Abstract
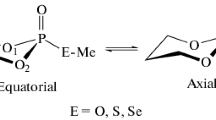
Different orientations of P(O) versus C(O) in P(O)NHC(O) skeleton have been discussed in two new phosphorus(V)-nitrogen compounds with formula XP(O)Y and XP(O)Z2 where X = NHC(O)C6H4(4-F) and Y = NHCH2C(CH3)2CH2NH (1), Z = NHC6H4(4-CH3) (2). Compound 1 is the first example of an aliphatic diazaphosphorinane with a gauche orientation which has been studied by X-ray crystallography; the P=O bond is in the equatorial position of the ring. Both compounds show n J(F,C) and m J(F,H) coupling constants (n = 1, 2, 3 and 4; m = 3 and 4) and 3 J(P,C) > 2 J(P,C). Quantum chemical calculations were performed with HF and Density Functional Theory (DFT) methods using 6−31+G(d,p) basis set. A tentative assignment of the observed vibrational bands for these molecules is discussed. Compound 1 shows a deshielded C atom of the carbonyl moiety (in 13C NMR spectrum) relative to that of 2, which is supported by IR spectroscopy in which the considerably lower C=O frequency is observed for 1. Comparing the X-ray crystallography and IR spectra of 1 and 2 shows that the acyclic compound 2, containing P=O and C=O bonds in an anti position, are involving in a stronger N–H···O=P hydrogen bond in crystal network. This leads to a weaker P=O and NC(O)NHP(O)–H bonds and stronger N···O interaction. The Namide–H is involved in an intramolecular N–H···O hydrogen bond.







Similar content being viewed by others
References
Gholivand K, Pourayoubi M (2004) Z Anorg Allg Chem 630:1330
Gholivand K, Shariatinia Z, Ansar S, Mashhadi SM, Daeepour F (2009) Struct Chem 20:481
Gubina KE, Shatrava JA, Ovchynnikov VA, Amirkhanov VM (2000) Polyhedron 19:2203
Gholivand K, Mahzouni HR, Pourayoubi M, Amiri S (2010) Inorg Chim Acta 363:2318
Gholivand K, Mostaanzadeh H, Koval T, Dusek M, Erben MF, Stoeckli-Evansd H, Della Védova CO (2010) Acta Crystallogr B 66:1
Sabbaghi F, Pourayoubi M, Toghraee M, Divjakovic V (2010) Acta Crystallogr E 66:o344
Pourayoubi M, Sabbaghi F (2009) J Chem Crystallogr 39:874
Yazdanbakhsh M, Eshtiagh-Hosseini H, Sabbaghi F (2009) Acta Crystallogr E 65:o78
Trush EA, Amirkhanov VM, Ovchynnikov VA, Swiatek-Kozlowska J, Lanikina KA, Domasevitch KV (2003) Polyhedron 22:1221
Gholivand K, Pourayoubi M, Shariatinia Z, Mostaanzadeh H (2005) Polyhedron 24:655
Gholivand K, Shariatinia Z (2007) Struct Chem 18:95
Gholivand K, Afshar F, Shariatinia Z, Zare K (2010) Struct Chem 21:629
Amirkhanov OV, Moroz OV, Znovjyak KO, Trush EA, Silva TY (2010) Acta Crystallogr E 66:o1102
Zhdanova LI, Byushkin VN, Malinovskii TI, Protsenko LD (1986) Proc Natl Acad Sci USSR 287:119
Zhdanova LI, Byushkin VN, Protsenko LD, Malinovskii TI (1986) Proc Natl Acad Sci USSR 288:1354
Rebrova ON, Byushkin VN, Malinovskii TI, Protsenko LD, Dneprova TN (1988) Proc Natl Acad Sci USSR 300:361
Rebrova ON, Byushkin VN, Malinovskii TI, Protsenko LD, Dneprova TN (1988) Proc Natl Acad Sci USSR 302:1108
Gholivand K, Mojahed F, Salehi M, Alizadehgan AM (2006) Anal Sci 22:x13
Rebrova ON, Byushkin VN, Malinovskii TI, Ovrutsky VM, Protsenko LD, Dneprova TN, Mazus MD (1992) Proc Natl Acad Sci USSR 324:103
Bruker APEX2, version 2009.7. Bruker SAINT, Version 7.66A. Bruker SADABS, Version 2009/1. Bruker XPREP, Version 2008/2. Bruker AXS, Inc. 5467 East Cheryl Parkway, Madison, WI 53711
Sheldrick GM (2008) Acta Crystallogr A 64:112
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cami R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, Revision E.01. Gaussian Inc., Wallingford, CT
Frisch A, Dennington RD, Keith TA, Millam J, Nielsen AB, Holder AJ, Hiscocks J (2007) Gauss View Version 4.1 User Manual. Gaussian Inc., Wallingford, CT
Lee CT, Yang WT, Parr RG (1988) Phys Rev B 37:785
Drago RS (1992) Physical methods for chemists, 2nd edn. Saunders College Publishing, Florida, p 254
Gholivand K, Pourayoubi M, Shariatinia Z (2007) Polyhedron 26:837
Gholivand K, Shariatinia Z, Pourayoubi M (2005) Z Anorg Allg Chem 631:961
Gholivand K, Mojahed F, Alizadehgan AM (2007) Pol J Chem 81:1829
Yazdanbakhsh M, Sabbaghi F, Boese R (2007) Z Kristallogr 222:331
Gholivand K, Mojahed F, Alizadehgan AM (2007) Pol J Chem 81:393
Gholivand K, Madani Alizadehgan A, Mojahed F, Soleimani P (2008) Polyhedron 27:1639
Gholivand K, Alizadehgan AM, Mojahed F, Firooz AA (2007) S Afr J Chem 60:91
Ovchynnikov VA, Amirkhanov VM, Timoshenko TP, Glowiak T, Kozlowski H (1998) Z Naturforsch B 53:481
Gholivand K, Della Védova CO, Erben MF, Mojahed F, Alizadehgan AM (2007) J Mol Struct 840:66
Gholivand K, Shariatinia Z, Mashhadi SM, Daeepour F, Farshidnasab N, Mahzouni HR, Taheri N, Amiri S, Ansar S (2009) Polyhedron 28:307
Gholivand K, Hosseini Z, Pourayoubi M, Shariatinia Z (2005) Z Anorg Allg Chem 631:3074
Gholivand K, Mostaanzadeh H, Shariatinia Z, Oroujzadeh N (2006) Main Group Chem 5:95
Gholivand K, Della Védova CO, Anaraki Firooz A, Madani Alizadehgan A, Michelini MC, Pis Diez R (2005) J Mol Struct 750:64
Gholivand K, Pourayoubi M, Mostaanzadeh H (2004) Anal Sci 20:x51
Mostaanzadeh H (2008) Anal Sci 24:x255
Rebrova ON, Byushkin VN, Malinovskii TI, Protsenko LD, Dneprova TN (1982) Proc Natl Acad Sci USSR 266:1391
Amirkhanov VM, Ovchynnikov VA, Glowiak T, Kozlowski H (1997) Z Naturforsch B 52:1331
Gubina KE, Ovchynnikov VA, Amirkhanov VM, Skopenko VV, Shishkin OV (2000) Z Naturforsch B 55:495
Sabbaghi F, Rostami Chaijan M, Pourayoubi M (2010) Acta Crystallogr E 66:o1754
Gholivand K, Mostaanzadeh H, Koval T, Dusek M, Erben MF, Della Védova CO (2009) Acta Crystallogr B 65:502
Gholivand K, Mojahed F, Madani Alizadehgan A, Bijanzadeh HR (2006) Z Anorg Allg Chem 632:1570
Dehghanpour S, Welter R, Hamady Barry A, Tabasi F (2008) Acta Crystallogr E 64:o633
Strasser CE, Sheng X, Safin DA, Raubenheimer HG, Luckay RC (2009) Acta Crystallogr E 65:o2926
Gholivand K, Shariatinia Z, Pourayoubi M, Farshadian S (2005) Z Naturforsch B 60:1021
Gholivand K, Shariatinia Z, Pourayoubi M (2006) Polyhedron 25:711
Gholivand K, Shariatinia Z, Pourayoubi M (2006) Z Anorg Allg Chem 632:160
Mahmoudi A, Hadjikhani R (2007) Z Kristallogr NCS 222:313
Mahmoudi A, Amin AH (2007) Acta Crystallogr E 63:o2031
Gubina KE, Amirkhanov VM (2000) Z Naturforsch B 55:1015
Rebrova ON, Byushkin VN, Zavodnik VE, Malinovskii TI, Protsenko LD, Dneprova TN (1985) Proc Natl Acad Sci USSR 280:1133
Bentrude WG, Setzer WN, Khan M, Sopchik AE, Ramli E (1991) J Org Chem 56:6127
Gholivand K, Pourayoubi M, Farshadian S, Molani S, Shariatinia Z (2005) Anal Sci 21:x55
Gholivand K, Della Védova CO, Erben MF, Mahzouni HR, Shariatinia Z, Amiri S (2008) J Mol Struct 874:178
Iriarte AG, Erben MF, Gholivand K, Jios JL, Ulic SE, Della Védova CO (2008) J Mol Struct 886:66
Thomas LC (1974) Interpretation of the infrared spectra of organophosphorus compounds. Heyden and Son, New York
Iriarte AG, Cutin EH, Erben MF, Ulic SE, Jios JL, Della Védova CO (2008) Vib Spectrosc 46:107
Acknowledgments
Support of this investigation by Ferdowsi University of Mashhad is gratefully acknowledged. The authors wish to thank Bruker AXS, Inc. (Madison, WI) for the use of one of their SMART X2S benchtop instruments.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tarahhomi, A., Pourayoubi, M., Rheingold, A.L. et al. Different orientations of C=O versus P=O in P(O)NHC(O) skeleton: the first study on an aliphatic diazaphosphorinane with a gauche orientation. Struct Chem 22, 201–210 (2011). https://doi.org/10.1007/s11224-010-9682-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-010-9682-y