Abstract
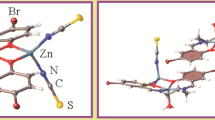
The structures of the chelate Zn(PDTC)2 and its dimeric form Zn2(PDTC)4 are investigated theoretically at B3LYP/cc-pVDZ level. The natural bond orbital (NBO) analysis has been performed to explore the metal–ligand coordination of these chelates. In Zn(PDTC)2, the sulfur atoms mainly use 3p sub-shells to coordinate with mixed (4s + 4p x + 4p y + 4p z ) orbital of zinc having sp 3 hybridization. In Zn2(PDTC)4, each zinc atom coordinates with one terminal and two bridging PDTC ligands. The contribution of bridging sulfur atoms in chelation is much more than terminal sulfurs. The bridging sulfur atoms use 3s and 3p sub-shells to coordinate with 4s and 4p sub-shells of metal center zinc. The charge transfer interactions between sulfur and metal center involving 4d, 5s, and 5p sub-shells of zinc are much feeble compared to those involving 4s and 4p sub-shells of zinc.


Similar content being viewed by others
References
Nieuwenhuizen PJ, Timal S, Haasnoot JG, Spek AL, Reedijk J (1997) Chem Eur J 3:1846
Nieuwenhuizen PJ, Ehlers AW, Haasnoot JG, Janse SR, Reedijk J, Baerends EJ (1999) J Am Chem Soc 121:163
Nieuwenhuizen PJ (2001) Appl Catal A 207:55
Debnath SC, Basu DK (2003) J Appl Polym Sci 52:597
Ouyang C, Wang S, Zhang Y, Zhang Y (2006) Polym Degrad Stab 91:795
Kurian JK, Peethambaran NR, Mary KC, Kuriakose B (2000) J Appl Polym Sci 78:304
Oliveira MRL, Perpetuo GJ, Janczak J, Rubinger MMM (2007) Polyhedron 26:163
Nieuwenhuizen PJ, Ehlers AW, Hofstraat JW, Janse SR, Nielen MWF, Reedijk J, Baerends EJ (1998) Chem Eur J 4:1816
Agarwal S, Aggarwal SG, Singh P (2005) Talanta 65:104
Vermeulen LA, Reinecke AJ, Reinecke SA (2001) Ecotoxicol Environ Saf B 48:183
Paz FAA, Neves MC, Trindade T, Klinowski J (2003) Acta Crystallogr E59:m1067
Munoz C, Pascual-Salcedo D, Castellanos MC, Alfranca A, Aragones J, Vara A, Redondo JM, de Landazuri MO (1996) Blood 88:3482
Cvek B, Dvorak Z (2007) Curr Pharm Des 13:3155
Kim CH, Kim JH, Hsu CY, Ahn YS (1999) FEBS Lett 449:28
Kim CH, Kim JH, Lee J, Ahn YS (2003) Toxicol Appl Pharmacol 190:189
Camps M, Boothroyd JC (2001) Exp Parasitol 98:206
Kang MS, Choi EK, Choi DH, Ryu SY, Lee HH, Kang HC, Koh JT (2008) FEMS Microbiol Lett 280:250
Choi EK, Lee HH, Kang MS, Kim BG, Lim HS, Kim SM, Kang IC (2010) J Microbiol 48:40
Vesna M, Di C, Lorena G, Alejandro D, Dolores F, Ping DQ (2008) Toxicol Appl Pharmacol 231(1):24
Shahid M, Rüffer T, Lang H, Awan SA, Ahmad S (2009) J Coord Chem 62:440
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, revision B.01. Gaussian, Inc, Pittsburgh
Hoyau S, Ohanessian G (1997) Chem Phys Lett 280:266
Kemper PR, Weis P, Bowers MT, Maitre P (1998) J Am Chem Soc 120:13494
Alcamí M, Luna A, Mó O, Yáñez M, Boutreau L, Tortajada J (2002) J Phys Chem A 106:2641
González L, Mó O, Yáñez M (1997) J Comput Chem 18:1124
González L, Mó O, Yáñez M (1998) J Chem Phys 109:139
González L, Mó O, Yáñez M (1997) J Phys Chem A 101:9710
Becke AD (1993) J Chem Phys 98:5648
Lee C, Yang W, Parr RG (1998) Phys Rev B 37:785
Dunning TH (1989) J Chem Phys 90:1007
Woon DE, Dunning TH (1993) J Chem Phys 98:1358
Reed AE, Weinstock RB, Weinhold F (1985) J Chem Phys 83:735
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghosh, D., Sen, K. & Das, A.K. Structure and coordination in mono and dinuclear Zn(II)-pyrrolidine dithiocarbamate complexes. Struct Chem 23, 227–235 (2012). https://doi.org/10.1007/s11224-011-9863-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-011-9863-3