, Phelps TG (1987) Molecular abundances in OMC-1-The chemical composition of interstellar molecular clouds and the influence of massive star formation. Astrophys J 315:621–645
Turner BE (1977) Microwave detection of interstellar ketene. Astrophys J 213:L75–L79
Frank P, Bhaskaran KA, Just Th (1986) High-temperature reactions of triplet methylene and ketene with radicals. J Phys Chem 90:3809–3814
Sun H, Tang YZ, Wang ZL, Pan XM, Li ZS, Wang RS (2005) DFT study on the mechanisms of the CH2CO + NCX (X = O, S) reactions. J Mol Struct Theochem 757:143–148
Washida N, Hatakeyama S, Takagi H, Kyogoku T, Sato S (1983) Reaction of ketenes with atomic oxygen. J Chem Phys 78:4533–4540
Gaffney JS, Atkinson R, Pitts JN Jr (1975) Relative rate constants for the reaction of O(3P) atoms with selected olefins, monoterpenes, and unsaturated aldehydes. J Am Chem Soc 97:6481–6483
Mack GPR, Thrush BA (1974) Reaction of oxygen atoms with carbonyl compounds Part 3. -Ketene. J Chem Soc Faraday Trans 1 70:1974
Zhou ZY, Fu H, Zhou XM, Cheng XL (2003) Mechanistic investigation on the multi-channel reaction of Cl + CH2CO. J Mol Struct THEOCHEM 620:207–214
Wallington TJ, Ball JC, Straccia AM, Hurley MD, Kaiser EW, Dill M, Schneider WF (1996) Kinetics and mechanism of the reaction of Cl atoms with CH2CO (ketene). Int J Chem Kinet 28:627–635
Maricq MM, Ball JC, Straccia AM, Szente JJ (1997) A diode laser study of the Cl + CH3CO reaction. Int J Chem Kinet 29:421–429
Hou H, Wang BS, Gu Y (2000) Ab initio mechanism and multichannel RRKM-TST rate constant for the reaction of Cl(2P) with CH2CO (ketene). J Phys Chem A 104:320–328
Grussdorf J, Nolte J, Temps F, Wagner HGg, (1994) Primary products of the elementary reactions of CH2CO with F, Cl, and OH in the gas phase Ber. Bunsenges Phys Chem 98:546–553
Cheng XL, Zhao YY, Zhou XM, Zhou ZY (2003) Reaction mechanism for the F+CH2CO reaction system based on density functional theory and vibrational mode analysis. J Mol Struct THEOCHEM 638:27–35
Lee J, Bozzelli JW (2003) Reaction of H plus ketene to formyl methyl and acetyl radicals and reverse dissociations. Int J Chem Kinet 35:20–44
Michael JV, Nava DF, Payne WA, Stief LJ (1979) Absolute rate constants for the reaction of atomic hydrogen with ketene from 298 to 500 K. J Chem Phys 70:5222–5227
Slemr F, Warneck P (1975) Reactions of atomic hydrogen with ketene and acetaldehyde. Ber Bunsenges Phys Chem 79:152–156
Hatakeyama S, Honda S, Akimoto H (1985) Reactions of ketene, methylketene, ethylketene, and dimethylketene with ozone in air. Bull Chem Soc Jpn 58:2411–2412
Oehlers C, Temps F, Gg WH, Wolf M (1992) Kinetics of the reaction of OH radicals with CH2CO. Ber Bunsenges Phys Chem 96:171–175
Brown AC, Canosa-Mas CE, Parr AD, Wayne RP (1989) Temperature dependence of the rate of the reaction between the OH radical and ketene. Chem Phys Lett 161:491–496
Semenikhin AS, Shubina EG, Savchenkova AS, Chechet IV, Matveev SG, Konnov AA, Mebel AM (2018) Mechanism and rate constants of the CH3 + CH2CO reaction: a theoretical study. Int J Chem Kinet 50:273–284
Sun H, He HQ, Hong B, Chang YF, An Z, Wang RS (2006) Theoretical study of the mechanism of CH2CO + CN reaction. Int J Quant Chem 106:894–905
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian, Inc. Wallingford CT
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functional. Theor Chem Account 120:215–241
Zhao Y, Truhlar DG (2008) Density functionals with broad applicability in chemistry. Acc Chem Res 41:157–167
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys 72:650–654
Clark T, Chandrasekhar J, Spitznagel GW, Schleyer PVR (1983) Efficient diffuse function-augmented basis sets for anion calculations. III. The 3–21+G basis set for first-row elements, Li–F. J Comp Chem 14:294–301
Gonzalez C, Schlegel HB (1989) An improved algorithm for reaction path following. J Chem Phys 90:2154–2161
Gonzalez C, Schlegel HB (1990) Reaction path following in mass-weighted internal coordinates. J Phys Chem 94:5523
Raghavachari K, Trucks GW, Pople JA, Head-Gordon M (1989) A fifth-order perturbation comparison of electron correlation theories. Chem Phys Lett 157:479–483
Dunning TH Jr (1989) Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J Chem Phys 90:1007–1023
Peiró-García J, Nebot-Gil I (2003) Ab initio study on the mechanism of the atmospheric reaction OH + O3 → HO2 + O2. Chem Phys Chem 4:843–847
Peiró-García J, Nebot-Gil I (2003) Ab initio study of the mechanism of the atmospheric reaction: NO2 + O3 → NO3 + O2. J Comput Chem 24:1657–1663
Rienstra-Kiracofe JC, Allen WD, Schaefer HF (2000) The C2H5 + O2 reaction mechanism: high-level ab initio characterizations. J Phys Chem A 104:9823–9840
Holbrook KA, Pilling MJ, Robertson SH (1996) Unimolecular reactions; J. Wiley, Chichester, UK
Smith IWM (1980) Kinetics and dynamics of elementary gas reactions. Butterworth, London, p 118
Johnston HS, Heicklen J (1962) Tunnelling corrections for unsymmetrical Eckart potential energy barriers. J Phys Chem 66:532–533
Eckart C (1930) The penetration of a potential barrier by electrons. Phys Rev 35:1303–1309
Bai FY, Tang NS, YZ, Pan XM, Zhao Z (2019) New insights into 3M3M1B: roles of water on the •OH-initiated degradation and aerosol formation in the presence of NOX (X=1, 2) and alkali. Phys Chem Chem Phys 21:17378–17392
Funding
This work was supported by the Natural Science Foundations of China (No. 21707062), Scientific Research Starting Foundation of Mianyang Normal University (No. QD2016A007) and by the Open Project Program of Beijing Key Laboratory of Flavor Chemistry, Beijing Technology and Business University (BTBU), Beijing 100048, China.
Author information
Authors and Affiliations
Contributions
Yongguo Liu, Huaming Du, Meilian Zhao, Yuxi Sun, Huirong Li Zhiguo Wang: calculation, data curation, formal analysis, investigation. Yunju Zhang: calculation, writing-review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, Y., Liu, Y., Du, H. et al. A theoretical study on gas-phase reaction of methylketene with OH: mechanism, kinetics, and insights. Struct Chem 33, 49–56 (2022). https://doi.org/10.1007/s11224-021-01811-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-021-01811-0