Abstract
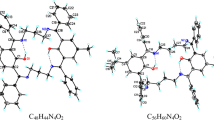
The structures of 8,8′-bis(3-phenylthioureidomethyl)-2,2′-binaphthalene (1), 8,8′-bis(3-butylthioureidomethyl)-2,2′-binaphthalene (2) and their complexes with anionic guests such as carboxylate ions (acetate, oxalate, malonate, succinate, glutarate, adipate, pimelate, suberate, and azelate), inorganic oxygen-containing anions (nitrate, sulfate, bicarbonate, hydrogen phosphate, and dihydrogen phosphate), and halide ions (fluoride, chloride, and bromide) were obtained using the ONIOM approach. The binding abilities of receptors 1 and 2 to anionic species in terms of binding energy, thermodynamic properties, and selectivity coefficient were obtained at the ONIOM(B3LYP/6-31G(d):AM1) and BSSE-corrected B3LYP/6-31G(d)//ONIOM(B3LYP/6-31G(d):AM1) levels of theory. The multipoint hydrogen bonding between receptors (either the receptor 1 or 2) and anionic guests were found. The hydrogen phosphate is the most preferable ion to bind with either the receptor 1 or 2.









Similar content being viewed by others
References
Kato R, Nishizawa S, Hayashita T, Teramae N (2001) Tetrahedron Lett 42:5053–5056. doi:10.1016/S0040-4039(01)00916-9
Lee DH, Lee KH, Hong J-I (2001) Org Lett 3:5–8. doi:10.1021/ol006690t
Lee DH, Lee HY, Lee KH, Hong J-I (2001) Chem Commun (Camb) 1188–1189. doi:10.1039/b102187g
Lee DH, Lee HY, Hong J-I (2002) Tetrahedron Lett 43:7273–7276. doi:10.1016/S0040-4039(02)01455-7
Jiménez D, Martínez-Máñnez R, Sancenón F, Soto J (2002) Tetrahedron Lett 43:2823–2825. doi:10.1016/S0040-4039(02)00363-5
Piatek P, Jurczak J (2002) Chem Commun (Camb) 20:2450–2451. doi:10.1039/b207335h
Gunnlaugsson T, Davis AP, O’Brien JE, Glynn M (2002) Org Lett 4:2449–2452. doi:10.1021/ol026004l
Fan A, Hong HK, Valiyaveettil S, Vittal JJ (2002) J Supramol Chem 2:247–254. doi:10.1016/S1472-7862(03)00079-0
Baerrigter H, Grave L, Nissink WM, Chrisstoffels LAJ, Vander Maas JH, Verboom W, de Jong F, Reindoudt DN (1998) Org Chem 63:4174–4180. doi:10.1021/jo972127l
Fan E, Van Arman SA, Kincaid D, Hamilton AD (1993) J Am Chem Soc 115:369–370. doi:10.1021/ja00054a066
Lee KH, Hong J-I (2000) Tetrahedron Lett 41:6083–6087. doi:10.1016/S0040-4039(00)00966-7
Wanno B, Rakrai W, Keawwangchai S, Morakot N, Morakot N, Nunthaboot N, Ruangpornvisuti V (2009) J Mol Struct THEOCHEM 902:33–40. doi:10.1016/j.theochem.2009.02.010
Ruangpornvisuti V (2004) J Mol Struct THEOCHEM 686:47–55. doi:10.1016/j.theochem.2004.08.007
Wanno B, Sang-aroon W, Tuntulani T, Pulpoka B, Ruangpornvisuti V (2003) J Mol Struct THEOCHEM 629:137–150. doi:10.1016/S0166-1280(03)00135-0
Ruangpornvisuti V (2004) J Mol Struct THEOCHEM 683:103–113. doi:10.1016/j.theochem.2004.06.022
Wanno B, Ruangpornvisuti V (2004) J Mol Struct THEOCHEM 685:57–71. doi:10.1016/j.theochem.2004.06.016
Wanno B, Ruangpornvisuti V (2006) J Mol Struct 787:76–89. doi:10.1016/j.molstruc.2005.11.006
Wanno B, Ruangpornvisuti V (2007) J Mol Model 13:65–77
Kondo S, Nagamine M, Yano Y (2003) Tetrahedron Lett 44:8801–8804. doi:10.1016/j.tetlet.2003.09.207
Kondo S, Sato M (2006) Tetrahedron 62:4844–4850. doi:10.1016/j.tet.2006.03.002
Becke AD (1988) Phys Rev A 38:3098–3100. doi:10.1103/PhysRevA.38.3098
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:385–789
Maseras F, Morokuma K (1995) J Comput Chem 16:1170–1179. doi:10.1002/jcc.540160911
Humbel S, Sieber S, Morokuma K (1996) J Chem Phys 105:1959–1967. doi:10.1063/1.472065
Dewar MJS, Reynolds CH (1986) J Comput Chem 2:140–143. doi:10.1002/jcc.540070206
Stewart JJP (1989) Comput Chem 10:209–220. doi:10.1002/jcc.540100208
Dapprich S, Komaromi I, Byun KS, Morokuma K, Frisch MJ (1999) J Mol Struct THEOCHEM 461:1–21. doi:10.1016/S0166-1280(98)00475-8
Remko M, Walsh OA, Richards WG (2001) J Phys Chem A 105:6926–6931. doi:10.1021/jp010637k
Remko M (2002) J Phys Chem A 106:5005–5010. doi:10.1021/jp020300y
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, Revision B.03, Gaussian, Inc., Pittsburgh, PA
Flükiger P, Lüthi HP, Portmann S, Weber J (2000) MOLEKEL 4.3: Swiss Center for Scientific Computing, Manno (Switzerland)
Boys SF, Bernadi F (1970) Mol Phys 19:553–566. doi:10.1080/00268977000101561
Mayer I, Surjan PR (1992) Chem Phys Lett 191:497–499. doi:10.1016/0009-2614(92)85415-7
Turi L, Dannenberg JJ (1993) J Phys Chem 97:2488–2490. doi:10.1021/j100113a002
Simon S, Duran M, Dannenberg JJ (1999) J Phys Chem A 103:1640–1643. doi:10.1021/jp9842188
Hamza A, Vibok A, Halasz GJ, Mayer I (2000) J Mol Struct THEOCHEM 427:501–502
Makowski M, Raczyska ED, Chmurzyski L (2001) J Phys Chem A 105:869–874. doi:10.1021/jp002458t
Long Y, Liu Y, Lei L, Nie L, Yao S (2001) Analyst (Lond) 126:1090–1094. doi:10.1039/b101478l
Acknowledgment
The authors gratefully acknowledge the Thailand Research Fund for financial support (Grant no. BRG5180016).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piyaauksornsak, S., Tangthongkul, T., Wanbayor, R. et al. Molecular structures of 8,8′-dithioureido-2,2′-binaphthalene derivatives and their anions recognition: an ONIOM investigation. Struct Chem 20, 767–780 (2009). https://doi.org/10.1007/s11224-009-9466-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-009-9466-4