Abstract
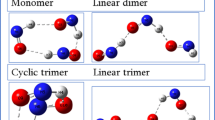
This work reports hydrogen bonding interaction in cyclic and ladder oligomers using density functional theory method. Many-body analysis technique has been used to study the nature of interactions between different molecules and their contribution to the binding energy of a respective hydrogen bonded oligomers. Hydrogen bonds in cyclic trimer to pentamer are stronger than those in corresponding ladder structures. Cyanamide monomer shows the lowest energy at B3LYP/aug-cc-pvdz level among different methods used here with the same basis set. The geometrical parameters for cyanamide monomer obtained at B3LYP/aug-cc-pvdz level are in excellent agreement with the experimental determinations. Cyclic structures are more stable than the ladder. In cyclic oligomers not only total two-body energies, but higher body energies also contribute significantly to the binding energy of a respective complex whereas in ladder, only total two-body energies contribute significantly and higher-body energies are almost negligible for cyanamide trimer to pentamer.




Similar content being viewed by others
References
Scheiner S (1997) Hydrogen bonding: a theoretical perspective. Oxford University Press, Oxford
Jeffrey GA (1997) An introduction to hydrogen bonding. Oxford University Press, New York
Desiraju GR, Steiner T (1999) The weak hydrogen bond in structural chemistry and biology. Oxford University Press, Oxford
Xantheas SS (1994) J Chem Phys 100:7523–7534
Xantheas SS (2000) Chem Phys 258:225–231
Xantheas SS, Dunning TH Jr (1993) J Chem Phys 99:8774–8792
Kulkarni A, Ganesh V, Gadre SR (2004) J Chem Phys 121:5043–5050
Chaudhari A, Lee SL (2004) J Chem Phys 120:7464–7469
Chaudhari A, Sahu PK, Lee SL (2004) J Chem Phys 120:170–174
Boo DW (2001) Bull Korean Chem Soc 22:693–698
Chaudhari A, Meraj G, Lee SL (2010) J Mol Mod 16:1559–1566
Milet A, Moszynski R, Wormer PES, van der Avoird AJ (1999) J Phys Chem A 103:6811–6819
Chaudhari A, Lee SL (2010) J Theor Comput Chem 9:177–187
Chaudhari A, Sahu PK, Lee SL (2005) Int J Quant Chem 101:67–72
Chaudhari A, Sahu PK, Lee SL (2004) J Mol Struc Theochem 683:115–119
Chaudhari A, Lee SL (2005) Chem Phys 310:281–285
Chaudhari A, Lee SL (2005) Int J Quant Chem 102:174–177
Mierzwicki K, Latajka Z (2000) Chem Phys Lett 325:465–472
Chaudhari A, Lee SL (2005) Int J Quant Chem 102:106–111
Chaudhari A (2010) Int J Quant Chem 110:1092–1099
Chaudhari A, Naganathappa M, Shinde MN, Kumbharkhane AC (2011) Int J Quant Chem 111:2972–2979
Brunsveld L, Folmer BJB, Meijer EW, Sijbesma RP (2001) Chem Rev 101:4071–4098
Ilhan F, Galow TH, Gray M, Clavier G, Rotello VM (2000) J Am Chem Soc 122:5895–5896
Pourcain C, Griffin AC (1995) Macromolecules 28:4116–4121
Vera F, Almuzar C, Orera I, Barbera J, Oriol L, Serrano JL, Sierra T (2008) J Polym Sci Part A 46:5528–5541
Ruokolainen J, Torkkeli M, Serimaa R, Komanschek E, Brinke GT, Ikkala O (1997) Macromolecules 30:2002–2007
Sheen YC, Lu CH, Huang CF, Kuo SW, Chang FC (2008) Polymer 49:4017–4024
Ruokolainen J, Saariaho M, Ikkala O, Brinke GT, Thomas EL, Torkkeli M, Serimaa R (1999) Macromolecules 32:1152–1158
Wu X, Zhang G, Zhang H (1998) Macromol Chem Phys 199:2101–2105
Bladon P, Griffin AC (1993) Macromolecules 26:6604–6610
Bouteiller L (2007) Adv Polym Sci 27:79–112
Shih RS, Lu CH, Kuo SW, Chang FC (2010) J Phys Chem C 114:2855–2862
Kriz J, Dybal J, Brus J (2006) J Phys Chem B 110:18338–18346
Kriz J, Dybal J (2005) J Phys Chem B 109:13436–13444
Kuo SW, Tung PH, Chang FC (2006) Macromolecules 39:9388–9395
Kuo SW, Tung PH, Lia CL, Jeoing KU, Chang FC (2008) Macromol Rapid Commun 29:229–233
Kuo SW, Tung PH, Chang FC (2009) Eur Polym J 45:1924–1934
Rakotondradany F, Whitehead MA, Leburs AM, Sleiman HF (2003) Chem Eur J 9:4771–4780
Berl V, Huc I, Khoury RG, Krische MJ, Lehn JM (2000) Nature 407:720–723
Berl V, Schmutz M, Krische MJ, Khoury RG, Lehn JM (2002) Chem Eur J 8:1227–1244
Lagowski JB (2002) J Mol Struct 589–590:125–137
Hassan S, Mohammmad RN (2003) J Mol Struct 626:143–158
Gambi A, Giumanini AG, Strazzolini P (2001) J Mol Struct 536:9–16
Malysheva L, Klymenko Y, Onipko A, Valiokas R, Licedber B (2003) Chem Phys Lett 370:451–459
Balbas A, Gonzales Tejera MJ, Tortajada J (2001) J Mol Struct 572:141–150
Groves C, Lewars E (2000) J Mol Struct 530:265–279
Pires MM, DeTuri VF (2007) J Chem Theor Comput 3:1073–1082
Wu H, Chaudhari A, Lee SL (2005) J Comput Chem 26:1543–1564
Sengwa RJ, Kaur K, Chaudhari R (2000) Polym Int 49:599–608
Sengwa RJ, Kaur K (2000) Polym Int 49:1314–1320
Sengwa RJ (2004) Polym Int 53:744–748
Sengwa RJ (1998) Polym Int 45:43–46
Sengwa RJ, Chaudhari R (2001) Polym Int 50:433–441
Greenwood NN, Earnshaw A (1984) Chemistry of the elements. Pergamon, Oxford, p. 341
Moffat JB, Vogt C (1970) J Mol Spectrosc 33:494–499
Riggs NV, Radom L (1985) Aust J Chem 38:835–840
Saebo S, Farnell L, Riggs NV, Radom L (1984) J Am Chem Soc 106:5047–5051
Thompson C, Glidewell C (1983) J Comput Chem 4:1–8
Vincent MA, Dykstra CE (1980) J Chem Phys 73:3838–3842
Ichikawa K, Hamada Y, Sugawara Y, Tsuboi M, Kato S, Morokuma K (1982) Chem Phys 72:301–312
Daoudi A, Pouchan C, Sauvaitre H (1982) Chem Phys Lett 91:477–483
Helgaker TU, Klewe B (1988) Acta Chem Scand A 42:269–272
Kapellos ST, Mavridis A (1993) J Mol Struct 279:151–156
Denner L, Luger P, Buschmann J (1988) Acta Crystallogr C44:1979–1981
Brown RD, Godfrey PD, Kleibomer B (1985) J Mol Spectrosc 114:257–273
Birk M, Winnewisser M (1986) Chem Phys Lett 123:382–385
Birk M, Winnewisser M (1993) J Mol Spectrosc 159:69–78
Kwon CH, Lee JH, Kim HL (2007) Bull Korean Chem Soc 28:1485–1488
Lee JH, Kang TY, Hwang H, Kwon CH, Kim HL (2008) Bull Korean Chem Soc 29:1685–1688
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Valiron P, Mayer I (1997) Chem Phys Lett 275:46–55
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, MontgomeryJA Jr, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES and Pople JA, (2003) Gaussian03. Gaussian Inc., Pittsburgh
Tyler JK, Sheridan J, Costain CC (1972) J Mol Spectrosc 43:248–261
Brown RD, Godfrey PD, Kleibomer B (1985) J Mol Spectrosc 14:257–273
Fletcher WH, Brown FB (1963) J Chem Phys 39:10
Hector R (1956) The Infrared spectra and structure of cyanamide (part I). The infrared spectra of formamide, N,N-dideuteroformamide and N-methylformamide (Part II), Ph.D. Thesis, California institute of Technology Pasadena, California
Bernstein MP, Bauschlicher CW Jr, Sandford SA (2004) Adv Space Res 33:40–43
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kharat, B., Deshmukh, V. & Chaudhari, A. Cyclic and ladder hydrogen bonded cyanamide oligomers: a density functional theory and many-body analysis approach. Struct Chem 23, 37–45 (2012). https://doi.org/10.1007/s11224-011-9841-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-011-9841-9