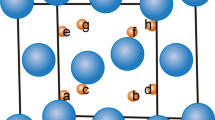
Abstract
LiK(BH4)2, with a 10.65 wt% H2 capacity, is a promising hydrogen storage candidate material. We describe all the possible metastable paths for three reactant mixtures associated with LiK(BH4)2, using first-principles thermodynamic calculations. However, with the first-principles calculations inherent disadvantage is absent from the vibrational entropy contribution to the prediction of the reaction mechanism, which indicates that the reaction mechanism of the reaction path prediction may not be the most stable. The study focuses on examining the possible metastable paths for the three reactants, using first-principles calculations. The objective of this study is to find the minimum free energy path among all the possible paths of the three reactants mixture. A clear conclusion is that the minimum free energy path predicted from first principles thermodynamic calculations is the most stable reaction paths at an appropriate H2 pressure range for all cases. An additional aim of paper is to assess whether the predicted mechanisms can be unambiguously described under the DFT uncertainty.




Similar content being viewed by others
References
Menedéz E, Garroni S, Lόpez-Ortega A, Estrader M, Liedke MO, Fassbender J, Solsona P, Surinach S, Baro MD, Nogués J (2010) J Phys Chem C 114:16818–16822
Schlapbach L, Züttel A (2001) Nature 414:353–358
Eberle U, Felderhoff M, Schuth F (2009) Angew Chem Int Ed 48:6608–6630
Grochala W, Edwards PP (2004) Chem Rev 104:1823
Soloveichik G, Her J-H, Stephens PW, Gao Y, Rijssenbeek J, Andrus M, Zhao JC (2008) Inorg Chem 47:4290–4298
Vajo JJ, Skeith SL, Mertens F (2005) J Phys Chem B 109:3719–3722
Vittetoe AW, Niemann MU, Srinivasan SS, McGrath K, Kumar A, Goswami DY, Stefanakos EK, Thomas S (2009) Int J Hydrogen Energy 34:2333–2339
Akbarzadeh AR, Ozolins V, Wolverton C (2007) Adv Mater 19:3233–3239
Nichkel EA, Jones NO, David WIF, Johnson SR, Lowton RL, Sommariva M, Edwards PP (2008) Angew Chem Int Ed 47:2817–2819
Xiao XB, Yu WY, Tang BY (2008) J Phys 20:445210–445216
Kim KC, Sholl DS (2010) J Phys Chem C 114:678–686
Kim KC, Kulkarni AD, Johnson JK, Sholl DS (2011) Phys Chem Chem Phys 13:21520–21529
Kim KC (2012) J Chem Phys 137:08412–084111
Baroni S, Gironcoli SD, Corso AD, Giannozzi P (2001) Rev Mod Phys 73:515–562
Refson K, Clark S, Tulip P (2006) Phys Rev B 73:195109–195115
Johnson JW, Brody JF (1982) J Electrochem Soc 129:2213–2219
Miwa K, Ohba N (2005) Towata S-i. Phys Rev B 71:195109–195115
Garroni S, Milanese C, Pottmaier D, Mulas G, Nolis P, Girella A, Caputo R, Olid D, Teixdor F, Baricco M, Marini A, Surinach S, Baro MD (2011) J Phys Chem C 115:16664–16671
Kresse G (1995) Furthmuller. J Comput Mater Sci 5:15–50
Caputo R, Garroni S, Olid D, Teixidor F, Surinach S, Baro MD (2010) Phys Chem Chem Phys 12:15093–15100
Alapati SV, Johnson JK, Sholl DS (2007) Phys Chem Chem Phys 9:1438–1452
Alapati SV, Johnson JK, Sholl DS (2007) J. Alloys Compd. 446–447:23–27
Alapati SV, Johnson JK, Sholl DS (2007) J Phys Chem C 111:1584–1591
Alapati SV, Johnson JK, Sholl DS (2008) J Phys Chem C 12:5258–5262
Alapati SV, Johnson JK, Sholl DS (2006) J. Phys. Chem. B 110:8769–8776
Ibikunle A, Goudy AJ, Yang H (2009) J Alloys Compd 475:110–115
Guo YJ, Jia JF, Wang XH, Ren Y, Wu HS (2013) Chem Phys 418:22–27
Ozolins V, Majzoub EH, Wolverton C (2009) J Am Chem Soc 131:230–237
Ozolins V, Majzoub EH, Wolverton C (2008) Phys Rev Lett 100:135501–135505
Kulkarni AD, Wang LL, Johnson DD, Sholl DS, Johnson JK (2010) J Phys Chem C 114:14601–14605
Kim KC, Allendorf MD, Stavila V, Sholl DS (2010) Phys Chem Chem Phys 12:9918–9926
Zavorotynska O, Corno M, Damin A, Spoto G, Ugliengo P, Baricco M (2011) J Phys Chem C 15:18890–18900
Acknowledgments
The work is supported by the National Natural Science Foundation of China (No. 21031003 and 21103101), and Key Project of Chinese Ministry of Education (No. 212022).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, YJ., Jia, JF. & Wu, HS. Validation of the reaction thermodynamics paths associated with LiK(BH4)2 by detection of metastable reaction paths from first-principles calculations. Struct Chem 26, 647–653 (2015). https://doi.org/10.1007/s11224-014-0516-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-014-0516-1